Breast cancer brain metastases: new directions in systemic therapy
Nancy U Lin
Dana-Farber Cancer Institute, Boston, MA, USA
Correspondence to: Nancy U Lin. Email: nlin@partners.org
Abstract
The management of patients with brain metastases from breast cancer continues to be a major clinical challenge. The standard initial therapeutic approach depends upon the size, location, and number of metastatic lesions and includes consideration of surgical resection, whole-brain radiotherapy, and stereotactic radiosurgery. As systemic therapies for control of extracranial disease improve, patients are surviving long enough to experience subsequent progression events in the brain. Therefore, there is an increasing need to identify both more effective initial treatments as well as to develop multiple lines of salvage treatments for patients with breast cancer brain metastases. This review summarises the clinical experience to date with respect to cytotoxic and targeted systemic therapies for the treatment of brain metastases, highlights ongoing and planned trials of novel approaches and identifies potential targets for future investigation.
Keywords: brain and nervous system, breast
Introduction
Historically, brain metastases were associated with very poor survival [1]. While brain metastases remain incurable, there is increasing optimism that advances in local and systemic therapies may provide clinical benefit in some subsets of patients, and there is increasing interest in exploring novel approaches to the prevention and management of this challenging clinical problem. This review summarises the clinical experience to date with respect to cytotoxic and targeted systemic therapies, highlights ongoing and planned trials and discusses potential targets and novel trial designs for future investigation.
Incidence and prognosis
Brain metastases are diagnosed in approximately 15% of unselected patients with advanced breast cancer [2]. Over time, it has become increasingly clear that the biology of the primary tumour influences the pattern of metastatic spread, including the likelihood of relapse in the central nervous system (CNS) [3–5]. As many as half of patients with HER2-positive advanced breast cancer will develop brain metastases at some point in the course of their disease [6–8]. Within the HER2-positive subset, hormone receptor status appears to further define the risk of CNS relapse, with patients having hormone receptor-negative/HER2-positive tumours experiencing increased risk of the CNS as site of first relapse compared with patients with hormone receptor-positive/HER2-positive tumours [9]. Patients with metastatic, triple-negative (ER, PR and HER2 negative) breast cancer are also at high risk, with 25–46% of patients developing brain metastases at some point in the course of their disease [4, 8]. Of interest, the timing of the CNS relapse also appears to vary by tumour subtype. Patients with non-luminal tumours (e.g. triple-negative cancers) appear to experience a shorter time to CNS relapse compared with patients with luminal tumours [5, 10].
In historical series, the median survival of unselected patients with breast cancer brain metastases treated with whole-brain radiotherapy (WBRT) has been reported at approximately five to six months [11]. More recent analyses have identified performance status and biologic subtype as major drivers of prognosis. For example, in a multi-institutional retrospective database of over 400 patients with breast cancer brain metastases, a prognostic model (the Diagnosis-Specific Graded Prognostic Assessment, DSGPA) using these factors (plus age) was able to distinguish between patients experiencing a two year median survival versus those with 3.4 months median survival [12, 13]. Across multiple retrospective studies, the most striking differences have been consistently noted between patients with HER2-positive breast cancer (who carry the most favourable prognosis) and those patients with triple-negative breast cancer [14–19]. Based on several lines of evidence, it is likely that improved systemic tumour control is a major contributing factor to this difference. First, although one must interpret retrospective data cautiously because of issues with patient selection, it has been observed by multiple groups that patients with HER2-positive tumours who continue on anti-HER2 therapy following the diagnosis of brain metastases fare better than those who received either no therapy, or chemotherapy without HER2-directed therapy [18, 19]. Second, as many as half of patients with HER2-positive brain metastases suffer a death primarily related to CNS (as opposed to systemic) progression; this is distinctly different from patients with triple-negative brain metastases, where patients most commonly die of uncontrolled systemic disease [6, 17].
Current standard of care
A full discussion of the current standard of care approaches is outside the scope of this review. Treatment guidelines for patients with brain metastases from solid tumours are available through the National Comprehensive Cancer Network (NCCN) and professional societies [20, 21]. Because of the relative paucity of breast cancer-specific prospective trials in this patient population, the recommendations are largely based upon data generated from trials of patients with solid tumours (of which lung cancer is most highly represented).
In general, the initial management of patients with brain metastases depends on (a) the number, size, and location of brain lesions; (b) the presence or absence of neurological symptoms; (c) the patient’s performance status and medical comorbidities; (d) the status of systemic metastases; (e) the availability of systemic treatment options; and (f) patient preference. In general, initial management will include some combination of surgical resection, radiosurgery, and/or WBRT, depending on the above factors. Systemic therapy could be a consideration either on a clinical trial, in the context of minimal CNS disease burden with rapidly progressive extracranial disease, or in select, well-informed patients as an alternative to localised therapies with close follow-up (i.e. a patient with small, asymptomatic CNS lesions). Among patients who have developed subsequent CNS progression after initial standard therapy, options include surgical resection, WBRT, stereotactic radiosurgery (SRS), off-label use of systemic therapy, consideration of a clinical trial, or best supportive care. Options will vary based on prior treatments received, response to prior treatments, location and number of the new or progressive CNS lesions, and the other patient and disease-related factors as listed above. For both initial and subsequent management decisions, a multidisciplinary approach is essential in making recommendations based upon an assessment of the patient in the context of their local therapy options, extracranial disease status, planned or ongoing systemic therapy, and clinical trial options.
Available data for systemic chemotherapy approaches
At present, there are no FDA-approved systemic therapies for the treatment of breast cancer brain metastases. A number of small, prospective trials of cytotoxic chemotherapy have been completed and are summarised in Table 1.
Temozolomide has been examined in a phase II trial of the National Cancer Institute of Canada Clinical Trials Group (NCI-CTG) [22]. This trial included 19 women with heavily pretreated metastatic breast cancer (MBC) and allowed patients with brain metastases (n=5) to participate. No objective responses were observed and the study was closed to further accrual due to lack of sufficient activity. A dose-dense temozolomide regimen has also been evaluated in a phase II study of solid tumour patients (n=51 with breast cancer) [23]. The objective response rate among breast cancer patients was 4%; however, the majority of the responses were transient, and median progression-free survival (PFS) was approximately two months.
Table 1: Prospective trials of chemotherapy for breast cancer brain metastases.
