Molecular profile of liquid biopsies: next generation biomarkers to improve lung cancer treatment
Fabrizio Bianchi
Molecular Medicine Program, European Institute of Oncology, Milan 20141, Italy
Correspondence to: Fabrizio Bianchi. Email: fabrizio.bianchi@ieo.eu
Abstract
Molecular profiling of liquid biopsies is now emerging as pivotal for cancer biomarker discovery. The low-invasive nature of the approach used for collecting biospecimens (i.e. blood, urine, saliva, etc.) may allow a widespread application of novel molecular diagnostics based on liquid biopsies. This is relevant, for example, in cancer screening programmes where it is essential to reduce costs and the complexity of screening tests in order to increase study compliance and effectiveness. Here, I discuss recent advances in biomarkers for the early cancer detection and prediction of chemotherapy response based on the molecular profiling of liquid biopsies.
Keywords: biomarkers, circulating biomarkers, circulating tumour DNA, lung cancer, microRNA
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Published: 24/11/2015; Received: 07/04/2015
Introduction
Despite significant improvements in the detection and treatment of cancer made in recent decades, which have had a positive impact on the overall survival of cancer patients [1], the prognosis of patients with advanced cancer (i.e. with regional or distant metastases) remains poor [1]. Lung cancer offers a prototypical example, while patients diagnosed with early-stage lung tumours (stage I–II) have a 5-year survival rate of ~40–50% [1], and up to ~90% for computerised tomography (CT)-detected stage-I tumour [2], the 5-year relative survival rate drops to 4–26% when the cancer is diagnosed at an advanced stage (~85% of all cases) [1]. The unfavourable prognosis of patients with advanced lung cancer is due to the metastatic spread of cancer cells to the regional (lymph nodes) or distant organs, which cannot be completely eradicated by surgery or multimodal therapy. In some cases, neoadjuvant chemotherapy (given before surgery) helps to reduce tumour size and to eradicate micrometastases in order to facilitate surgery in patients with stage-IIIA lung cancer [3]. However, patients treated with conventional chemotherapy (i.e. platinum based) frequently develop resistance to therapy due to the selection of molecular alterations in tumour cells, which make them resistant to most of the available chemotherapeutic agents [4]. For this reason, several investigators now focus their studies on the identification of cancer gene mutations, aimed at characterizing the molecular mechanisms that underlie chemoresistance, which may help to identify more effective therapeutic strategies for chemorefractant cancer [5].
With the advent of next-generation sequencing (NGS), which allows simultaneous sequencing of all coding genes in the human genome in a few days, it is now possible to deeply sequence hundreds of tumours to map the mutational landscape of cancer including mutations possibly involved in chemoresistance [6, 7]. Results from these studies are expected to change the way that we treated patients with cancer: from the concept of ‘one drug fits all’ to the concept of ‘personalised medicine’ using molecular targeted drugs. However, the successful treatment of neoplastic diseases is hindered by the complexity of the cancer genome which was confirmed by recent NGS studies [8–11]. The molecular complexity of cancer augments with disease progression, favoured by the acquisition of genomic instability in cancer [10, 12, 13]. In such scenario, new methods to anticipate diagnosis of cancer may increase the frequency of early diagnosis when surgery and chemotherapy are more effective. Recent results from large trials for the early detection of lung cancer showed a 20% reduction of mortality in individuals who underwent screening [14]. However, the number of screened individuals required to detect one lung cancer patient is still too high (~1 patient out of 100 screened individuals) and the high false-positive rate in CT screening (most of the lung nodules detected are benign, ~96%) represents a limit to the widespread applicability of screening programmes [14]. Thus, there is an urgent need to identify additional epidemiologic risk factors (other than age, smoking status) and cancer biomarkers to better identify the high-risk population, thus reducing the number of unnecessary exams.
