The role of the robotic technique in minimally invasive surgery in rectal cancer
Paolo Pietro Bianchi1, Fabrizio Luca2, Wanda Petz1, Manuela Valvo2, Sabine Cenciarelli3, Massimiliano Zuccaro3 and Roberto Biffi3
1 Unit of Minimally Invasive Surgery, European Institute of Oncology, 20141 Milano, Italy
2 Unit of Integrated Abdominal Surgery, European Institute of Oncology, 20141 Milano, Italy
3 Division of Abdomino-Pelvic and Minimally-Invasive Surgery, European Institute of Oncology, 20141 Milano, Italy
Correspondence to: Roberto Biffi. Email: roberto.biffi@ieo.it
Abstract
Laparoscopic rectal surgery is feasible, oncologically safe, and offers better short-term outcomes than traditional open procedures in terms of pain control, recovery of bowel function, length of hospital stay, and time until return to working activity. Nevertheless, laparoscopic techniques are not widely used in rectal surgery, mainly because they require a prolonged and demanding learning curve that is available only in high-volume and rectal cancer surgery centres experienced in minimally invasive surgery. Robotic surgery is a new technology that enables the surgeon to perform minimally invasive operations with better vision and more intuitive and precise control of the operating instruments, promising to overcome some of the technical difficulties associated with standard laparoscopy. The aim of this review is to summarise the current data on clinical and oncological outcomes of minimally invasive surgery in rectal cancer, focusing on robotic surgery, and providing original data from the authors’ centre.
Keywords: rectal cancer, total mesorectal excision, laparoscopic surgery, robotic surgery.
Introduction
The most important advance in rectal cancer surgery over the last 30 years was the development, affirmation, and standardisation of total mesorectal excision (TME) [1]. TME is associated with reduction in local recurrence rate to <10% and improved survival in some series [2]. TME aims to completely remove the rectum with its perirectal fat, at the same time maintaining the integrity of the mesorectal fascia and preserving the autonomic nerves.
The available literature on minimally invasive TME for rectal cancer, as illustrated by the findings in a recent meta-analysis of randomised control trials [3], demonstrates that laparoscopic TME is feasible and associated with better short-term outcomes than open surgery, while maintaining equivalent oncologic safety. Nevertheless, laparoscopic TME for rectal cancer is not widely practiced [4–7]. The main reasons for this are the prolonged and demanding learning curve, and the technically challenging nature of the operations themselves, related to the lack of adequate space within the pelvis and difficulty of manoeuvring the rigid and cumbersome laparoscopic instruments [8]. It has been estimated that over 50 operations [9] are required for an experienced colorectal surgeon to achieve proficiency with minimally invasive techniques and such experience can only be obtained at high-volume centres. One way of overcoming this limitation would be to make minimally invasive operations technically easier, thereby reducing learning time and increasing the attractiveness of minimally invasive approaches for young surgeons [10–12].
Robotic surgery is a sophisticated new technology that improves the traditional laparoscopic surgery in different ways: it has high-definition three-dimensional vision, it translates the surgeon’s hand movements into precise movements of the instruments inside the patient, the camera is held and moved by the first surgeon, and a fourth robotic arm is available as a fixed retractor. All these aspects increase surgeon comfort and reduce fatigue [13]. The aim of this paper is to review the available data on robotic surgery for rectal cancer in comparison with traditional laparoscopic technique.
Laparoscopic surgery in rectal cancer
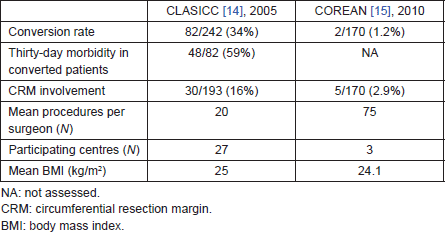
A minimally invasive approach to TME has been shown to be feasible and oncologically safe in several trials, the two largest being the CLASICC trial [14], which recruited colon and rectal cancer patients, and the COREAN [15] trial, which recruited rectal cancer patients only. There was some concern about the higher rate of circumferential resection margin (CRM) involvement among rectal cancer patients undergoing low anterior resection in the laparoscopic arm (12.4%) of the CLASICC trial compared with the open arm (6.3%). This was probably due to the greater technical difficulty associated with the laparoscopic approach for rectal cancer—as indicated by higher conversion rate in the rectal laparoscopic than colon laparoscopic subgroup (34% versus 25%). However, in this study, the difference in CRM involvement did not translate into a difference in five-year disease-free survival between laparoscopically assisted and open surgery [16].