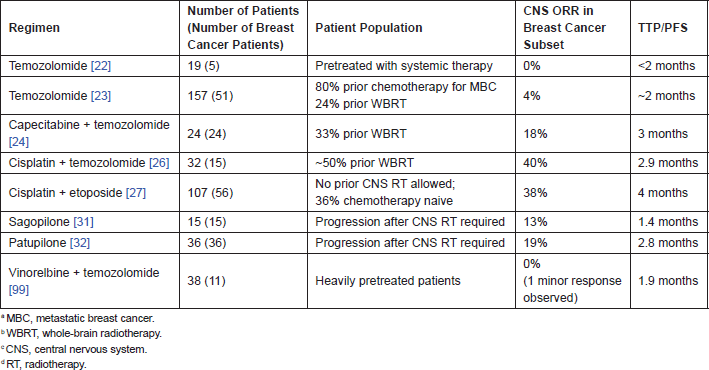
Capecitabine is an active agent in patients with MBC. With respect to breast cancer brain metastases, capecitabine has been evaluated in a phase I study in combination with temozolomide, with a reported response rate of 18% [24]. CNS activity has also been reported in a case series from Memorial Sloan-Kettering Cancer Center, with capecitabine given as a single agent [25].
Platinum agents have also been prospectively evaluated, although in combination with other cytotoxic agents rather than as monotherapy. In a phase II study evaluating the combination of cisplatin and temozolomide, six out of 15 (40%) of breast cancer patients achieved a partial CNS response [26]. For the 32 total patients enrolled in the study, median time to progression (TTP) was 2.9 months. In a separate study evaluating the combination of cisplatin and etoposide, the CNS response rate was 38% with a median TTP of four months [27]. It should be noted that in the latter study, patients were not allowed to have received prior CNS radiotherapy, and about one-third were chemotherapy naive. Additionally, although there has been increasing interest in the use of platinum agents in certain subsets of breast cancer, the systemic response rate in unselected, pretreated patients has been low [28]. Still, for patients who have seen relatively little prior systemic therapy and who have progressive brain metastases, off-label use of platinum agents could be a plausible treatment option.
Epothilones are tubulin-stabilising agents with activity against multiple solid tumours. At present, only ixabepilone is commercially available and is approved for the treatment of refractory MBC. Negative results of a trial in patients with primary brain tumours and the existence of investigation alepothilones with excellent blood–brain barrier (BBB) permeability have limited efforts to formally test the effects of ixabepilone in patients with breast cancer brain metastases [29]. Sagopilone is an epothilone B analogue that readily crosses the BBB and is not a P-glycoprotein substrate [30]. Despite its preclinical promise, its single-agent activity in the phase II study breast cancer brain metastasis study was only modest, and there currently are no plans to further develop the drug in this setting [31]. Patupilone, another BBB-permeable epothilone, has also demonstrated CNS activity in breast cancer, and future trials are under consideration [32].
Finally, some regimens, while not evaluated in prospective clinical trials, have reported efficacy in case series and include CMF (cyclophosphamide, methotrexate and 5-fluorouracil), anthracycline-based regimens and high-dose intravenous methotrexate [33, 34].
Available data for targeted therapy approaches
Endocrine therapies remain the oldest and most established targeted treatments for the management of patients with breast cancer. In the setting of CNS disease, there have been multiple case reports describing responses to tamoxifen, aromatase inhibitors and megestrol acetate, although no prospective trials have been conducted [35–37]. Unfortunately, the reality is that most patients with ER-positive breast cancers have developed endocrine-refractory disease by the time brain metastases are detected. However, among the subset who have had minimal prior endocrine therapy exposure and/or who have had prior sustained responses to treatment, endocrine therapy may be an option that could be considered.
Trastuzumab does not appear to cross the intact BBB [38]. Although it should be acknowledged that trastuzumab levels have not been directly measured in human brain metastasis samples, nevertheless, clinically, it has been repeatedly observed that a substantial proportion of patients with HER2-positive MBC develop isolated CNS progression in the setting of continued systemic disease control on trastuzumab [6]. A number of studies have evaluated the efficacy of lapatinib, a small molecule tyrosine kinase inhibitor that targets both EGFR and HER2, in the setting of HER2-positive breast cancer brain metastases (Table 2). The response rate to single-agent lapatinib in the refractory setting was underwhelming; however, when given in combination with capecitabine, CNS response rates ranging from 18% to 38% have been reported [39–44]. More recently, lapatinib plus capecitabine has been studied in the upfront setting in the LANDSCAPE trial, prior to the use of WBRT or other local therapies for the treatment of brain metastases. Approximately two-thirds of patients achieved a CNS objective response, and the median TTP was 5.5 months, with one-year survival exceeding 70% (Bachelot et al, Lancet Oncology, in press) [45]. Results of this study have prompted ongoing European efforts to organise a potential phase III trial directly comparing lapatinib plus capecitabine with WBRT for initial treatment of patients with HER2-positive brain metastases, a study which will be needed before this regimen becomes part of routine upfront care.
Table 2: Studies of Lapatinib in HER2-positive breast cancer brain metastases.
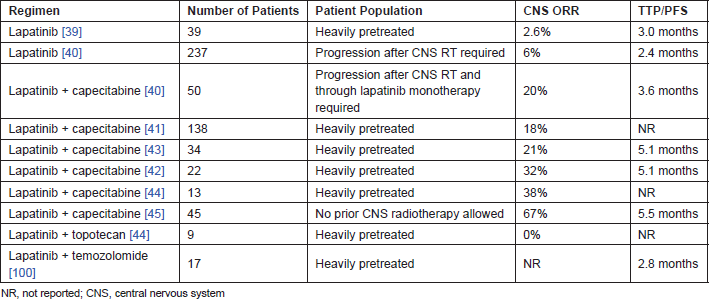
Lapatinib plus capecitabine has also been compared with trastuzumab plus capecitabine in the CEREBEL trial, with a primary endpoint of the development of CNS metastases [46]. Of note, patients were required to have CNS screening at baseline, and this led to 20% of the study patients having asymptomatic brain metastases detected, leading to study exclusion. The study did not meet its primary endpoint because of a paucity of CNS events in both arms. It is likely that the study was inconclusive as the highest-risk population for developing brain metastases was screened out due to the screening procedures.
New targets and avenues of investigation
Despite some notable advances in recent years, there remains much progress to be made. Commercially available treatment options for patients with progressive brain metastases after surgical or radiotherapy approaches remain limited, and there are still no systemic regimens that have gained a formal indication in this setting. Fortunately, the landscape is changing. There are a number of novel agents of interest and an increasing number of trials designed to evaluate the efficacy of these compounds in the CNS (Table 3).