Circulating microRNA as new valuable biomarkers for lung cancer screening
The molecular profile of liquid biopsies (i.e. blood, urine, saliva) is an attractive field for cancer biomarkers discovery, both for the relative low invasivity of the procedure for collecting material and for the possibility to obtain multiple samples from the same patients at different times. Recently, the variety of circulating molecules analysable in blood samples has been further expanded with the discovery of circulating free-RNA and, in particular, of a specific class of small RNA called microRNAs that are remarkably stable in body fluids [15–17]. MicroRNAs (miRNAs) are short non-coding RNA molecules function as endogenous triggers of the mRNA interference pathway and are involved in the regulation of many cellular processes, including differentiation, proliferation and apoptosis [17, 18]. The expression of miRNAs is often deregulated in human tumours, both in a tissue- and in a cancer-specific manner [19]. Importantly, fluctuations of circulating miRNAs were shown to be associated with many malignant and non-malignant diseases, including lung cancer [15, 20, 21]. Contrary to circulating tumour-DNA (ct-DNA), where there are evidences that only a fraction of early stages tumours may release sufficient quantities of circulating DNA to be detected in the blood [22], circulating free-miRNAs (cf-miRNA) appear to be excellent candidates for bloodborne tumour markers for the early diagnosis of different tumours [23, 24].
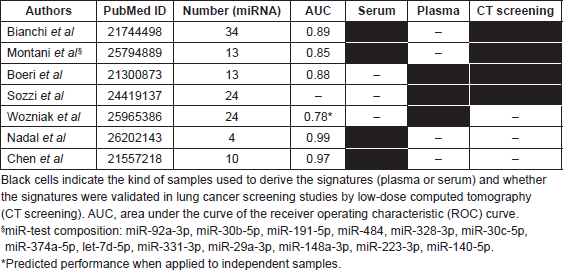
In line with this, our group has recently characterised cf-miRNA signatures that correlate with the presence of asymptomatic, early stage, lung cancer [25, 26]. We initially analysed cf-miRNA in serum samples from 174 individuals enrolled in the COSMOS (Continuing Observation of SMOking Subjects) trial (ClinicalTrials.gov Identifier: NCT01248806 [27]). This trial assessed the efficacy of annual LDCT scans in detecting early-stage lung cancer in over 5000 high-risk individuals and validated simple and non-invasive guidelines for the diagnostic work up of screen-detected lung nodules [27, 28]. From the miRNA analysis, we derived a 34 serum cf-miRNA signature (Table 1) that can identify individuals with asymptomatic, early stage, lung cancer (AUC = 0.89, p < 0.0001) [25]. We used this signature to develop a diagnostic test, the miR-test (Table 1), which confirmed an AUC of 0.85, accuracy of 74.9%, sensitivity of 77.8% and specificity of 74.8% in an additional independent cohort of 1115 high-risk individuals from the COSMOS trial [29]. Another study, COSMOS 2, is ongoing and is screening by CT and miR-test ~10.000 high-risk subjects from eight different health institutions. Results of this study will confirm whether miR-test may better identify high-risk candidates for low-dose CT screening, thus reducing screening costs and augment feasibility. Initial results of the screening will be available in 2017.
Other cf-miRNA signatures for lung cancer early detection have recently been described [26, 30–33], though few of them have been validated in lung cancer screening studies. Almost half of these signatures were discovered by analysing serum samples, while the others were derived from plasma samples (Table 1). There are differences in both quantities and species of cf-miRNAs when analysing serum or plasma samples due to differences in the chemical composition and in the technical preparation of these two biological specimens [34, 35]. This should be taken into account when searching for overlapping cf-miRNA in the different studies or during validation of cf-miRNA biomarkers using external cohorts. However, Wozniak et al. recently described an analysis of multiple cf-miNA signatures derived both from serum (including our 34-miRNA signature) and plasma samples, in a case–control study of 100 lung cancer patients and 100 non-cancer controls, whose plasma samples were profiled by TaqMan Human MicroRNA Array [33]. Interestingly, our 34-miRNA serum signature performed well in this case–control cohort of plasma samples (AUC = 0.78) that suggests the existence of a core of cf-miRNAs which quantities and species do not vary significantly in the serum or plasma samples.