Furthermore, in the CLASICC trial, morbidity and mortality rates were high among laparoscopic cases (colon and rectal cancer) converted to open surgery. Thirty-day morbidity was 29% in laparoscopic cases and 45% in converted cases; in-hospital mortality was 1% in laparoscopic cases and 9% in converted cases. Some of this extra morbidity could have been due to more advanced cancers requiring conversion, but the longer operating times, greater technical difficulty, and need for a wide open incision in the converted cases, almost certainly contributed to these findings. Conversion to open surgery can also result in greater local recurrence rate [17]. Finally, participation in the CLASICC trial required that involved surgeons had to have performed at least 20 previous laparoscopic colorectal resections; it is possible that many operators were still in the learning phase of their activity at the time when the CLASICC study started (Table 1).
The COREAN trial was designed to assess the safety and efficacy of laparoscopic surgery in comparison with open surgery for cT3N0-2 mid and low rectal cancer without distant metastasis after preoperative chemoradiation therapy. The seven surgeons who contributed to this trial had done a median of 75 previous laparoscopic colorectal resections, and the three hospitals involved were high-volume centres, with over 200 rectal cancers treated annually. The trial found no differences for CRM positivity or TME quality, but documented short-term advantages for laparoscopic surgery over open one. The conversion rate was 1.2% (2/170). The lack of cT4 cases, the lower mean body mass index of patients (24.1 kg/m² versus CLASICC 26 kg/m2), and greater surgeon experience matured in high-volume centres, may all have contributed to low conversion rate and good oncologic results of the COREAN study (Table 1).
Table 1. Main differences in results between CLASICC and COREAN trials.
Notwithstanding these findings, laparoscopic rectal resection is still a challenging procedure, even in expert hands [18]. The recent guidelines of the European Association for Endoscopic Surgery recommend that it should be performed only by expert surgeons at high-volume centres and in selected patients (cT4 cases should be excluded); if these conditions are met, the laparoscopic approach can be an acceptable alternative treatment option to conventional open surgery [19], maintaining its well-established advantages (less pain, less blood loss, shorter hospital stay, and better quality of life in the early postsurgical period).
Technical aspects of minimally invasive TME
Laparoscopic low rectal resection is second only to reversal of Hartmann’s procedure in terms of colorectal operation difficulty [8]. The main problems are the lack of space within the rigid pelvis and the small operating field, so that exposure of the lateral and anterior rectal wall in particular is hindered. Furthermore, the view with the laparoscopic camera is often obstructed by smoke from cautery instruments, and a good view of the operating field often depends on the skill of the camera assistant, who must be experienced, thoroughly familiar with all the steps of the procedure, and also have an excellent relationship with the operating surgeon, understanding where he/she wants to go and what he/she wants to see. The robotic technology seems to offer some advantages to overcome the above-mentioned technical difficulties of laparoscopic procedure.
The only robotic system so far available is the da Vinci Surgical System (Intuitive Surgical, Mountain View, California, United States). This consists of a console, some distance from the patient where the surgeon sits while operating, and the robotic cart with its three or four robotic arms, which is in close proximity to the patient. Various instruments, attached to the robotic arms, are inserted into the patient through small incisions. The console provides a magnified high-definition three-dimensional view of the operating field through a stereo-endoscope fitted to one of the robotic arms, whose visual field and angle of vision are adjustable.
The surgeon manipulates two-finger joysticks (finger and wrist movements) at the console while watching the surgical field through the visual system. Movements of the surgeon’s hands are transmitted through the robot to the robotic arms, so that equivalent but scaled movements of the surgical instruments are made within the surgical field. The instruments have seven degrees of movement freedom and hand tremor is filtered out [13, 20]. The da Vinci system appears able to overcome many of the problems that characterise laparoscopic rectal resection. Initially, the three-dimensional high-definition camera is under the direct control of the surgeon, and the double optical system makes consistently clear vision more likely. Furthermore, the tremor-filtered, multiple-degree-of-movement instruments, which can be scaled relative to the surgeon’s hand movements, permit more precise dissection in the narrow space and render the more difficult steps of the operation easier. Tissue retraction with the robot is also easier, since the third arm can be used as a fixed retractor, and is always manipulated by surgeon (Figure 1) [21]. These potential advantages within the narrow space of the pelvis have been shown to be translated into concrete advantages for robotic radical prostatectomy [22], and in our opinion the excellent ergonomics and precise dissection offered by the robotic system can be helpful in colorectal surgery also for vessel ligation and flexure mobilisation. This is why we have standardised a ‘fully robotic’ technique for left colon and rectal resection [23].
Literature review
Robotic TME has been introduced mainly in centres with a high volume of rectal cancer treated per year and by surgeons who are experts in minimally invasive surgery [24–38]. The published studies are small, not randomised, single centre series; despite the low evidence of data available, five meta-analyses [39–43] have already been published on this technique, indicating the marked interest in robotic surgery for rectal cancer. In the published series, the robotic technique is not standardised, rectal resection can be performed with a full robotic technique or using a hybrid procedure with some steps laparoscopy performed, but in all the studies, the TME step was performed robotically [24–28, 30–38]. Overall, the published studies are mainly retrospective or prospective single centre series, comparing mainly robotic rectal resection to laparoscopy, but recently some studies comparing robotic rectal resection with open surgery have also been published [44–46].