The ideal agent would reach therapeutic concentrations in the brain, be active against breast cancer in both intracranial and extracranial sites (including in patients who have received multiple prior lines of systemic therapy), have a favourable toxicity profile, and demonstrably benefit patients. Two examples are the third-generation taxane TPI-287 and the peptide-taxane conjugate GRN1005. TPI-287 is a microtubule-stabilising agent designed to circumvent the MDR-1 drug efflux resistance mechanism and is also highly permeable across the intact BBB. In a preclinical model using intracardiac injection of 231-BR cells, treatment with TPI-287 inhibited outgrowth of brain metastases, an effect that was not seen with paclitaxel, ixabepilone, or nab-paclitaxel [47]. GRN1005 (previously ANG1005) is a peptide (angiopep-2)-taxane conjugate that targets the lipoprotein receptor-related protein 1, which is upregulated on the surface of the BBB as well as in brain metastases, and reaches therapeutic concentrations in the brain in preclinical models [48, 49]. In the phase I study in patients with brain metastases from solid tumours, CNS activity was observed across multiple tumour types, including breast cancer and patients with taxane-refractory disease [50]. The most commonly observed adverse events were neutropenia, anaemia, and fatigue. Based upon the data outlined above, phase II trials of both TPI-287 and GRN1005 are now ongoing. However, interim analysis of the phase II trial of GRN 1005 in breast cancer brain metastases did not meet its efficacy endpoint. Follow up and analysis of the entire study population will be required before reaching conclusions about the future of the compound.
Table 3: Selected trials for breast cancer brain metastases.
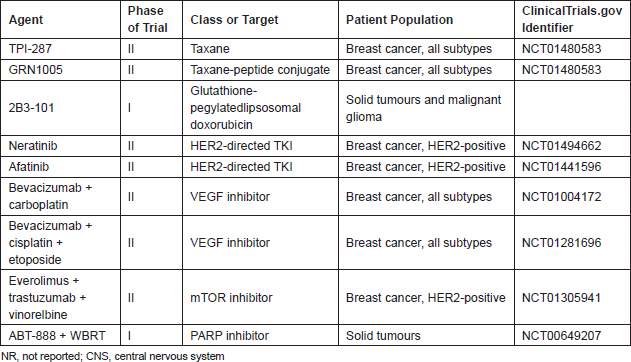
In light of the activity observed with lapatinib, other HER2-directed tyrosine kinase inhibitors are also being evaluated for CNS activity. For example, neratinib has been evaluated in phase II trials in HER2-positive MBC patients for the treatment of extracranial disease. Approximately half of patients treated in the first-line setting achieved an objective response; in the refractory setting, the response rate was 24%, with a median PFS of 22 weeks [51]. Based on these promising findings, neratinib is now being evaluated in a multicentre study for the treatment of patients with HER2-positive breast cancer and progressive CNS metastases. Similarly, afatinib is an irreversible inhibitor of EGFR and HER2, with preliminary evidence of activity in the phase II non-CNS metastatic setting, which is currently in clinical trials for the treatment of breast cancer brain metastases [52]. Other HER2-targeting compounds of interest, including those with excellent BBB penetration, are in the early stages of clinical development.
Beyond HER2, other targets of interest include tumour-associated angiogenesis, the PI3K/mTOR pathway, and the DNA repair pathway. With respect to angiogenesis, it is well documented that breast cancer brain metastases are highly angiogenic and associated with abnormal tumour-associated vasculature [53]. In preclinical models, VEGF promotes the growth of breast cancer brain metastases, and this growth is inhibited by anti-angiogenic agents [54]. There are preliminary data supporting an effect of bevacizumab on brain metastases and reassuring safety data from the standpoint of CNS haemorrhage [55, 56]. Moreover, bevacizumab has an established role in the treatment of primary brain tumours [57, 58]. It is expected that additional data regarding the efficacy of bevacizumab/chemotherapy combinations for the treatment of breast cancer brain metastases will be available within the next year (NCT01004172; NCT01281696) and, if promising, could lead to additional trials in the near future. It should be noted that radiographic response to bevacizumab should be interpreted with some caution, given its anti-oedema effects, which may lead to decreases in gadolinium leakage and potential under-calling of tumour burden. While in patients with primary brain tumours, specific modifications of response criteria have been added to account for some of these issues, there are as of yet no standard criteria for assessment of CNS response in the setting of solid tumour brain metastases [59].
Another pathway of considerable active interest in breast cancer is the PI3K/mTOR pathway. Earlier this year, everolimus, a rapamycin analogue that inhibits mTOR signalling, was approved in combination with exemestane for patients with ER-positive MBC, on the basis of a randomised trial demonstrating a substantial improvement in PFS [60]. In a randomised phase II trial, everolimus also demonstrated additive benefit when given in conjunction with tamoxifen [61]. Everolimus is also under active investigation [62, 63] in the setting of HER2-positive breast cancer. Notably, everolimus has demonstrated efficacy against a rare type of brain tumour (subependymal giant cell astrocytoma, SEGA), suggesting it reaches therapeutic levels in the brain [64]. A phase II study evaluating everolimus (in combination with trastuzumab and vinorelbine) among patients with HER2-positive breast cancer brain metastases is ongoing.
In tandem with ongoing studies evaluating mTOR inhibitors in breast cancer, there is a tremendous amount of effort being placed in evaluating pan- and alpha-specific PI3 kinase inhibitors. Approximately one-third of HER2-positive breast cancers contain a PIK3CA somatic mutation, and PTEN loss is present in up to half of triple-negative breast cancers [65]. PIK3CA mutations are also commonly present in ER-positive breast cancers [66]. Of interest, anecdotal reports of CNS activity have been reported with BEZ235 and BKM120 in a patient with HER2-positive and triple-negative breast cancer, respectively [67, 68]. In particular, BKM120 is associated with mood alterations that are thought to be related its ability to cross the intact BBB (Zhao J, personal communication, September 2012), and there is interest in further evaluating this compound in the CNS.
Finally, although the enthusiasm for PARP inhibitors waned somewhat with the presentation of the negative phase III iniparib trial, it now appears that iniparib is not a true PARP inhibitor [69]. Other PARP inhibitors have shown clear single-agent activity in BRCA mutation carriers, which is notable given reports of a high incidence of brain metastases in BRCA carriers with breast cancer [70–72]. Their role in non-BRCA-associated breast cancer is still uncertain, but it is an area of continued active investigation. A phase I study of the PARP inhibitor ABT-888 in combination with WBRT is ongoing, and there is interest in evaluating the combination of PARP inhibitors plus chemotherapy or other targeted approaches for brain metastases in the future.
Leptomeningeal metastases
Among patients with leptomeningeal metastases, breast cancer is the most commonly represented solid tumour [73]. Still, leptomeningeal metastases are a fairly infrequent occurrence in most breast cancer patients. In series from Korea and the United States, only 3–5% of patients with HER2-positive MBC were reported to have developed leptomeningeal metastases [6, 7, 74]. The incidence does appear higher than this in patients with ER-positive lobular breast cancers and in patients with triple-negative breast cancer, although the point estimates have varied [4, 75–77].
