The role of palliative radiotherapy for haemostasis in unresectable gastric cancer: a single-institution experience
Cheng Lee Chaw1, Paddy G Niblock1, Cheng Shu Chaw2 and Douglas J Adamson1
1 Princess Alexandra Centre, Oncology Department, Ninewells Hospital & Medical School, Dundee DD1 9SY, Scotland, UK
2 Faculty of Applied Science, Department of Pharmacy, Health and Well Being, University of Sunderland SR1 3SD, UK
Correspondence to: Cheng Lee Chaw. Email: c.chaw@nhs.net
Abstract
Purpose: To evaluate the outcomes of patients with gastric cancer bleeding who had been treated with palliative radiotherapy with haemostatic intent.
Methods and materials: Fifty-two gastric cancer patients aged 52–92 years (median 78 years) with active bleeding or anaemia resulting from inoperable gastric cancer were treated with short-course radiotherapy. Responses to radiotherapy treatment were evaluated based on the changes of haemoglobin level, number of transfusions received before and after radiotherapy, and overall median survival.
Results: Thirty-nine (75%) patients received single 8 Gy fraction, and 13 (25%) patients received 20 Gy in five daily fractions. The need for transfusion was evaluable in 44 patients, and the response rate was 50%, with less requirement for blood transfusions within four weeks of radiotherapy. There was also an increase in mean haemoglobin level (0.66 ± 1.12 g/dl, p < 0.01) after radiotherapy in 35 evaluable patients. The overall median survival (calculated from last day of treatment to date of death) was 160 days (95% CI of 119–201 days), making actuarial 12-month survival 15%.
Conclusion: Palliative short-course radiotherapy is a reasonably effective treatment that can provide durable palliation of bleeding in gastric cancer.
Keywords: bleeding, gastric cancer, haemostasis, palliative, radiotherapy.
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Gastric cancer is the fourth most common malignancy in men and the fifth most common malignancy in women globally. It accounts for 8% of the total number of cases of cancer and 10% of annual deaths from cancer worldwide [1]. The current curative treatment for gastric cancer is surgery, but most patients are diagnosed with locally advanced or metastatic disease at presentation. The prognosis of these patients is poor, and a five-year survival rate of 10–20% has been reported [2, 3].
The local symptoms caused by gastric cancer are pain, obstruction (dysphagia, vomiting), and bleeding (melaena, haematemesis). Overt gastrointestinal bleeding from gastric cancer at presentation is uncommon but does occur in up to 10% of patients [4], and this can be a very distressing symptom for both patients and their family members. Chronic bleeding from gastric cancer can cause anaemia and subsequently fatigue and anorexia. Anaemia can also interfere with symptom control, and control of bleeding is important to improve the quality of life in these patients [5].
There are several treatment modalities for gastric bleeding, including palliative surgical resection, endoscopic treatment, and radiotherapy, but little research into which treatment is the most effective option has been performed.
Gastric cancer bleeding can be relieved by palliative resection; however, only fit patients can undergo surgery, and it is associated with significant mortality approaching 10% as well as morbidity in about a third of patients [6–8]. Surgery may provide rapid relief from symptoms, but prolongation of survival is noted in only 6% of patients and improvement in the quality of life following palliative resection is unclear [7].
Endoscopic intervention including argon plasma coagulation has been reported to achieve haemostasis in 67% of patients with gastroduodenal tumour bleeding [9]; however, this is associated with risk of perforation (5–15%), and the re-bleeding rate is high [9].
Radiotherapy has been used to palliate bleeding from other malignant tumours such as lung, cervical, and bladder cancers [10, 11]. Radiation causes denudation of the intima of the blood vessels supplying the tumour, leading to capillary necrosis and the formation of thrombosis, which blocks the lumen of blood vessels, achieving haemostasis [4]. The response rate to this treatment has been reported to be as high as 50–90% [2–4, 12, 13], but the retrospective nature of these reports needs to be taken into account.
Here, we report the results of a single-centre retrospective study on ‘short-course’ radiotherapy to palliate patients with symptomatic locally advanced gastric cancer and evaluate its effectiveness in relieving symptoms of bleeding or anaemia.