Circulating tumour-DNA mutational screening can identify actionable mutations for lung cancer treatment
Pioneer studies demonstrated the presence of circulating free DNA (cf-DNA) in the blood and their potential applicability for screening pathological conditions including cancer [36, 37]. Subsequent studies further analysed cf-DNA and demonstrated that part of these DNA may be specifically released by cancer cells (circulating tumour DNA, ct-DNA) [38], thus corroborating the use of ct-DNA as a cancer biomarker [39–41]. An advantage of screening ct-DNA in the blood rather than circulating proteins (e.g. PSA, CA125, CEA) is that the former can be checked also for tumour-specific mutations which dramatically increase the specificity of a blood test for cancer diagnosis and monitoring. Importantly, ct-DNA screening was shown to be effective to determine EGFR mutation status in patients with metastatic lung cancer who acquired resistance to anti-EGFR tyrosine kinase inhibitors (TKI) [42–44]. For example, the acquisition of the EGFR T790M resistance mutation was shown to be traceable in ct-DNA before treatment with anti-EGFR TKI [45], which suggests that ct-DNA screening may allow the anticipation of third-generation anti-EGFR TKIs in the case of acquired resistance to first-line anti-EGFR TKI therapy (gefitinib, afatinib, erlotinib). However, the sensitivity of this assay across different tumour types and stages still represents a concern that should be addressed before clinical application [46].
Table 1. Published cf-miRNA signatures for lung cancer early detection.
In addition, the recent application of NGS in screening cf-DNA allowed for searching known or novel mutations in an ‘unsupervised’ manner, which eliminates the requirement of a prior knowledge of relevant mutations thus expanding the possibility of identifying novel mutations involved in cancer progression and chemoresistance [47]. An important application of NGS for screening ct-DNA was recently proposed by Newman et al, where a relatively inexpensive, yet highly sensitive, method called CAPP-Seq was developed to screen for 521 exons and 13 introns from a total of 139 mutated genes [22]. The method was sensitive enough to monitor treatment response in patients with lung cancer and to predict mutations possibly responsible for chemoresistance [22].
Conclusion
I envision that the molecular analysis of liquid biopsies will become the gold standard for developing non-invasive tests for personalised medicine. Through a ‘simple’ blood test clinicians will be able to anticipate diagnosis of cancer thus augmenting the probability of curative treatments for patients with cancer. In addition, the possibility to collect multiple samples (even in the case of metastatic disease) from the same patient will help to better monitor the therapeutic response, and to aid oncologists to anticipate second line (molecularly targeted) chemotherapy, in the case of acquired chemotherapy resistance. It will also be essential to overcome the current technological limitations which impede the large-scale application of these molecular tests. Indeed, the extraction and analysis of circulating nucleic acids require expensive and complex equipment that is still available only in few genomics laboratories worldwide. Furthermore, the sensitivity of these molecular tests should be improved in order to augment the detection rate and avoid false-negative results that are detrimental for cancer screening programmes. The possibility to analyse circulating biomarkers directly in the blood without sample pre-processing and using point-of-care health diagnostic technologies will favour massive application of such molecular tests.
Acknowledgments
I would like to thank Ugo Cavallaro for the numerous insightful scientific discussions and for his help in preparing this manuscript. This work was supported by grants from the DOD (FY13) Lung Cancer Research Program (LCRP) Clinical Exploration Award (LC130638) and from the Italian Association for Cancer Research (AIRC; IG12818). This work was presented at the WP2 EurocanPlatform meeting: European recommendations for biomarker-based chemoprevention trials, October 13–14, 2014, at the European Institute of Oncology, Milan, Italy.