There are very few prospective trials to guide therapy. Unfortunately, survival remains poor across breast cancer subtypes [78]. Intrathecal chemotherapy can be effective, but even with good patient selection, long-term responses are uncommon [79, 80]. Case reports and case series have been published on the experience with endocrine therapy, high-dose intravenous methotrexate, capecitabine, and irinotecan, and off-label use could be considered in select circumstances [25, 81–83]. Finally, in patients with elevated intracranial pressure, placement of a ventriculoperitoneal shunt can have clear palliative benefits and is a consideration within the context of multidisciplinary management [84, 85].
From an investigational standpoint, given that HER2 amplification appears to be retained in patients with HER2-positive primary tumours and leptomeningeal involvement, intrathecal trastuzumab is the subject of two ongoing prospective trials in France and in the United States (www.clinicaltrials.gov; NCT01373710, NCT01325207) [86]. A number of case reports have been published in the medical literature, although on close review, in many cases, patients received multiple concurrent therapies, making isolation of the true effect of intrathecal trastuzumab a challenge [87–89]. At present, off-label use of intrathecal trastuzumab is not recommended, given that the commercial drug supply is not formulated for intrathecal use [90]. Newer anti-HER2 agents, such as pertuzumab or TDM1, which have demonstrated activity against extracranial metastases, could also be of interest in patients with leptomeningeal disease when given intrathecally. However, these would need to be studied carefully for both safety and efficacy and, as of now, would not be recommended for off-label use in this setting [91–93].
Challenges and opportunities in clinical trial design
Patients with brain metastases have historically been excluded from the vast majority of clinical trials in cancer. As patients with brain metastases are included in trials and as more trials are opened that specifically assess the CNS efficacy of novel treatments, there is an increasing need to standardise response criteria for assessment of brain metastases. Because neither the RECIST nor Macdonald criteria were developed for the specific purpose of evaluating patients with CNS metastases, there are some gaps and inconsistencies in how they are applied in brain metastasis studies, and many groups have either modified the criteria or developed new criteria. The Response Assessment in Neuro-Oncology (RANO) Metastatic Working Group is currently taking on the task of formulating new response guidelines in an effort to reduce this heterogeneity across studies.
A traditional clinical trial paradigm has been to evaluate novel systemic approaches in the refractory setting (i.e., in patients whose CNS disease has progressed after radiotherapy) in patients with measurable disease and with objective response as the primary endpoint. Provocative data in a small study of patients treated with lapatinib and capecitabine as upfront therapy for HER2-positive brain metastases raise the question of whether systemic therapy might someday take the place of radiotherapy-based approaches and raise difficult questions as to what type of data would be required to comfortably make that shift [45]. In particular, in a hypothetical phase III study directly comparing WBRT with systemic therapy, CNS response, or CNS progression-free survival may not be the most appropriate primary endpoint(s). In a small, randomised study comparing SRS alone to SRS with WBRT, although intracranial control was superior in the SRS WBRT arm, neurocognitive outcomes were inferior, suggesting that intracranial control is not directly translatable to improved patient outcomes [94]. Yet, designating progression-free survival as the primary endpoint leaves open questions about the allowable systemic therapies in the WBRT arm and about the treatment of CNS and non-CNS progression in the statistical plan.
Another trial design that has been proposed is to consider ‘secondary prevention’; that is, to assess the ability of novel approaches to prevent further CNS progression among patients with a limited number of brain metastases treated with SRS alone. To date, there have not been any clinical breast cancer studies utilising this model, although there is significant interest in launching such a study. Ultimately, it would be ideal to develop therapies that could prevent the occurrence of brain metastases altogether. For example, in preclinical models, targeting of polo-like kinase 1, α(V)-integrins, b-raf, and histone deacetylase decreases brain metastatic outgrowth and could be potentially directed translated into clinical trials in the future [95–98].
Conclusions
Breast cancer brain metastases continue to pose a difficult challenge for patients and clinicians. For the majority of patients, the most appropriate initial therapy remains a radiotherapy-based approach, although select patients may benefit from surgery, and systemic therapy could be considered in some circumstances. Although there are still no systemic therapies with an FDA indication for the treatment of breast cancer brain metastases, there is emerging evidence of activity of a number of regimens, and there are an increasing number of clinical trials targeting this patient population.
Acknowledgments
Funding support from the Breast Cancer Research Foundation.
Disclosures:
Research support: Genentech, BoehringerIngelheim, GlaxoSmithKline, Geron, Bayer
Consultant: GlaxoSmithKline (<$10K), Novartis ($10K), Geron (uncompensated), to-BBB (uncompensated)
References
1. Zimm S, Wampler GL, Stablein D, et al (1981) Intracerebral metastases in solid-tumor patients: natural history and results of treatment Cancer 48 384–94 DOI: 10.1002/1097-0142(19810715)48:2<;384::AID-CNCR2820480227>;3.0.CO;2-8 PMID: 7237407