Methods and materials
Patients
Two hundred and ninety-eight consecutive patients who were diagnosed with gastric cancer between May 2001 and December 2010 and referred for oncological treatment were analysed retrospectively. The patients regarded as having gastric cancer on endoscopy findings and who had a pathological diagnosis of only high-grade dysplasia were included. Furthermore, those patients who presented with gastrointestinal bleeding or anaemia and were treated with short-course palliative radiotherapy were eligible for inclusion in this retrospective study. The diagnosis of gastrointestinal bleeding was established either by endoscopic findings or from the clinical symptoms of patients (melaena or haemetemesis) supported by evidence of a low or falling haemoglobin level. In our region, palliative radiotherapy to the stomach alone is generally reserved for patients unfit for other treatment strategies such as chemotherapy or surgery.
The patients’ gender, age, histology, stage, and treatment factors (use of chemotherapy before and after radiation treatment, postradiation transfusion and mean haemoglobin level before and after radiotherapy) as well as radiotherapy parameters (field arrangements, total dose, dose fractionation regimens) were extracted from the patient records.
Treatment procedures
All patients with gastrointestinal bleeding received external beam radiotherapy using high-energy (6 or 10 MV) photons from a linear accelerator. The patients were treated supine with parallel-opposed pair of anterior/posterior fields, and the radiotherapy was delivered either as a single fraction of 8 or 20 Gy in five daily fractions over one week. The 20 Gy treatment in five fractions was usually given to patients with a better performance status or those who had dysphagia caused by tumour obstructing the lower oesophagus.
Response assessment
The efficacy of the short-course radiotherapy was evaluated. With reference to previous studies by other groups [2, 4, 12, 13], we defined a positive radiotherapy response if
• patients subsequently required a blood transfusion but did not need one in the four weeks after radiotherapy was given (‘transfusion group’) or
• patients did not require transfusion at all and survived for three months or more (‘Non-transfusion group’).
We also evaluated the efficacy of the short-course radiotherapy on the basis of improvement in haemoglobin levels in patients after the radiotherapy.
The difference between the mean haemoglobin values before and after radiotherapy was therefore reviewed. The mean preradiotherapy haemoglobin level was obtained from the blood test results from the day of admission to hospital with gastrointestinal bleeding to the first fraction of radiotherapy. Hence the value reflects the underlying disease and effects of transfusion only. The mean postradiotherapy haemoglobin level reflects the four-week period starting with the last fraction of radiotherapy. This parameter was used to measure the effect of radiotherapy: a decrease from the mean preradiotherapy haemoglobin level was considered to be indicative of re-bleeding.
We were also keen to determine the overall survival of this group of patients, which we calculated from the day the patient received the last fraction of radiotherapy.
Statistical analysis
Statistical analyses were carried out with the SPSS (Statistical Package for Social Sciences) version 18. Survival was estimated by the Kaplan–Meier method. Univariate analysis with log rank test was performed to study different factors correlating with survival. The alteration of haemoglobin levels was analysed with paired t-test; p-values <0.05 were considered to be statistically significant.
Results
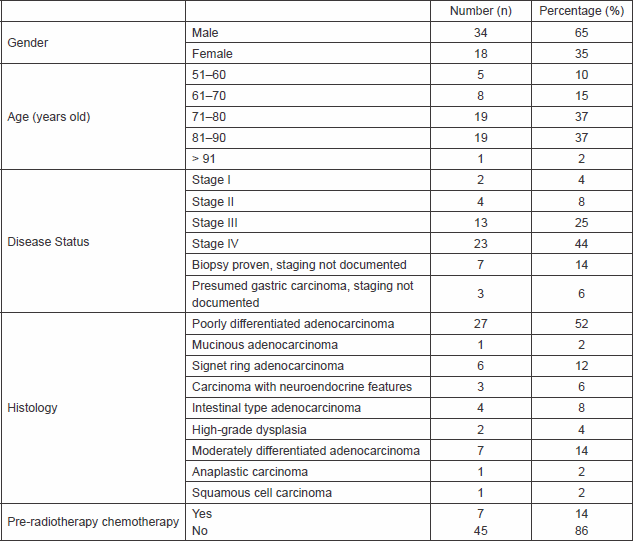
Sixty-four of the 298 patients were referred for palliative radiotherapy for haemostatic intent. However, 12 of the 64 patients did not receive palliative radiotherapy after the initial consultation either because the oncologist assessed that they were not fit for treatment or there was no immediate indication for the treatment. Fifty-two patients received palliative radiotherapy for control of bleeding symptoms of locally advanced gastric cancer. The clinical details of these 52 patients are summarised in Table 1. The median age of the patients was 78 years (range 52–92 years); they were predominantly male (n = 34, 65%). The median age for the male patients was 76 years (range 52–90 years), and the median age for the female patients (n = 18, 35%) was 81 years (range 61–92 years).