References
1. Siegel R, Ma J and Zou Z et al (2014) Cancer statistics CA Cancer J Clin 64 9–29 DOI: 10.3322/caac.21208 PMID: 24399786
2. International Early Lung Cancer Action Program, Henschke CI and Yankelevitz DF et al (2006) Survival of patients with stage I lung cancer detected on CT screening N Engl J Med 355 1763–1771 DOI: 10.1056/NEJMoa060476 PMID: 17065637
3. Gilligan D, Nicolson M and Smith I et al (2007) Preoperative chemotherapy in patients with resectable non-small cell lung cancer: results of the MRC LU22/NVALT 2/EORTC 08012 multicentre randomised trial and update of systematic review Lancet 369 1929–1937 DOI: 10.1016/S0140-6736(07)60714-4 PMID: 17544497
4. Seve P and Dumontet C (2005) Chemoresistance in non-small cell lung cancer Curr Med Chem Anticancer Agents 5 73–88 DOI: 10.2174/1568011053352604 PMID: 15720263
5. Lonning PE and Knappskog S (2013) Mapping genetic alterations causing chemoresistance in cancer: identifying the roads by tracking the drivers Oncogene 32 5315–5330 DOI: 10.1038/onc.2013.48 PMID: 23474753
6. Cancer Genome Atlas Research N (2012) Comprehensive genomic characterization of squamous cell lung cancers Nature 489 519–525 DOI: 10.1038/nature11404 PMID: 22960745 PMCID: 3466113
7. Cancer Genome Atlas Research N (2014) Comprehensive molecular profiling of lung adenocarcinoma Nature 511 543–550 DOI: 10.1038/nature13385 PMID: 25079552 PMCID: 4231481
8. Gerlinger M, Rowan AJ and Horswell A et al (2012) Intratumor heterogeneity and branched evolution revealed by multiregion sequencing N Engl J Med 366 883–892 DOI: 10.1056/NEJMoa1113205 PMID: 22397650
9. Navin N, Kendall J and Troge J et al (2011) Tumour evolution inferred by single-cell sequencing Nature 472 90–94 DOI: 10.1038/nature09807 PMID: 21399628 PMCID: 4504184
10. de Bruin EC, McGranahan N and Mitter R et al (2014) Spatial and temporal diversity in genomic instability processes defines lung cancer evolution Science 346 251–256 DOI: 10.1126/science.1253462 PMID: 25301630
11. Zhang J, Fujimoto J and Zhang J et al (2014) Intratumor heterogeneity in localized lung adenocarcinomas delineated by multiregion sequencing Science 346 256–259 DOI: 10.1126/science.1256930 PMID: 25301631 PMCID: 4354858
12. Lee AJ, Endesfelder D and Rowan AJ et al (2011) Chromosomal instability confers intrinsic multidrug resistance Cancer Res 71 1858–1870 DOI: 10.1158/0008-5472.CAN-10-3604 PMID: 21363922 PMCID: 3059493
13. Sotillo R, Schvartzman JM and Socci ND et al (2010) Mad2-induced chromosome instability leads to lung tumour relapse after oncogene withdrawal Nature 464 436–440 DOI: 10.1038/nature08803 PMID: 20173739 PMCID: 2841716
14. Aberle DR, Adams AM and Berg CD et al (2011) Reduced lung-cancer mortality with low-dose computed tomographic screening N Engl J Med 365 395–409 DOI: 10.1056/NEJMoa1102873 PMID: 21714641 PMCID: 4356534
15. Chen X, Ba Y and Ma L et al (2008) Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases Cell Res 18 997–1006 DOI: 10.1038/cr.2008.282 PMID: 18766170
16. Schwarzenbach H, Hoon DS and Pantel K (2011) Cell-free nucleic acids as biomarkers in cancer patients Nat Rev Cancer 11 426–437 DOI: 10.1038/nrc3066 PMID: 21562580