2. Barnholtz-Sloan JS, Sloan AE, Davis FG, et al (2004) Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System J Clin Oncol 22 2865–72 DOI: 10.1200/JCO.2004.12.149 PMID: 15254054
3. Pestalozzi BC, Zahrieh D, Price KN, et al (2006) Identifying breast cancer patients at risk for Central Nervous System (CNS) metastases in trials of the International Breast Cancer Study Group (IBCSG) Ann Oncol 17 935–44 DOI: 10.1093/annonc/mdl064 PMID: 16603601
4. Lin NU, Vanderplas A, Hughes ME, et al (2012) Clinicopathologic features, patterns of recurrence, and survival among women with triple-negative breast cancer in the National Comprehensive Cancer Network Cancer DOI: 10.1002/cncr.27581 PMID: 23270461 PMCID: 3492207
5. Arvold ND, Oh KS, Niemierko A, et al (2012) Brain metastases after breast-conserving therapy and systemic therapy: incidence and characteristics by biologic subtype Breast Cancer Res Treat DOI: 10.1007/s10549-012-2243-x
6. Bendell JC, Domchek SM, Burstein HJ, et al (2003) Central nervous system metastases in women who receive trastuzumabbased therapy for metastatic breast carcinoma Cancer 97 2972–7 DOI: 10.1002/cncr.11436 PMID: 12784331
7. Brufsky AM, Mayer M, Rugo HS, et al (2011) Central nervous system metastases in patients with HER2-positive metastatic breast cancer: incidence, treatment, and survival in patients from registHER Clin Cancer Res 17 4834–43 DOI: 10.1158/1078-0432.CCR-10-2962 PMID: 21768129
8. Kennecke H, Yerushalmi R, Woods R, et al (2010) Metastatic Behavior of Breast Cancer Subtypes J Clin Oncol DOI: 10.1200/JCO.2009.25.9820
9. Vaz-Luis I, Ottesen RA, Hughes ME, et al (2012) Impact of hormone receptor status on patterns of recurrence and clinical outcomes among patients with human epidermal growth factor-2-positive breast cancer in the National Comprehensive Cancer Network: a prospective cohort study Breast Cancer Res 14 R129 DOI: 10.1186/bcr3324 PMID: 23025714
10. Berghoff A, Bago-Horvath Z, De Vries C, et al (2012) Brain metastases free survival differs between breast cancer subtypes Br J Cancer 106 440–6 DOI: 10.1038/bjc.2011.597 PMID: 22233926 PMCID: 3273356
11. Fokstuen T, Wilking N, Rutqvist LE, et al (2000) Radiation therapy in the management of brain metastases from breast cancer Breast Cancer Res Treat 62 211–6 DOI: 10.1023/A:1006486423827 PMID: 11072785
12. Sperduto PW, Kased N, Roberge D, et al (2011) Effect of Tumor Subtype on Survival and the Graded Prognostic Assessment for Patients With Breast Cancer and Brain Metastases Int J Radiat Oncol Biol Phys DOI: 10.1002/1097-0142(19810715)48:2<384::AIDCNCR2820480227>3.0.CO;2-8 PMID: 7237407
13. Sperduto PW, Kased N, Roberge D, et al (2011) Summary Report on the Graded Prognostic Assessment: An Accurate and Facile Diagnosis-Specific Tool to Estimate Survival for Patients With Brain Metastases J Clin Oncol DOI: 10.1200/JCO.2011.38.0527
14. Anders CK, Deal AM, Miller CR, et al (2011) The prognostic contribution of clinical breast cancer subtype, age, and race among patients with breast cancer brain metastases Cancer 117 1602–11 DOI: 10.1002/cncr.25746 PMID: 21472708
15. Melisko ME, Moore DH, Sneed PK, et al (2008) Brain metastases in breast cancer: clinical and pathologic characteristics associated with improvements in survival J Neurooncol 88 359–65 DOI: 10.1007/s11060-008-9578-5 PMID: 18398574
16. Eichler AF, Kuter I, Ryan P, et al (2008) Survival in patients with brain metastases from breast cancer: the importance of HER-2 status Cancer 112 2359–67 DOI: 10.1002/cncr.23468 PMID: 18361426
17. Lin NU, Claus E, Sohl J, et al (2008) Sites of distant recurrence and clinical outcomes in patients with metastatic triple-negative breast cancer: high incidence of central nervous system metastases Cancer 113 2638–45 DOI: 10.1002/cncr.23930 PMID: 18833576
18. Niwinska A, Murawska M, Pogoda K (2010) Breast cancer subtypes and response to systemic treatment after whole-brain radiotherapy in patients with brain metastases J Clin Oncol 28 abstract 1029 DOI: 10.1002/cncr.25391 PMID: 20549816
19. Dawood S, Broglio K, Esteva FJ, et al (2008) Defining prognosis for women with breast cancer and CNS metastases by HER2 status Ann Oncol 19 1242 –8 DOI: 10.1093/annonc/mdn036 PMID: 18334512
20. National Comprehensive Cancer Network (NCCN) (2012) Clinical practice guidelines in oncology: Central Nervous System Cancer available from www.nccn.org/professionals/physician_gls/pdf/cns
21. Linskey ME, Kalkanis SN (2010) Evidence-linked, clinical practice guidelines-getting serious; getting professional J Neurooncol 96 1–5 DOI: 10.1007/s11060-009-0070-7 PMID: 2810238
22. Trudeau ME, Crump M, Charpentier D, et al (2006) Temozolomide in metastatic breast cancer (MBC): a phase II trial of the National Cancer Institute of Canada - Clinical Trials Group (NCIC-CTG). Ann Oncol 17 952–6 DOI: 10.1093/annonc/mdl056 PMID: 16565212
23. Siena S, Landonio G, Beaietta E (2003) Multicenter phase II study of temozolomide therapy for brain metastasis in patients with malignant melanoma, breast cancer, and non-small cell lung cancer Proc Am Soc Clin Oncol 22 abstract 407
24. Rivera E, Meyers C, Groves M, et al (2006) Phase I study of capecitabine in combination with temozolomide in the treatment of patients with brain metastases from breast carcinoma Cancer 107 1348–54 DOI: 10.1002/cncr.22127 PMID: 16909414
25. Ekenel M, Hormigo AM, Peak S, et al (2007) Capecitabine therapy of central nervous system metastases from breast cancer J Neurooncol 85 223–7 DOI: 10.1007/s11060-007-9409-0 PMID: 17611719
26. Christodoulou C, Bafaloukos D, Linardou H, et al (2005) Temozolomide (TMZ) combined with cisplatin (CDDP) in patients with brain metastases from solid tumors: a Hellenic Cooperative Oncology Group (HeCOG) Phase II study J Neurooncol 71 61–5 DOI: 10.1007/s11060-004-9176-0 PMID: 15719277