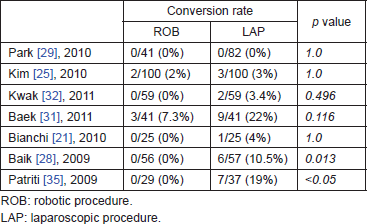
In comparison with standard laparoscopy, the most important finding for robotic rectal surgery is the lower rate of conversion to open surgery. In the meta-analysis in [39], robotic rectal resection was associated with a significant reduction of conversion rate in comparison with laparoscopy. In the meta-analysis in [40], conversion rates and estimated blood loss were significantly lower in robotic rectal cancer patients than in rectal cancer patient undergoing conventional laparoscopic resection (Table 2). The meta-analysis in [41] also reported a significantly lower conversion rate among robotic patients.
Conversion to open surgery is an important indicator of proficiency, generally being ~10–15% in laparoscopic rectal surgery, except for the COREAN trial, where the conversion rate was 1.1%. A preliminary publication of the COLOR II trial reported a 17% conversion rate to open surgery [47]. An RCT [robotic versus laparoscopic resection for rectal cancer (ROLARR)]—comparing laparoscopic with robotic rectal resection for cancer—is ongoing, and its first end-point is the conversion rate to open surgery [48].
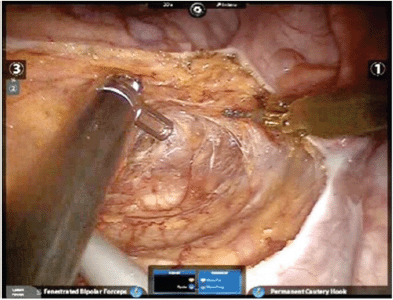
Figure 1. Robotic TME.
Table 2. Conversion rate in robotic and laparoscopic surgery for rectal cancer.
Learning for minimally invasive surgery is a crucial issue, because laparoscopic colorectal surgery has a steep learning curve, particularly for rectal cancer, and robotic surgery seems to reduce the learning curve to 15/20 cases, as calculated in [49], or even avoided, as reported in [50].
In our Institute (European Institute of Oncology, Milano, Italy) three senior consultants approached the robotic technique in rectal surgery. One of them was highly experienced in laparoscopic surgery with >100 laparoscopic colorectal resections. In 90 cases of robot-assisted rectal resections performed by the expert laparoscopic team, the operative time did not change, as well as leakage (4%) and conversion rates (2%), thus confirming the data from the clinical series in [50]. The other two surgeons had a solid experience in open colorectal surgery but were less expert in laparoscopy; nevertheless, the learning curve was brief (ten cases for each surgeon) and the operative time reduced to <240 min in a short span of time [51]. Therefore, it is possible to assume that for an experienced colorectal surgeon the impressive facilitation of the performance of complex laparoscopic procedures offered by the robotic system does not imply to complete an advanced laparoscopy learning period. The overall series of the IEO group is presented in Table 3.
Clinical and oncological outcomes
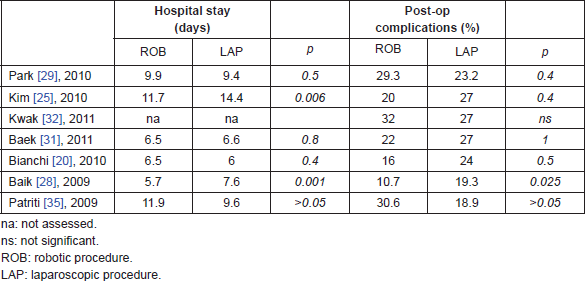
Looking at short-term clinical outcomes, robotic surgery did not show significant differences from laparoscopic technique in all the analyzed studies. Overall complications rate and length of hospital stay are similar for both techniques, and this can be easily explained because the operation performed robotically is the same than in laparoscopy, with the aid of a better technology. In comparison with open surgery, robot-assisted surgery maintains all the advantages of minimally invasive procedures [44–46] (Table 4).
Table 3. Personal series: results of robotic resection for distal rectal cancer.
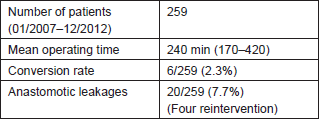
Table 4. Short-term clinical outcomes of robotic and laparoscopic surgery for rectal cancer.
