Background: Lorlatinib, an anaplastic lymphoma kinase (ALK)–inhibitor, is approved as frontline as well as subsequent line of therapy in ALK-rearranged advanced non-small cell lung cancer (NSCLC). There is limited literature about safety and efficacy of lorlatinib in Indian patients.
Materials and methods: This was a retrospective multicentre study on patients with ALK-rearranged advanced NSCLC received lorlatinib as second line and beyond between May 2017 and December 2021. ALK was tested either by immunohistochemistry or fluorescent in-situ hybridisation. Clinicopathologic features, treatment details, toxicity and outcomes were analysed.
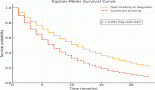
Results: A total of 38 patients were enrolled with a median age of 54 years (range: 30–72) and male: female ratio of 20:18. Fifteen (44%) patients had brain metastases at baseline. Lorlatinib use was – second line in 11 (29%), third line in 21 (55%) and fourth line in 4 (11%) of patients, respectively. The best radiologic response to lorlatinib was – complete response in 9 (24%), partial response in 17 (46%), stable disease in 9 (24%) and progressive disease in 2 (5%) of patients, respectively. After a median follow-up of 76.6 months (95% CI: 68.9–100), the median progression-free survival (PFS) of lorlatinib was not reached (95% CI: 24.3–not reached) and median overall survival (OS) of the whole cohort was 93.1 months (95% CI: 62–not reached). Both median PFS (p = 0.48) and median OS (p = 0.74) was similar between second line and later line use of lorlatinib. Thirty-three (87%) patients experienced treatment-related toxicity and six (16%) patients required dose modification.
Conclusion: Lorlatinib was highly efficacious in terms of overall response rate, median PFS and median OS in this small real-world cohort of advanced ALK+ve NSCLC with a manageable safety profile.