27. Franciosi V, Cocconi G, Michiara M, et al (1999) Front-line chemotherapy with cisplatin and etoposide for patients with brain metastases from breast carcinoma, nonsmall cell lung carcinoma, or malignant melanoma: a prospective study Cancer 85 1599–605 DOI: 10.1002/(SICI)1097-0142(19990401)85:7<;1599::AID-CNCR23>;3.0.CO;2-# PMID: 10193952
28. Gradishar WJ, Stephenson P, Glover DJ, et al (2001) A Phase II trial of cisplatin plus WR-2721 (amifostine) for metastatic breast carcinoma: an Eastern Cooperative Oncology Group Study (E8188). Cancer 92 2517–22 DOI: 10.1002/1097-0142-(20011115)92:10<;2517::AID-CNCR1602>;3.0.CO;2-Z PMID: 11745184
29. Peereboom DM, Supko JG, Carson KA, et al (2010) A phase I/II trial and pharmacokinetic study of ixabepilone in adult patients with recurrent high-grade gliomas J Neurooncol 100 261–8 DOI: 10.1007/s11060-010-0190-0 PMID: 20449631
30. Hoffmann J, Fichtner I, Lemm M, et al (2009) Sagopilone crosses the blood-brain barrier in vivo to inhibit brain tumor growth and metastases Neurooncol 11 158–66 DOI: 10.1215/15228517-2008-072 PMCID: PMC2718987
31. Freedman RA, Bullitt E, Sun L, et al (2011) A Phase II Study of Sagopilone (ZK 219477; ZK-EPO) in Patients With Breast Cancer and Brain Metastases Clin Breast Cancer DOI: 10.1016/j.clbc.2011.03.024
32. Murphy C, Nulsen B, Rump M (2009) Phase II trial of patupilone in patients (pts) with breast cancer brain metastases (BCBM) progressing or recurring after whole brain radiotherapy (WBXRT). ASCO Breast Cancer Symp
33. Lassman AB, Abrey LE, Shah GD, et al (2006) Systemic high-dose intravenous methotrexate for central nervous system metastases J Neurooncol 78 255–60 DOI: 10.1007/s11060-005-9044-6
34. Boogerd W, Dalesio O, Bais EM, et al (1992) Response of brain metastases from breast cancer to systemic chemotherapy Cancer 69 972–80 DOI: 10.1002/1097-0142(19920215)69:4<;972::AID-CNCR2820690423>;3.0.CO;2-P PMID: 1735089
35. Salvati M, Cervoni L, Innocenzi G, et al (1993) Prolonged stabilization of multiple and single brain metastases from breast cancer with tamoxifen Report of three cases Tumori 79 359–62 PMID: 8116083
36. Madhup R, Kirti S, Bhatt ML, et al (2006) Letrozole for brain and scalp metastases from breast canceróa case report Breast 15 440–2 DOI: 10.1016/j.breast.2005.07.006
37. Stewart DJ, Dahrouge S (1995) Response of brain metastases from breast cancer to megestrol acetate: a case report J Neurooncol 24 299–301 DOI: 10.1007/BF01052847 PMID: 7595761
38. Pestalozzi BC, Brignoli S (2000) Trastuzumab in CSF J Clin Oncol 18 2349–51 PMID: 10829059
39. Lin NU, Carey LA, Liu MC, et al (2008) Phase II trial of lapatinib for brain metastases in patients with human epidermal growth factor receptor 2-positive breast cancer J Clin Oncol 26 1993–9 DOI: 10.1200/JCO.2007.12.3588 PMID: 18421051
40. Lin NU, Dieras V, Paul D, et al (2009) Multicenter phase II study of lapatinib in patients with brain metastases from HER2-positive breast cancer Clin Cancer Res 15 1452–9 DOI: 10.1158/1078-0432.CCR-08-1080 PMID: 19228746
41. Boccardo F, Kaufman B, Baselga J, et al (2008) Evaluation of lapatinib (Lap) plus capecitabine (Cap) in patients with brain metastses (BM) from HER2 breast cancer (BC) enrolled in the Lapatinib Expanded Access Program (LEAP) and French Authorisation Temporaire d’Utilisation (ATU). J Clin Oncol 26 abstract 1094
42. Metro G, Foglietta J, Russillo M, et al (2010) Clinical outcome of patients with brain metastases from HER2-positive breast cancer treated with lapatinib and capecitabine Ann Oncol 22 625–30 DOI: 10.1093/annonc/mdq434 PMID: 20724575
43. Sutherland S, Ashley S, Miles D, et al Treatment of HER2-positive metastatic breast cancer with lapatinib and capecitabine in the lapatinib expanded access programme, including efficacy in brain metastases--the UK experience Br J Cancer 102 995–1002 PMID: 20179708 PMCID: 2844035
44. Lin NU, Eierman W, Greil R, et al (2011) Randomized phase II study of lapatinib plus capecitabine or lapatinib plus topotecan for patients with HER2-positive breast cancer brain metastases J Neurooncol DOI: 10.1007/s11060-011-0629-y PMCID: 3097340
45. Bachelot T, Romieu CG, Campone M, et al (2011) LANDSCAPE: An FNCLCC phase II study with lapatinib (L) and capecitabine (C) in patients with brain metastases (BM) from HER2-positive( ) metastatic breast cancer (MBC) before whole-brain radiotherapy (WBR). J Clin Oncol 29 abstract 209
46. Pivot X, Semiglazov V, Żurawski B, et al (2012) CEREBEL (EGF111438): An open label randomized phase III study comparing the incidence of CNS metastases in patients (pts) with HER2 Metastatic Breast Cancer (MBC), treated with Lapatinib plus Capecitabine (LC) versus Trastuzumab plus Capecitabine (TC) ESMO 2012 Annual Meeting: LBA11
47. Fitzgerald DP, Emerson DL, Qian Y, et al (2012) TPI-287, a New Taxane Family Member, Reduces the Brain Metastatic Colonization of Breast Cancer Cells Mol Cancer Ther 11 1959–67 DOI: 10.1158/1535-7163.MCT-12-0061 PMID: 22622283
48. Thomas FC, Taskar K, Rudraraju V, et al (2009) Uptake of ANG1005, a novel paclitaxel derivative, through the blood-brain barrier into brain and experimental brain metastases of breast cancer Pharm Res 26 2486–94 DOI: 10.1007/s11095-009-9964-5 PMID: 19774344
49. Regina A, Demeule M, Che C, et al (2008) Antitumour activity of ANG1005, a conjugate between paclitaxel and the new brain delivery vector Angiopep-2. Br J Pharmacol 155 185–97 DOI: 10.1038/bjp.2008.260 PMID: 18574456
50. Kurzrock R, Gabrail N, Chandhasin C, et al (2012) Safety, pharmacokinetics, and activity of GRN1005, a novel conjugate of angiopep-2, a peptide facilitating brain penetration, and paclitaxel, in patients with advanced solid tumors Mol Cancer Ther 11 308–16 DOI: 10.1158/1535-7163.MCT-11-0566