Few data are available on sexual and urinary dysfunctions, which is a crucial issue to be evaluated, because laparoscopy did not add advantages in term of sexual/urinary dysfunctions preservation [52, 53]. Moreover, the MRC CLASICC trial raised a concern that laparoscopic TME may be associated with increased rates of male sexual dysfunction compared with those of conventional open TME [54]. Otherwise, in robotic prostate resections, a better preservation of genito/urinary functions compared with open or laparoscopic procedure has been demonstrated [22]. To date, only three studies—evaluating genitourinary function after robotic rectal surgery—have been published [55–57]; although different methods and questionnaires have been used by the authors for assessment of this topic, there are some converging evidences in all these studies. A similar trend for sexual function has been observed, with a decrease at one month after surgery and subsequent progressive recovery, and all the authors agree that an easier identification of the nerves and of the planes of dissection is provided by the robotic system (Table 5).
Further research is needed to provide definitive scientific evidence that enhanced precision given by the robotic system permits better preservation of the hypogastric plexus and therefore better functional results. We should also fill the gap in our knowledge about sexual function and quality of life in women treated for rectal cancer, as the vast majority of the trials for both open and minimally invasive surgery assessed male patients only. To the best of the authors’ knowledge, our study represents the largest series ever published and the only evaluating also female sexual function [57].
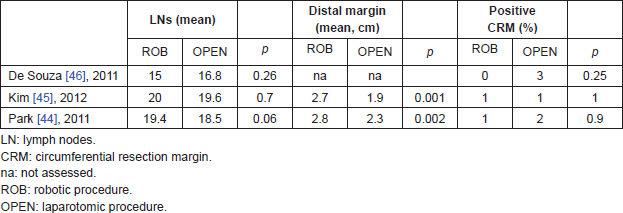
Oncologic safety has been definitely demonstrated for laparoscopy approach, with some concern about the CRM involvement, as reported in CLASICC trial [14]; the robotic series so far available demonstrate comparable oncologic results with laparoscopy. In [29], the number of lymph nodes retrieved was superior in robotic group, but percentage of involvement of CRM and radial margins were the same as laparoscopy (Table 6). In terms of oncologic safety, open rectal resection is still considered the standard surgical approach. In [45], 200 patients were prospectively evaluated (100 open and 100 robotic), the mean distal resection margin was significantly longer in the robotic than in the open group (2.7 versus 1.9 cm, P = 0.001). Moreover, multivariate analysis that included potent parameters, i.e. SSO types, preoperative CRT, T-category, and growth pattern, showed that patients with distal resection margin ≥2 cm were significantly more likely to be in the robotic than in the open group (OR, 2.415; 95% CI, 1.233–4.73; P = 0.01). Positive CRMs were observed in one patient from each of the two groups. The mean number of harvested lymph nodes and lymphovascular or perineural invasion was similar in the two groups. In the retrospective analysis of de Souza [46], there were not significant differences in distal and circumferential margin involvement, but in robotic group none CRM was involved and in open group, three out of 46 patients had a positive CRM (Table 7). In our early experience of first 25 robotic rectal resections compared with 25 laparoscopic rectal resections there was one (4%) positive CRM in the laparoscopic group and none in the robotic group, a difference not statistically significant. The number of harvested lymph nodes was slightly superior in the robotic (18) than in laparoscopic group (17), but again the difference was not statistically significant [21]. In all the meta-analyses comparing laparoscopic with robotic rectal resection, a significant difference in the quality of oncologic resection failed to be demonstrated [39–43]. In summary, oncologic safety of robotic rectal resection was clearly demonstrated in term of proximal, distal and circumferential margin clearance, and number of retrieved lymph nodes. No differences in CRM involvement were detected in comparison with open series, and concern expressed in the CLASICC trial for CRM involvement of laparoscopy-treated cases is not confirmed in the available robotic series.
Table 5. Urinary and sexual dysfunctions results in robotic rectal resection.
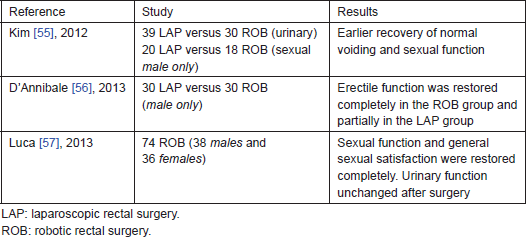
Table 6. Oncologic results of robotic and laparoscopic surgery for rectal cancer.
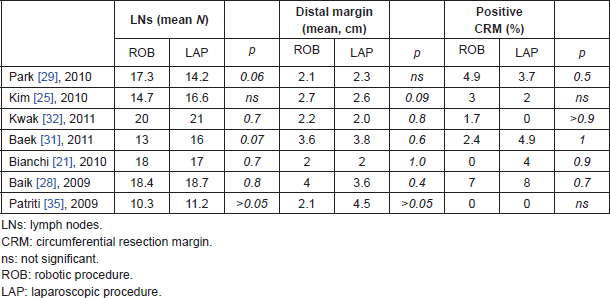
Table 7. Oncologic results of open and robotic surgery for rectal cancer.
