Introduction: In the earliest cases of COVID-19, a higher percentage of severe and fatal cases was observed in patients with cancer, including those with haematological malignancies. However, patients with chronic myeloid leukaemia (CML) had better prognoses, suggesting that tyrosine kinase inhibitors (TKIs) may have a therapeutic effect against SARS-CoV-2. This study describes the clinical and epidemiological characteristics of patients with CML receiving the TKIs tested for SARS-CoV-2 in Tegucigalpa, Honduras.
Methodology: An Analytical cross-sectional study was conducted. The sample included patients with Philadelphia chromosome-positive (Ph+) CML, who had been tested at least once for COVID-19 at the Emma Romero de Callejas Cancer Centre (CCERC). Sociodemographic and clinical variables were both analysed. Epi Info 7.2.4.0 and Stata/MP 16.0 were used to collect and analyse data. The COVID-19 positivity percentage and the association between severity and the TKI used were determined using Fisher’s exact test and odds ratio (OR). Data were gathered from clinical records with approval of CCERC institutional management.
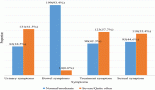
Results: One hundred and forty-nine patients with Ph+ CML were included; 20.1% were COVID-19-positive; 56% were male; mean age was 46 years; 81% were receiving imatinib, with a mean treatment duration of 6 years; 55% achieved a BCR -ABL molecular response ≤ 0.1% (IS). Twenty-one percent had comorbidities. COVID-19 was asymptomatic in 38.7% of patients, mild in 35.5% and severe in 9.7%. One patient died, a fatality rate of 3.2%. No statistical association was found between disease severity and treatment with imatinib versus second-line TKI (OR: 0.833, p: 0.8493, 95% CI: 0.098–10.998).
Conclusion: Despite high COVID-19 positivity in CML when compared with the literature, this study found a lower fatality rate. The type of TKI used or molecular response at the time of infection was not associated with case severity. Determining the effectiveness of imatinib or other TKIs as a COVID-19 treatment requires randomised clinical trials.