51. Burstein HJ, Sun Y, Dirix LY, et al (2010) Neratinib, an irreversible ErbB receptor tyrosine kinase inhibitor, in patients with advanced ErbB2-positive breast cancer J Clin Oncol 28 1301–7 DOI: 10.1200/JCO.2009.25.8707 PMID: 20142587
52. Lin NU, Winer EP, Wheatley D, et al (2012) A phase II study of afatinib (BIBW 2992), an irreversible ErbB family blocker, in patients with HER2-positive metastatic breast cancer progressing after trastuzumab Breast Cancer Res Treat 133 1057–65 DOI: 10.1007/s10549-012-2003-y PMID: 22418700 PMCID: 3387495
53. Bullitt E, Lin NU, Smith JK, et al (2007) Blood vessel morphologic changes depicted with MR angiography during treatment of brain metastases: a feasibility study Radiology 245 824–30 DOI: 10.1148/radiol.2453061889 PMID: 17954616
54. Kim LS, Huang S, Lu W, et al (2004) Vascular endothelial growth factor expression promotes the growth of breast cancer brain metastases in nude mice Clin Exp Metastasis 21 107–18 DOI: 10.1023/B:CLIN.0000024761.00373.55 PMID: 15168728
55. Besse B, Lasserre SF, Compton P, et al (2010) Bevacizumab safety in patients with central nervous system metastases Clin Cancer Res 16 269–78 DOI: 10.1158/1078-0432.CCR-09-2439
56. Lu YS, Chen WW, Ling CH, et al (2012) Bevacizumab, etoposide, and cisplatin (BEEP) in brain metastases of breast cancer progressing from radiotherapy: results of the first stage of a multicenter phase II study J Clin Oncol 30 abstract 1079 DOI: 23150706 PMCID: 3295566
57. Vredenburgh JJ, Desjardins A, Herndon JE, 2nd, et al (2007) Bevacizumab plus irinotecan in recurrent glioblastoma multiforme J Clin Oncol 25 4722–9 DOI: 10.1200/JCO.2007.12.2440 PMID: 17947719
58. Chamberlain MC (2011) Bevacizumab for the treatment of recurrent glioblastoma Clin Med Insights Oncol 5 117–29 DOI: 10.4137/CMO.S7232 PMID: 21603247 PMCID: 3095028
59. Wen PY, Macdonald DR, Reardon DA, et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group J Clin Oncol 28 1963–72 DOI: 10.1200/JCO.2009.26.3541 PMID: 20231676
60. Baselga J, Campone M, Piccart M, et al (2012) Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer N Engl J Med 366 520–9 DOI: 10.1056/NEJMoa1109653
61. Bachelot T, Bourgier C, Cropet C, et al (2012) Randomized Phase II Trial of Everolimus in Combination With Tamoxifen in Patients With Hormone Receptor-Positive, Human Epidermal Growth Factor Receptor 2-Negative Metastatic Breast Cancer With Prior Exposure to Aromatase Inhibitors: A GINECO Study J Clin Oncol 30 2718–24 DOI: 10.1200/JCO.2011.39.0708 PMID: 22565002
62. Morrow PK, Wulf GM, Ensor J, et al (2011) Phase I/II study of trastuzumab in combination with everolimus (RAD001) in patients with HER2-overexpressing metastatic breast cancer who progressed on trastuzumab-based therapy J Clin Oncol 29 3126–32 DOI: 10.1200/JCO.2010.32.2321 PMID: 21730275 PMCID: 3157979
63. Andre F, Campone M, O’Regan R, et al (2010) Phase I study of everolimus plus weekly paclitaxel and trastuzumab in patients with metastatic breast cancer pretreated with trastuzumab J Clin Oncol 28 5110–5 DOI: 10.1200/JCO.2009.27.8549 PMID: 20975068
64. Curran MP (2012) Everolimus: in patients with subependymal giant cell astrocytoma associated with tuberous sclerosis complex Paediatr Drugs 14 51–60 DOI: 10.2165/11207730-000000000-00000
65. Gonzalez-Angulo AM, Ferrer-Lozano J, Stemke-Hale K, et al (2011) PI3K pathway mutations and PTEN levels in primary and metastatic breast cancer Mol Cancer Ther 10 1093–101 DOI: 10.1158/1535-7163.MCT-10-1089 PMID: 21490305 PMCID: 3112276
66. Santarpia L, Qi Y, Stemke-Hale K, et al (2012) Mutation profiling identifies numerous rare drug targets and distinct mutation patterns in different clinical subtypes of breast cancers Breast Cancer Res Treat 134 333–43 DOI: 10.1007/s10549-012-2035-3 PMID: 22538770
67. Bendell JC, Rodon J, Burris HA, et al (2012) Phase I, dose-escalation study of BKM120, an oral pan-Class I PI3K inhibitor, in patients with advanced solid tumors J Clin Oncol 30 282–90 DOI: 10.1200/JCO.2011.36.1360
68. Krop IE, Saura C, Rodon Ahnert J, et al (2012) A phase I/IB dose-escalation study of BEZ235 in combination with trastuzumab in patients with PI3-kinase or PTEN altered HER2 metastatic breast cancer J Clin Oncol 30 abstract 508 PMID: 22649126
69. O’Shaughnessy J, Schwartzberg LS, Danso MA, et al (2011) A randomized phase III study of iniparib (BSI-201) in combination with gemcitabine/carboplatin (G/C) in metastatic triple-negative breast cancer (TNBC). J Clin Oncol 29 abstract 1007
70. Kaye SB, Lubinski J, Matulonis U, et al (2012) Phase II, open-label, randomized, multicenter study comparing the efficacy and safety of olaparib, a poly (ADP-ribose) polymerase inhibitor, and pegylated liposomal doxorubicin in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer J Clin Oncol 30 372–9 DOI: 10.1200/JCO.2011.36.9215
71. Foulkes WD, Stefansson IM, Chappuis PO, et al (2003) Germline BRCA1 mutations and a basal epithelial phenotype in breast cancer J Natl Cancer Inst 95 1482–5 DOI: 10.1093/jnci/djg050 PMID: 14519755
72. Lee LJ, Alexander B, Schnitt SJ, et al (2011) Clinical outcome of triple negative breast cancer in BRCA1 mutation carriers and noncarriers Cancer 117 3093–100 DOI: 10.1002/cncr.25911 PMID: 21264845
73. Wasserstrom WR, Glass JP, Posner JB (1982) Diagnosis and treatment of leptomeningeal metastases from solid tumors: experience with 90 patients Cancer 49 759–72 DOI: 10.1002/1097-0142(19820215)49:4<;759::AID-CNCR2820490427>;3.0.CO;2-7 PMID: 6895713
74. Nam BH, Kim SY, Han HS, et al (2008) Breast cancer subtypes and survival in patients with brain metastases Breast Cancer Res 10 R20 DOI: 10.1186/bcr1870 PMID: 18307763 PMCID: 2374976
75. Lamovec J, Bracko M (1991) Metastatic pattern of infiltrating lobular carcinoma of the breast: an autopsy study J Surg Oncol 48 28–33 DOI: 10.1002/jso.2930480106 PMID: 1653879