Thirty-nine (75%) patients received a single 8 Gy treatment and 13 (25%) patients received 20 Gy in five daily fractions. Two patients were subsequently re-treated with palliative radiotherapy (both re-treatments were with an 8 Gy single fraction). One of them received 8 Gy single fraction on two occasions for haemostatic intent, and these radiotherapy treatments were given eight months apart. The other patient was initially treated with 20 Gy in five fractions for palliation of dysphagia, and further radiotherapy treatment was given one year later for haemostatic intent. There were seven (14%) patients treated with systemic chemotherapy before receiving short-course radiotherapy for controlling the bleeding symptom, and only three (6%) patients received systemic treatment after palliative radiotherapy for control of the disease, as would be expected for an unfit cohort of patients.
Table 1. Patients’ demographics.
Treatment outcomes
Response to radiotherapy
Eight of the 52 patients had received palliative radiotherapy for haemostasis but did not have any documentation of transfusion data. Hence the response to short-course treatment could not be evaluated in these patients. The 44 remaining evaluable patients were categorised into two groups in terms of transfusion requirement (Table 2).
Non-transfusion group: Fifteen patients did not receive any transfusion after palliative radiotherapy. One patient died within a week and six patients died within a month. The remained eight patients survived beyond three months and did not require transfusion; hence the response rate to radiotherapy in this group was 53% (8/15).
Transfusion group: Twenty-nine patients required postradiation transfusion. Of the 29 patients, fifteen (52%) required transfusion within four weeks; hence palliative radiotherapy was deemed ineffective. Fourteen of the 29 patients required transfusion at least a month or more after radiotherapy. Hence, the ‘response rate’ to radiotherapy was 48% (14 of 29). We also reviewed the number of transfusions that patients required until they died (Table 2). Thirteen (45%) of the 29 patients required one transfusion before they died, and the rest required multiple transfusions before death.
Thus, 22 of 44 patients (50%), that is, 14 from transfusion group and 8 from non-transfusion group, responded to radiotherapy.
Mean haemoglobin difference before and after radiotherapy
Only 35 patients had complete preradiotherapy and postradiotherapy haemoglobin levels recorded, and in these patients, the differences in mean values were evaluated. The blood results of the remaining 17 patients were not evaluable due to incomplete documentation. The mean haemoglobin value before palliative radiotherapy for these 35 patients was 9.53 g/dl, whereas the mean haemoglobin level after radiotherapy treatment was 10.2 g/dl. There was a statistically significant improvement in mean haemoglobin level after radiotherapy compared with the preradiotherapy haemoglobin (p < 0.01, Figure 1).
Table 2. Number of transfusions required by 44 patients before they died.
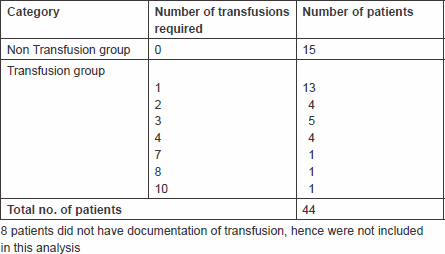
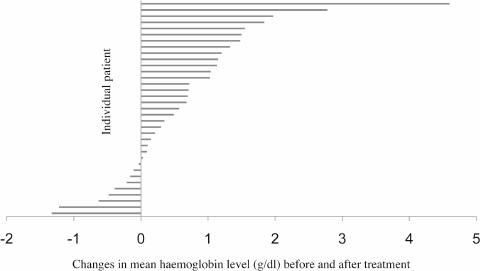
Figure 1. Differences in mean haemoglobin level before and after palliative radiotherapy in 35 patients. Twenty-six patients had an increase in mean haemoglobin level after treatment, whereas nine patients had a fall. The overall increment in mean haemoglobin level was 0.66 ±1.12g/dl in 35 patients, p < 0.01.