Costs of robotic surgery
Robotic surgery is expensive, due to the cost of robot purchase, maintenance, and instruments. Therefore, a careful analysis of costs and of cost/effectiveness is mandatory, even if it is difficult to perform because of the lack of available data about the potential benefits of robotic surgery. A recent costs analysis in [58] demonstrates that robotic surgery is more expensive with a significantly lower hospital profit in a robotic group compared with a laparoscopic group. The authors underlined the necessity to demonstrate with controlled randomised trials potential oncological and clinical benefits of robotic TME to justify its costs.
On the contrary, the interesting study [59] demonstrates that robotic gastric bypass is less expensive than the equivalent laparoscopic and open operations. The economic advantage is correlated to the lower number of leakages and consequent increasing of the hospital stay costs, and to the absence of mechanical staplers use in the robotic subgroup. It is unlikely that in rectal surgery mechanical staplers will be abandoned, but the possibility to perform a safe robotic rectal suture—associated to the transanal removal of the specimen—is already demonstrated, as described in [60].
The costs/analysis in [61] on robotic prostatectomy summarises accurately the current status of the debate on costs of robotic surgery. The authors underline that robotic surgery provides similar postoperative outcomes of laparoscopic surgery but a reduced learning curve, although costs are currently high, increased competition from manufacturers, and wider dissemination of the technology could drive down costs.
Turchetti et al [62] performed a review of the literature on the costs of robotic surgery. They conclude that all the evaluated studies performed a pure costs/analysis, without an evaluation of the effectiveness of robotic surgery. The higher costs of robotic surgery are mainly due to the high purchase and maintenance costs for the robot and to a lesser extent to the longer operating room time. However, emerging evidence shows that operating room time decreases with experience in the use of the robot. Turchetti et al overlooked two aspects of robotic surgery, the possibility to facilitate some difficult minimally invasive procedures and the changing role of the assistant at the operating table. Concerning the possibility to increase the number of surgeons able to perform advanced minimally invasive procedures, the authors conclude that if this will be demonstrated by prospective studies the high initial investment in robotic technology may be more than justified.
One of the key steps to pursue a successful robotic programme is the surgical volume. It is strictly connected to the learning curve and to the quality of outcomes. Three to five cases per week during the initiation of the programme are necessary to obtain continuity in the learning curve. Palmer et al [63] reported a significant increase in surgical volume since the introduction of the robotic programme, from 40 to 350 cases per year within five years. The cost of surgery can be evaluated with an analysis of the variable costs and the fixed costs. Variable costs are related to all those activities that are necessary to produce the surgical performance (such as disposable tools, medications, and so on). Fixed costs are represented by the overall operating room time dedicated to robotics and the purchase of the system. It is clear that a high surgical volume centre can have an impact in terms of variable costs reduction; hence, the best chance to increase surgical volume and therefore to reduce costs is to share the use of the robotic system with all surgical teams, including urologists, gynaecologists, thoracic surgeons, and other specialties [64].
Conclusions
Minimally invasive surgery for rectal cancer is still confined to only a few centres, despite the evidence of better short-term clinical outcomes and at least the same oncologic safety of traditional open surgery. The reason for this low diffusion is mainly technical, and robotic surgery seems to overcome some of the limitations of laparoscopy, offering an easy technique with less fatigue for the surgeons and a better possibility of teaching for the beginners. The main concern is related to the high costs of robotic surgery, which can be sustained by a careful economic plan and by the use of the robot spread between different surgical specialties. Furthermore, if it will be demonstrated that an easier technique such as robotics may increase the number of minimally invasive rectal resections, the following costs/effectiveness analysis probably will be in favour of robotic surgery. The results of the ROLARR randomised controlled clinical trial are awaited to assess potential clinical and oncological benefits of robotic surgery in rectal cancer, which can justify its higher costs, as well as an enlargement of competition between the companies producing robots, that could reduce the costs of instrumentation.