76. Gauthier H, Guilhaume M-N, Bidard FC, et al (2009) Meningeal carcinomatosis in patients with breast cancer: clinical features and prognostic factors in 91 patients from a single institution Cancer Res 69 abstract 1090
77. Altundag K, Bondy ML, Mirza NQ, et al (2007) Clinicopathologic characteristics and prognostic factors in 420 metastatic breast cancer patients with central nervous system metastasis Cancer 110 2640–7 DOI: 10.1002/cncr.23088 PMID: 17960791
78. Lee S, Ahn HK, Park YH, et al (2011) Leptomeningeal metastases from breast cancer: intrinsic subtypes may affect unique clinical manifestations Breast Cancer Res Treat 129 809–17 DOI: 10.1007/s10549-011-1682-0 PMID: 21785952
79. Jaeckle KA, Phuphanich S, Bent MJ, et al (2001) Intrathecal treatment of neoplastic meningitis due to breast cancer with a slow-release formulation of cytarabine Br J Cancer 84 157–63 DOI: 10.1054/bjoc.2000.1574 PMID: 11161370
80. Glantz MJ, Jaeckle KA, Chamberlain MC, et al (1999) A randomized controlled trial comparing intrathecal sustained-release cytarabine (DepoCyt) to intrathecal methotrexate in patients with neoplastic meningitis from solid tumors Clin Cancer Res 5 3394–402 PMID: 10589750
81. Ozdogan M, Samur M, Bozcuk HS, et al (2003) Durable remission of leptomeningeal metastasis of breast cancer with letrozole: a case report and implications of biomarkers on treatment selection Jpn J Clin Oncol 33 229–31 DOI: 10.1093/jjco/hyg046 PMID: 12865466
82. Tham YL, Hinckley L, Teh BS, et al (2006) Long-term clinical response in leptomeningeal metastases from breast cancer treated with capecitabine monotherapy: a case report Clin Breast Cancer 7 164–6 DOI: 10.3816/CBC.2006.n.028 PMID: 16800978
83. Tetef ML, Margolin KA, Doroshow JH, et al (2000) Pharmacokinetics and toxicity of high-dose intravenous methotrexate in the treatment of leptomeningeal carcinomatosis Cancer Chemother Pharmacol 46 19–26 DOI: 10.1007/s002800000118 PMID: 10912573
84. Lee SH, Kong DS, Seol HJ, et al (2011) Ventriculoperitoneal shunt for hydrocephalus caused by central nervous system metastasis J Neurooncol 104 545–51 DOI: 10.1007/s11060-010-0512-2 PMID: 21274592
85. Omuro AM, Lallana EC, Bilsky MH, et al (2005) Ventriculoperitoneal shunt in patients with leptomeningeal metastasis Neurology 64 1625–7 DOI: 10.1212/01.WNL.0000160396.69050.DC PMID: 15883329
86. Park IH, Kwon Y, Ro JY, et al (2010) Concordant HER2 status between metastatic breast cancer cells in CSF and primary breast cancer tissue Breast Cancer Res Treat 123 125–8 DOI: 10.1007/s10549-009-0627-3
87. Platini C, Long J, Walter S (2006) Meningeal carcinomatosis from breast cancer treated with intrathecal trastuzumab Lancet Oncol 7 778–80 DOI: 10.1016/S1470-2045(06)70864-6 PMID: 16945774
88. Stemmler HJ, Mengele K, Schmitt M, et al (2008) Intrathecal trastuzumab (Herceptin) and methotrexate for meningeal carcinomatosis in HER2-overexpressing metastatic breast cancer: a case report Anticancer Drugs 19 832–6 DOI: 10.1097/CAD.0b013e32830b58b0 PMID: 18690096
89. Stemmler HJ, Schmitt M, Harbeck N, et al (2006) Application of intrathecal trastuzumab (Herceptintrade mark) for treatment of meningeal carcinomatosis in HER2-overexpressing metastatic breast cancer Oncol Rep 15 1373–7 PMID: 16596213
90. Siderov J (2006) Care with intrathecal trastuzumab Lancet Oncol 7 888 DOI: 10.1016/S1470-2045(06)70917-2 PMID: 17081914
91. Baselga J, Cortes J, Kim SB, et al (2011) Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer N Engl J Med 366 109–19 DOI: 10.1056/NEJMoa111321 PMID: 22149875
92. Blackwell KL, Miles D, Gianni L, et al (2012) Primary results from EMILIA, a phase III study of trastuzumab emtansine (T-DM1) versus capecitabine (X) and lapatinib (L) in HER2-positive locally advanced or metastatic breast cancer (MBC) previously treated with trastuzumab (T) and a taxane J Clin Oncol 30 LBA1 PMID: 22689807
93. Krop IE, Beeram M, Modi S, et al (2010) Phase I study of trastuzumab-DM1, an HER2 antibody-drug conjugate, given every 3 weeks to patients with HER2-positive metastatic breast cancer J Clin Oncol 28 2698–704 DOI: 10.1200/JCO.2009.26.2071 PMID: 20421541
94. Chang EL, Wefel JS, Hess KR, et al (2009) Neurocognition in patients with brain metastases treated with radiosurgery or radio-surgery plus whole-brain irradiation: a randomised controlled trial Lancet Oncol DOI: 10.1016/S1470-2045(09)70263-3
95. Wu YJ, Muldoon LL, Gahramanov S, et al (2012) Targeting alpha(V)-integrins decreased metastasis and increased survival in a nude rat breast cancer brain metastasis model J Neurooncol 110 27–36 DOI: 10.1007/s11060-012-0942-0 PMID: 22842979
96. Gril B, Palmieri D, Qian Y, et al (2011) Pazopanib reveals a role for tumor cell B-Raf in the prevention of HER2 breast cancer brain metastasis Clin Cancer Res 17 142–53 DOI: 10.1158/1078-0432.CCR-10-1603 PMCID: 3059742
97. Qian Y, Hua E, Bisht K, et al (2011) Inhibition of Polo-like kinase 1 prevents the growth of metastatic breast cancer cells in the brain Clin Exp Metastasis 28 899–908 DOI: 10.1007/s10585-011-9421-9 PMID: 21953073
98. Palmieri D, Lockman PR, Thomas FC, et al (2009) Vorinostat inhibits brain metastatic colonization in a model of triple-negative breast cancer and induces DNA double-strand breaks Clin Cancer Res 15 6148–57 DOI: 10.1158/1078-0432.CCR-09-1039 PMID: 19789319
99. Iwamoto FM, Omuro AM, Raizer JJ, et al (2008) A phase II trial of vinorelbine and intensive temozolomide for patients with recurrent or progressive brain metastases J Neurooncol 87 85–90 DOI: 10.1007/s11060-007-9491-3
100. De Azambuja E, Lemort M, Rossari JR, et al (2011) Phase I study of lapatinib (L) and temozolomide (T) combination for the treatment of progressive brain metastases (BM) in HER2-positive metastatic breast cancer patients (Pts) (LAPTEM, LAP 111172). J Clin Oncol 29 abstract 570