Median survival
The overall median survival of patients in this study was 160 days (95% CI of 119–201 days) ranging from 6 days to 1053 days. At the time of analysis, four patients were still alive (Figure 2). The median survival of those 12 patients who did not receive palliative radiotherapy was 49 days (95% CI of 18–80 days).
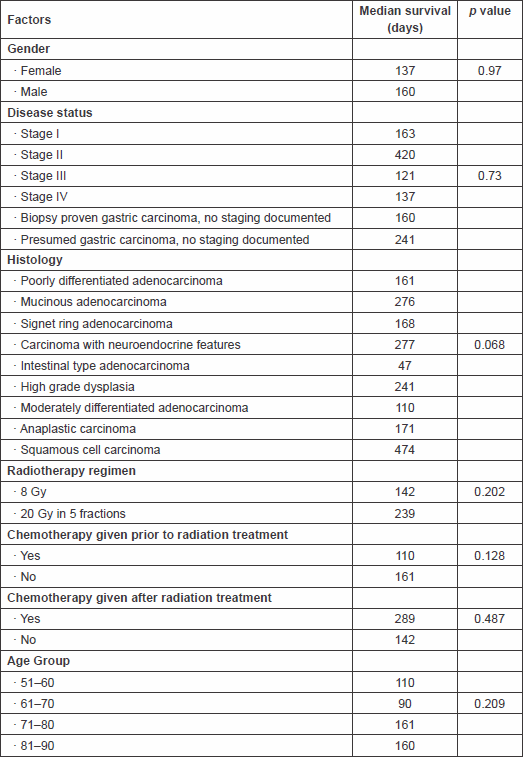
The survival rates at 3, 6, and 12 months were 65%, 23%, and 15%, respectively. Univariate analysis on different pretreatment factors (stage, histology, radiotherapy dose, etc.) that might have an impact on survival was performed, but as shown in Table 3, no statistically significant difference was found.
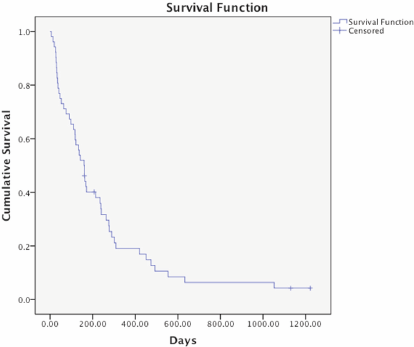
Figure 2. Kaplan–Meier survival curve of 52 patients receiving palliative radiotherapy. The overall median survival of these patients was 160 days (95% CI 119–201 days). Four patients were alive and censored at the time of analysis.
Table 3. Factors that could potentially have an impact on median survival time.