References
1. MacFarlane JK, Ryall RD and Heald RJ (1993) Mesorectal excision for rectal cancer Lancet 341(8843) 457–60 DOI: 10.1016/01406736(93)90207-W PMID: 8094488
2. Heald RJ et al (1998) Rectal cancer: the Basingstoke experience of total mesorectal excision 1978–1997 Arch Surg 133(8) 894–9 DOI: 10.1001/archsurg.133.8.894 PMID: 9711965
3. Ohtani H et al (2011) A meta-analysis of the short- and long-term results of randomized controlled trials that compared laparoscopy-assisted and conventional open surgery for rectal cancer J Gastrointest Surg 15 1375–85 DOI: 10.1007/s11605-011-1547-1 PMID: 21557014
4. Staudacher C and Vignali A (2010) Laparoscopic surgery for rectal cancer: the state of the art World J Gastrointest Surg 2(9) 275–82 DOI: 10.4240/wjgs.v2.i9.275 PMID: 21160896 PMCID: 2999691
5. Jensen TCP and Wai LL (2009) Laparoscopic resection for rectal cancer: a review Ann Surg Oncol 16 3038–47 DOI: 10.1245/s10434-009-0603-5
6. Kuhry E et al (2008) Long-term results of laparoscopic colorectal cancer resection Cochrane Database Syst Rev (2) CD003432 PMID: 18425886
7. Lee JK, Delaney CP and Lipman JM (2012) Current state of the art in laparoscopic colorectal surgery for cancer: update on the multi-centric international trials Ann Surg Innov Res 6(1) 5 DOI: 10.1186/1750-1164-6-5 PMID: 22846394 PMCID: 3444362
8. Jamali FR et al (2008) Evaluating the degree of difficulty of laparoscopic colorectal surgery Arch Surg 143(8) 762–7 DOI: 10.1001/archsurg.143.8.762 PMID: 18711036
9. Kayano H et al (2011) Evaluation of the learning curve in laparoscopic low anterior resection for rectal cancer Surg Endosc 25 2972–9 DOI: 10.1007/s00464-011-1655-8 PMID: 21512883
10. Li JCM et al (2012) Institution learning curve of laparoscopic colectomy—a multi-dimensional analysis Int J Colorectal Dis 27(4) 527–33 DOI: 10.1007/s00384-011-1358-6
11. Shah PR, Joseph A and Haray PN (2005) Laparoscopic colorectal surgery: learning curve and training implications Postgrad Med J 81 537–40 DOI: 10.1136/pgmj.2004.028100 PMID: 16085749 PMCID: 1743324
12. IJ Park et al (2009) Multidimensional analysis of the learning curve for laparoscopic colorectal surgery: lessons from 1,000 cases of laparoscopic colorectal surgery Surg Endosc 23 839–46 DOI: 10.1007/s00464-008-0259-4 PMID: 19116741
13. Spinoglio G et al (2009) Robotic laparoscopic surgery with the da Vinci® system: an early experience Surg Technol Int 18 70–4 PMID: 19579191
14. Guillou PJ et al (2005) Short-term endpoints of conventional versus laparoscopic assisted surgery in patients with colorectal cancer (MRC CLASICC trial): multicentre, randomised controlled trial Lancet 365 1718–26 DOI: 10.1016/S0140-6736(05)66545-2 PMID: 15894098
15. Sung-Bum Kang et al (2010) Open versus laparoscopic surgery for mid or low rectal cancer after neoadjuvant chemoradiotherapy (COREAN trial): short-term outcomes of an open-label randomised controlled trial Lancet Oncol 11 637–45 DOI: 10.1016/S1470-2045(10)70131-5
16. Jayne DG et al (2010) Five-year follow-up of the Medical Research Council CLASICC trial of laparoscopically assisted versus open surgery for colorectal cancer Br J Surg 97(11) 1638–45 DOI: 10.1002/bjs.7160 PMID: 20629110
17. Rottoli M et al (2009) Laparoscopic rectal resection for cancer: effects of conversion on short-term outcome and survival Ann Surg Oncol 16(5) 1279–86 DOI: 10.1245/s10434-009-0398-4 PMID: 19252948
18. Bianchi PP et al (2007) Laparoscopic surgery in rectal cancer: a prospective analysis of patient survival and outcomes Dis Colon Rectum 50(12) 2047–53 DOI: 10.1007/s10350-007-9055-9 PMID: 17906896
19. Siegel R et al (2011) Laparoscopic extraperitoneal rectal cancer surgery: the clinical practice guidelines of the European Association for Endoscopic Surgery (EAES) Surg Endosc 25 2423–40 DOI: 10.1007/s00464-011-1805-z PMID: 21701921
20. Taylor GW and Jayne DG (2007) Robotic applications in abdominal surgery: their limitations and future developments Int J Med Robot 3 3–9 Review DOI: 10.1002/rcs.115 PMID: 17441019
21. Bianchi PP et al (2010) Robotic versus laparoscopic total mesorectal excision for rectal cancer: a comparative analysis of oncological safety and short-term outcomes Surg Endosc 24(11) 2888–94 DOI: 10.1007/s00464-010-1134-7 PMID: 20526623