17. Yendamuri S and Kratzke R (2011) MicroRNA biomarkers in lung cancer: MiRacle or quagMiRe? Transl Res 157 209–215 DOI: 10.1016/j.trsl.2010.12.015 PMID: 21420031
18. Krol J, Loedige I and Filipowicz W (2010) The widespread regulation of microRNA biogenesis, function and decay Nat Rev Genet 11 597–610 PMID: 20661255
19. Volinia S, Calin GA and Liu CG et al (2006) A microRNA expression signature of human solid tumors defines cancer gene targets Proc Natl Acad Sci USA 103 2257–2261 DOI: 10.1073/pnas.0510565103 PMID: 16461460 PMCID: 1413718
20. Shen J, Liu Z and Todd NW et al (2011) Diagnosis of lung cancer in individuals with solitary pulmonary nodules by plasma microRNA biomarkers BMC cancer 11 374 DOI: 10.1186/1471-2407-11-374 PMID: 21864403 PMCID: 3175224
21. Hu Z, Chen X and Zhao Y et al (2010) Serum microRNA signatures identified in a genome-wide serum microRNA expression profiling predict survival of non-small-cell lung cancer J Clin Oncol 28 1721–1726 DOI: 10.1200/JCO.2009.24.9342 PMID: 20194856
22. Newman AM, Bratman SV and To J et al (2014) An ultrasensitive method for quantitating circulating tumor DNA with broad patient coverage Nat Med 20 548–554 DOI: 10.1038/nm.3519 PMID: 24705333 PMCID: 4016134
23. Lin PY and Yang PC (2011) Circulating miRNA signature for early diagnosis of lung cancer EMBO Mol Med 3 436–437 DOI: 10.1002/emmm.201100155 PMID: 21739607 PMCID: 3377090
24. Madhavan D, Cuk K and Burwinkel B (2013) Cancer diagnosis and prognosis decoded by blood-based circulating microRNA signatures Front Genet 4 116 DOI: 10.3389/fgene.2013.00116 PMID: 23802013 PMCID: 3689027
25. Bianchi F, Nicassio F and Marzi M et al (2011) A serum circulating miRNA diagnostic test to identify asymptomatic high-risk individuals with early stage lung cancer EMBO Mol Med 3 495–503 DOI: 10.1002/emmm.201100154 PMID: 21744498 PMCID: 3377091
26. Boeri M, Verri C and Conte D et al (2011) MicroRNA signatures in tissues and plasma predict development and prognosis of computed tomography detected lung cancer Proc Natl AcadSci USA 108 3713–3718 DOI: 10.1073/pnas.1100048108
27. Veronesi G, Bellomi M and Mulshine JL et al (2008) Lung cancer screening with low-dose computed tomography: a non-invasive diagnostic protocol for baseline lung nodules Lung Cancer 61 340–349 DOI: 10.1016/j.lungcan.2008.01.001 PMID: 18308420
28. Pelosi G, Sonzogni A and Veronesi G et al (2008) Pathologic and molecular features of screening low-dose computed tomography (LDCT)-detected lung cancer: a baseline and 2-year repeat study Lung Cancer 62 202–214 DOI: 10.1016/j.lungcan.2008.03.012 PMID: 18450320
29. Montani F, Marzi M and Dezi F et al (2015) miR-Test: a blood test for lung cancer early detection JNCI DOI: 10.1093/jnci/djv063 PMID: 25794889
30. Chen X, Hu Z and Wang W et al (2012) Identification of ten serum microRNAs from a genome-wide serum microRNA expression profile as novel noninvasive biomarkers for nonsmall cell lung cancer diagnosis Int J Cancer 130 1620–1628 DOI: 10.1002/ijc.26177
31. Nadal E, Truini A and Nakata A et al (2015) A novel serum 4-microRNA signature for lung cancer detection Sci Rep 5 12464 DOI: 10.1038/srep12464 PMID: 26202143
32. Sozzi G, Boeri M and Rossi M et al (2014) Clinical utility of a plasma-based miRNA signature classifier within computed tomography lung cancer screening: a correlative MILD trial study J Clin Oncol 32 768–773 DOI: 10.1200/JCO.2013.50.4357 PMID: 24419137