Discussion
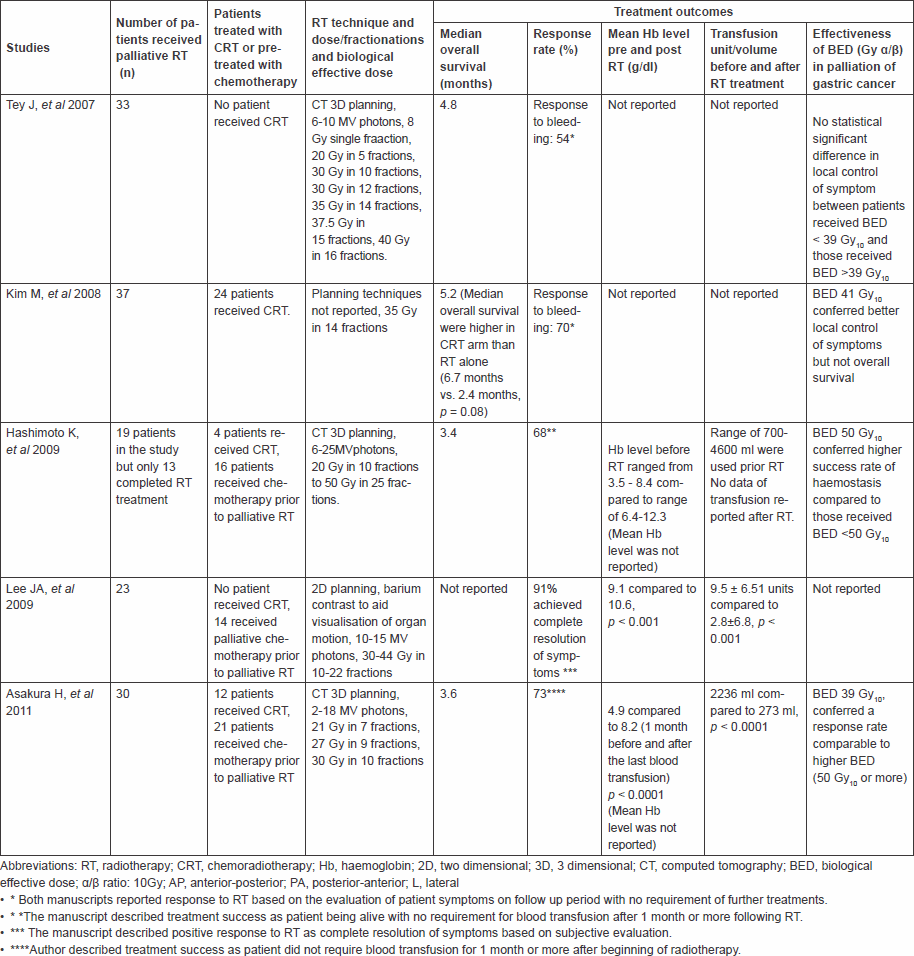
There are five retrospective studies in recent years that have described the benefits of using radiotherapy to palliate bleeding in patients with unresectable gastric cancer [2–4, 12, 13]. The response rates for controlling bleeding in these studies varied from 50–90%, such wide range is due to the difference in the definition of treatment success. The reported overall median survival with palliative radiotherapy varied from 3–5.2 months (Table 4). These retrospective studies, however, had relatively small sample sizes of 19–37 patients, and no randomised controlled trial to evaluate the efficacy of short-course radiotherapy on the palliation of symptomatic gastric cancer has been published.
Our study evaluated the outcomes of 52 patients with gastric cancer bleeding treated with radiotherapy in a single regional centre over a ten-year period, with the sample size larger than those reported previously [2–4, 12, 13]. Like all retrospective works, the results need to be interpreted with caution. It is difficult to obtain retrospective data for each patient in a standard way, and the information on performance status or symptom improvement may be hard to obtain in all cases. The strengths of our series are that all patients in our catchment population (approximately 400,000) were treated in a single oncology radiotherapy centre by only a few consultants using a similar technique. We therefore believe that these results reflect outcomes typical for all-comers within a population rather than subgroups selected for study, for example, in clinical trials with specific exclusion criteria. We believe case ascertainment to be high for similar reasons of easy data retrieval from the comprehensive and mainly electronic records of one regional Scottish centre.
Table 4: Summary of the 6 retrospective studies on efficacy of palliative radiotherapy treatment in advanced gastric cancer.
The majority (73%) of the patients were elderly patients over 70 years of age and were unfit to receive radical treatment or palliative systemic treatment. A small number of patients in our study had been diagnosed with early-stage disease (stages I and II) but owing to significant co-morbidities were fit neither for surgery nor for palliative chemotherapy; therefore palliative radiotherapy was given for patients who were symptomatic with bleeding. As none of our patients received concurrent chemoradiation treatment, and only 14% of patients (7 of 52) received chemotherapy prior to radiotherapy, this has provided a useful description of the natural history of patients who are treated with radiotherapy alone. Six percent (3 of 52) of these patients received systemic treatment after radiotherapy. The overall median survival in our group of 160 days was unlikely to be confounded by the use of other treatments, and this result was similar to the result shown by Tey et al [12].