22. Ficarra V et al (2012) Systematic review and meta-analysis of studies reporting potency rates after robot-assisted radical prostatectomy Eur Urol 62(3) 418–30 DOI: 10.1016/j.eururo.2012.05.046 PMID: 22749850
23. Luca F et al (2009) Full robotic left colon and rectal cancer resection: technique and early outcome Ann Surg Oncol 16 1274–78 DOI: 10.1245/s10434-009-0366-z PMID: 19242762
24. Smith AL et al (2012) Dual-console robotic surgery compared to laparoscopic surgery with respect to surgical outcomes in a gynecologic oncology fellowship program Gynecol Oncol 126(3) 432–6 DOI: 10.1016/j.ygyno.2012.05.017 PMID: 22613352
25. Kim NK and Kang J (2010) Optimal total mesorectal excision for rectal cancer: the role of robotic surgery from an expert’s view J Korean Soc Coloproctol 26(6) 377–87 DOI: 10.3393/jksc.2010.26.6.377
26. Baek JH et al (2010) Oncologic outcomes of robotic-assisted total mesorectal excision for the treatment of rectal cancer Ann Surg 251 882–6 DOI: 10.1097/SLA.0b013e3181c79114 PMID: 20395863
27. Baik SH et al (2008) Robotic tumor-specific mesorectal excision of rectal cancer: short-term outcome of a pilot randomized trial Surg Endosc 22 1601–8 DOI: 10.1007/s00464-008-9752-z PMID: 18270772
28. Baik SH et al (2009) Robotic versus laparoscopic low anterior resection of rectal cancer: short-term outcome of a prospective comparative study Ann Surg Oncol 16 1480–7 DOI: 10.1245/s10434-009-0435-3 PMID: 19290486
29. Park JS et al (2010) Robotic-assisted versus laparoscopic surgery for low rectal cancer: case-matched analysis of short-term outcomes Ann Surg Oncol 17 3195–202 DOI: 10.1245/s10434-010-1162-5 PMID: 20589436
30. Pigazzi A et al (2006) Robotic-assisted laparoscopic low anterior resection with total mesorectal excision for rectal cancer Surg Endosc 20 1521–5 DOI: 10.1007/s00464-005-0855-5 PMID: 16897284
31. Baek JH, Pastor C and Pigazzi A (2011) Robotic and laparoscopic total mesorectal excision for rectal cancer: a case-matched study Surg Endosc 25 521–5 DOI: 10.1007/s00464-010-1204-x
32. Kwak JM et al (2011) Robotic vs laparoscopic resection of rectal cancer: short-term outcomes of a case-control study Dis Colon Rectum 54 151–6 DOI: 10.1007/DCR.0b013e3181fec4fd PMID: 21228661
33. Jiménez Rodríguez RM et al (2011) Prospective randomised study: robotic-assisted versus conventional laparoscopic surgery in colorectal cancer resection Cir Esp 89 432–8 DOI: 10.1016/j.ciresp.2011.01.017 PMID: 21530948
34. Popescu I et al (2010) The minimally invasive approach, laparoscopic and robotic, in rectal resection for cancer. A single center experience Acta Chir Iugosl 57 29–35 DOI: 10.2298/ACI1003029P PMID: 21066980
35. Patriti A et al (2009) Short- and medium-term outcome of robot-assisted and traditional laparoscopic rectal resection JSLS 13 176–83 PMID: 19660212 PMCID: 3015932
36. Hellan M et al (2007) Short-term outcomes after robotic-assisted total mesorectal excision for rectal cancer Ann Surg Oncol 14(11) 3168–73 DOI: 10.1245/s10434-007-9544-z PMID: 17763911
37. Spinoglio G et al (2008) Robotic colorectal surgery: first 50 cases experience Dis Colon Rectum 51(11) 1627–32 DOI: 10.1007/s10350-008-9334-0 PMID: 18484134
38. Ng KH et al (2009) Robotic-assisted surgery for low rectal dissection: from better views to better outcome Singapore Med J 50(8) 763–7 PMID: 19710972
39. Memon S et al (2012) Robotic versus laparoscopic proctectomy for rectal cancer: a meta-analysis Ann Surg Oncol 19 2095–101 DOI: 10.1245/s10434-012-2270-1 PMID: 22350601
40. Yang Y et al Robot-assisted versus conventional laparoscopic surgery for colorectal disease, focusing on rectal cancer: a meta-analysis Ann Surg Oncol 19(12) 3727–36 DOI: 10.1245/s10434-012-2429-9 PMID: 22752371
41. Lin S et al (2011) Meta-analysis of robotic and laparoscopic surgery for treatment of rectal cancer World J Gastroenterol 17(47) 5214–20 DOI: 10.3748/wjg.v17.i47.5214
42. Gonzalez Fernandeza AM and Mascarenas Gonzalez JF (2012) Revision de conjunto escision mesorrectal total laparoscopica versus asistida por robot en el tratamiento del cancer de recto: un metaanalisis Cir Esp 90(6) 348–54 DOI: 10.1016/j.ciresp.2012.03.004