33. Wozniak MB, Scelo G and Muller DC et al (2015) Circulating microRNAs as non-invasive biomarkers for early detection of non-small-cell lung cancer PLoS One 10 e0125026 DOI: 10.1371/journal.pone.0125026 PMID: 25965386 PMCID: 4428831
34. Heegaard NH, Schetter AJ and Welsh JA et al (2012) Circulating micro-RNA expression profiles in early stage non-small-cell lung cancer Int J Cancer 130 1378–1386 DOI: 10.1002/ijc.26153 PMCID: 3259258
35. Wang K, Yuan Y and Cho JH et al (2012) Comparing the microRNA spectrum between serum and plasma PloS one 7 e41561 DOI: 10.1371/journal.pone.0041561 PMID: 22859996 PMCID: 3409228
36. Mandel P and Metais P (1948) Les acides nucleiques du plasma sanguin chez l’homme [in French] C R Seances Soc Biol Fil 142 241–243 PMID: 18875018
37. Girardi AJ, Sweet BH and Slotnick VB (1962) Development of tumors in hamsters inoculated in the neonatal period with vacuolating virus, SV-40 Proc Soc Exp Biol Med 109 649–660 DOI: 10.3181/00379727-109-27298 PMID: 13898881
38. Leon SA, Shapiro B and Sklaroff DM et al (1977) Free DNA in the serum of cancer patients and the effect of therapy Cancer Res 37 646–650 PMID: 837366
39. Vasioukhin V, Anker P and Maurice P et al (1994) Point mutations of the N-ras gene in the blood plasma DNA of patients with myelodysplastic syndrome or acute myelogenous leukaemia Br J Haematol 86 774–779 DOI: 10.1111/j.1365-2141.1994.tb04828.x PMID: 7918071
40. Sorenson GD, Pribish DM and Valone FH et al (1994) Soluble normal and mutated DNA sequences from single-copy genes in human blood Cancer Epidemiol Biomarkers Prev 3 67–71 PMID: 8118388
41. Misale S, Yaeger R and Hobor S et al (2012) Emergence of KRAS mutations and acquired resistance to anti-EGFR therapy in colorectal cancer Nature 486 532–536 PMID: 22722830 PMCID: 3927413
42. Karachaliou N, Mayo-de Las Casas C and Queralt C et al (2015) Association of EGFR L858R mutation in circulating free DNA with survival in the EURTAC trial JAMA Oncol 1 149–157 DOI: 10.1001/jamaoncol.2014.257 PMID: 26181014
43. Luo J, Shen L and Zheng D (2014) Diagnostic value of circulating free DNA for the detection of EGFR mutation status in NSCLC: a systematic review and meta-analysis Sci Rep 4 6269 DOI: 10.1038/srep06269 PMID: 25201768
44. Douillard JY, Ostoros G and Cobo M et al (2014) Gefitinib treatment in EGFR mutated caucasian NSCLC: circulating-free tumor DNA as a surrogate for determination of EGFR status J Thorac Oncol 9 1345–1353 DOI: 10.1097/JTO.0000000000000263 PMID: 25122430 PMCID: 4224589
45. Maheswaran S, Sequist LV and Nagrath S et al (2008) Detection of mutations in EGFR in circulating lung-cancer cells N Engl J Med 359 366–377 DOI: 10.1056/NEJMoa0800668 PMID: 18596266 PMCID: 3551471
46. Bettegowda C, Sausen M and Leary RJ et al (2014) Detection of circulating tumor DNA in early- and late-stage human malignancies Sci Transl Med 6 224ra24 DOI: 10.1126/scitranslmed.3007094 PMID: 24553385 PMCID: 4017867
47. Murtaza M, Dawson SJ and Tsui DW et al (2013) Non-invasive analysis of acquired resistance to cancer therapy by sequencing of plasma DNA Nature 497 108–112 DOI: 10.1038/nature12065 PMID: 23563269