Different dose fractionation schedules varying from single 8 Gy to dose of 50 Gy in 25 fractions have been published in previous papers [2–4, 12, 13]. Two of the studies suggested the use of higher biological effective dose (BED) of over 39 Gy [2, 3] would provide better local control and also survival. Tey et al suggested that there was no statistically significant difference in median survival between lower and higher BED, but the result favoured the use of higher dose, which tended to confer longer survival [12]. Our study could lend support to this, as the median survival of the 39 patients receiving a single 8 Gy versus the 13 patients treated with 20 Gy in five fractions was 142 versus 239 days, respectively, p = 0.202 (Table 3); however, this finding is likely be confounded by the selection of patients with better performance status for the longer fractionation and by the preferences of the radiotherapist. Assuming alpha/beta (α/β) ratio was 10, the BED in our study ranged from 14.4 Gy (8 Gy single fraction) to 28 Gy (20 Gy in five fractions), which was at least 1.5 times less than the dose applied in the previous studies [2, 3, 12, 13], although the results appear similar. Our study has shown that the use of lower dose treatment has shown a similar median survival compared with higher dose treatment in this, generally unfit, patient cohort.
Previous studies evaluated the response of radiotherapy by reviewing the pre- and postradiotherapy haemoglobin levels and also the reduction of transfusion units or amount of transfused blood after radiotherapy [2, 4, 13]. However, it is unclear from these studies at what stage and how often after radiotherapy, the haemoglobin level was checked. Lee et al had commented on the challenging aspects of assessing the efficacy of radiotherapy [4]. This correctly states that a single haemoglobin level as an end point to evaluate response is flawed, as haemoglobin level does not reflect acute gastrointestinal bleeding directly, and because the level of haemoglobin varies with gender and also comorbidity. Therefore, Lee et al. had suggested the use of mean value of haemoglobin level and mean value of reduction in transfusion units required postradiotherapy for a period of three months. A mean haemoglobin level is more likely to reflect the true effect of radiotherapy.
We evaluated the response to treatment by calculating the mean value of haemoglobin levels preradiotherapy and postradiotherapy. There was a statistically significant increase in the level of haemoglobin after radiotherapy. This finding was comparable with the results reported in the previous studies (Table 4), which showed an increase in the level of postradiotherapy haemoglobin although the values did not return to the reference range in all cases. The likely explanation was that anaemia persisted because of underlying disease and comorbidity of patients, even though radiotherapy has achieved haemostatic effects.
Most studies deemed radiotherapy treatment effective if patients did not require transfusion within four weeks or more after radiotherapy [2, 4, 13] in which the cutoff point of four weeks seems arbitrary. For the purpose of comparison of response to short-course radiotherapy, we adopted the similar approach. We assessed the response of short-course radiotherapy by recording the need of transfusion within four weeks after completing radiotherapy treatment. In our study, 15 patients did not require transfusion; however, half of this non-transfusion group died within a month; therefore the radiotherapy treatment was not considered effective. For the ‘transfusion group’ (n = 29), which comprised patients receiving transfusion within four weeks after radiotherapy, nearly half of them (13 of 29) required only one transfusion before they died. The rest required multiple transfusions and three patients of this group required transfusion on more than five occasions, so it is evident that the survival of these patients was prolonged by the transfusion treatment, but initial haemostasis was achieved by radiotherapy. It is notable that in a group perceived to have a dire outlook, almost one in seven patients were alive one year after completing radiotherapy, and it may be that this simple treatment provides excellent palliation at small cost to the individual.
To date, there are no accurate measurable parameters to evaluate the effectiveness of radiotherapy for gastric cancer bleeding. Theoretically, haemostatic effects can be accurately assessed by direct view of the bleeding source through an endoscopic approach in conjunction with examination of the haemoglobin level at regular intervals. However, this approach is not suitable or particularly helpful in patients who have advanced disease. In our retrospective study, the change in mean haemoglobin and also the requirement of transfusion were used as surrogates to assess efficacy of treatment.