43. Trastulli S et al (2012) Robotic resection compared with laparoscopic rectal resection for cancer: systematic review and metaanalysis of short-term outcome Colorectal Dis 14(4) e134–56 DOI: 10.1111/j.1463-1318.2011.02907.x
44. Park JS et al (2011) S052: a comparison of robot assisted, laparoscopic, and open surgery in the treatment of rectal cancer Surg Endosc 25 240–8 DOI: 10.1007/s00464-010-1166-z
45. Kim JC et al (2012) Open versus robot-assisted sphincter-saving operations in rectal cancer patients: techniques and comparison of outcomes between groups of 100 matched patients Int J Med Robotics Comput Assist Surg 8(4) 468–75 DOI: 10.1002/rcs.1452
46. deSouza AL et al (2011) A comparison of open and robotic total mesorectal excision for rectal adenocarcinoma Dis Colon Rectum 54 275–82 DOI: 10.1007/DCR.0b013e3182060152 PMID: 21304296
47. Buunen M et al (2009) COLOR II A randomized clinical trial comparing laparoscopic and open surgery for rectal cancer Dan Med Bull 56(2) 89–91 PMID: 19486621
48. Collinson FJ et al (2012) An international, multicentre, prospective, randomised, controlled, unblinded, parallel-group trial of robotic-assisted versus standard laparoscopic surgery for the curative treatment of rectal cancer Int J Colorectal Dis 27 233–41 DOI: 10.1007/s00384-011-1313-6
49. Bokhari MB et al (2011) Learning curve for robotic-assisted laparoscopic colorectal surgery Surg Endosc 25(3) 855–60 DOI: 10.1007/s00464-010-1281-x PMCID: 3044842
50. Akmal Y et al (2012) Robot-assisted total mesorectal excision: is there a learning curve? Surg Endosc 26(9) 2471–6 DOI: 10.1007/s00464-012-2216-5 PMID: 22437950
51. Pigazzi A et al (2010) Multicentric study on robotic tumor specific mesorectal excision or the treatment of rectal cancer Ann Surg Oncol 17 1614–20 DOI: 10.1245/s10434-010-0909-3 PMID: 20087780
52. Sartori CA et al (2011) NO urinary and sexual disorders after laparoscopic TME for rectal cancer in males J Gastrointest Surg 15 637–43 DOI: 10.1007/s11605-011-1459-0 PMID: 21327977
53. Morino M et al (2009) Male sexual and urinary function after laparoscopic total mesorectal excision Surg Endosc 23 1233–40 DOI: 10.1007/s00464-008-0136-1
54. Jayne DG et al (2005) Bladder and sexual function following resection for rectal cancer in a randomized clinical trial of laparoscopic versus open technique Br J Surg 92 1124–32 DOI: 10.1002/bjs.4989 PMID: 15997446
55. Kim JY et al (2012) A comparative study of voiding and sexual function after total mesorectal excision with autonomic nerve preservation for rectal cancer: laparoscopic versus robotic surgery Kim Ann Surg Oncol 19(8) 2485–93 DOI: 10.1245/s10434012-2262-1
56. D’Annibale A et al (2013) Total mesorectal excision: a comparison of oncological and functional outcomes between robotic and laparoscopic surgery for rectal cancer Surg Endosc 27(6) 1887–95 DOI: 10.1007/s00464-012-2731-4
57. Luca F et al (2013) Impact of robotic surgery on sexual and urinary functions after fully robotic nerve-sparing total mesorectal excision for rectal cancer Ann Surg 257(4) 672–8 DOI: 10.1097/SLA.0b013e318269d03b
58. Se-Jin Baek et al (2012) Robotic versus conventional laparoscopic surgery for rectal cancer: a cost analysis from a single institute in korea World J Surg 36(11) 2722–9 DOI: 10.1007/s00268-012-1728-4
59. Monika E et al (2012) Reducing cost of surgery by avoiding complications: the model of robotic Roux-en-Y gastric bypass Obes Surg 22 52–61 DOI: 10.1007/s11695-011-0422-1
60. Choi GS et al (2009) A novel approach of robotic-assisted anterior resection with transanal or transvaginal retrieval of the specimen for colorectal cancer Surg Endosc 23(12) 2831–5 DOI: 10.1007/s00464-009-0484-5 PMID: 19440794
61. Ahmed K et al (2012) Assessing the cost effectiveness of robotics in urological surgery—a systematic review BJU Int 110(10) 1544–56 DOI: 10.1111/j.1464-410X.2012.11015.x PMID: 22443296
62. Turchetti G et al (2012) Economic evaluation of da Vinci-assisted robotic surgery: a systematic review Surg Endosc 26(3) 598–606 DOI: 10.1007/s00464-011-1936-2
63. Palmer KJ et al (2008) Launching a successful robotic surgery program J Endourol 22(4) 819–24 DOI: 10.1089/end.2007.9824 PMID: 18419223
64. Rocco B et al (2009) Building a robotic program Scand J Surg 98 72–5 PMID: 19799044