In conclusion, our study suggests that radiotherapy is a good option for patients with gastric cancer bleeding as shown by the overall median survival and the improvement of mean haemoglobin levels after radiotherapy. Either a single 8 Gy fraction or 20 Gy in five fractions, corresponding to a BED of 14.4to 28 Gy, appears to provide similar response rates to those treatments of higher BED (Table 4). Further investigations with control group not receiving radiotherapy for palliation of gastric cancer bleeding and larger sample size would provide more evidence of the efficacy of this treatment in controlling symptomatic bleeding, and as in most palliative treatments, it would be important to assess the effect of treatment on quality of life, although such data are difficult to collect from patients who are frail and unfit.
RT: radiotherapy; CRT: chemoradiation; Hb: haemoglobin; 2D: two dimensional; 3D: three dimensional; CT: computed tomography; BED: biological effective dose; PA: posterior-anterior; L: lateral
Conflict of interest notification and funding disclosure
All the authors of this manuscript declare that there are no financial disclosures or conflicts of interest that could be perceived as prejudicing the impartiality of the research reported.
Acknowledgments
The authors would like to thank Dr R. Casasola, Dr M. Ferguson, Prof. A Munro who provided information on patients in this study. They would also like to thank Dr Z Shi for his help in statistical analysis.
References
1. Thrumurthy SG, Chaudry MA, Hochhauser D, et al (2013) The diagnosis and management of Gastric Cancer BMJ 347 f6367 DOI: 10.1136/bmj.f6367 PMID: 24191271
2. Hashimoto K, Mayahara H, Takashima A, et al (2009) Palliative radiation therapy for haemorrhage of unresectable gastric cancer: a single institute experience J Cancer Res Clin Oncol 135 1117–23 DOI: 10.1007/s00432-009-0553-0 PMID: 19205735
3. Kim MM, Rana V, Janjan NA, et al (2008) Clinical benefit of palliative radiation therapy in advanced gastric cancer Acta Oncologica 47 421–7 DOI: 10.1080/02841860701621233
4. Lee JA, Lim DH, Park W, et al (2009) Radiation therapy for gastric cancer bleeding Tumori 95 726–30
5. Pereira J and Phan T (2004) Management of bleeding in patients with advanced cancer Oncologist 9(5) 561–70 DOI: 10.1634/theoncologist.9-5-561 PMID: 15477642
6. Saidi RF, ReMine SG, DuDrik PS, et al (2006) Is there a role for palliative gastrectomy in patients with stage IV gastric cancer? World Surg 30 21–7 DOI: 10.1007/s00268-005-0129-3
7. Greenall MJ and Barr H (2000) Carcinoma of Stomach Oxford Textbook of Surgery 2nd edn, ed PJ Morris, WC Wood (Oxford University Press) pp 1325–6
8. Lupasco C, Andronic D, Ursulescu C, et al (2010) Palliative gastrectomy in patients with stage IV gastric cancer – our recent experience Chirurgia Bucur 105 473–6
9. Loftus EV, Alexander GL, Ahlquist DA, et al (1994) Endoscopic Treatment of Major bleeding from advanced gastroduodenal malignant lesions Mayo Clin Proc 69 736–40 DOI: 10.1016/S0025-6196(12)61090-8 PMID: 8035627
10. Ferries FD, Bezjak A and Rosenthal SG (2001) The palliative uses of radiation therapy in surgical oncology patients Surg Oncol Clin N Am 10 185–201
11. Onsrud M, Hagen B and Stricket T (2001) 10Gy single fraction pelvic irradiation for palliation and life prolongation in patients with cancer of cervix and corpus uteri Gynecol Oncol 82 167–71 DOI: 10.1006/gyno.2001.6233 PMID: 11426980
12. Tey J, Back MF, Shakespear TP, et al (2007) The role of palliative radiation therapy in symptomatic locally advanced gastric cancer Int J Radiation Oncology Biol Phys 67 385–8 DOI: 10.1016/j.ijrobp.2006.08.070
13. Asakura H, Hashimoto T, Haranda H, et al (2011) Palliative radiotherapy for bleeding from advanced gastic cancer: is a schedule of 30Gy in 10 fractions adequate J Cancer Res Clin Oncol 137 125–30 DOI: 10.1007/s00432-010-0866-z






