Haematological, renal and hepatic toxicity profiles of infusional 5-fluorouracil versus capecitabine in African gastrointestinal cancer patients: a retrospective cohort study
Tinashe Adrian Mazhindu1,2, Ntokozo Ndlovu1,2, Margaret Z Borok3, Kevin Grimes4 and Collen Masimirembwa2
1Department of Oncology, Medical Physics and Imaging Sciences, Faculty of Medicine and Health Sciences, University of Zimbabwe, Harare, Zimbabwe
2African Institute of Biomedical Science and Technology, Harare, Zimbabwe
3Department of Internal Medicine, Faculty of Medicine and Health Sciences, University of Zimbabwe, Harare, Zimbabwe
4Department of Chemical and Systems Biology, Stanford University School of Medicine, Stanford University, Stanford, CA 94305, USA
Abstract
Introduction: Infusional 5-fluorouracil (5-FU) or capecitabine are commonly used in the management of gastrointestinal tract. So far very few studies have evaluated non dermatological, neurological and intestinal treatment related toxicities between these two fluoropyrimidine in African patients – evaluating the healthcare burden related to hospitalisations, blood transfusions and use of granulocyte colon-stimulating factor (G-CSF) – all of which are costly and scarce in Africa.
Methods: We conducted a 10-year retrospective cohort study of black African patients with gastrointestinal cancer who received either 5-FU or capecitabine-based chemotherapy – extracted and analysed data on treatment related adverse events (TRAEs) incidence, severity and management.
Results: A total of 179 participants were analysed: 100 received 5-FU and 79 received capecitabine. The incidence of any TRAE was 75/100 (75%) in the 5-FU group and 51/79 (65%) in the capecitabine group relative risk (RR 0.86, 95% confidence interval (CI) 0.7–1.05; p = 0.15). Severe TRAEs occurred at similar rates: 33% for both groups (RR 0.99, 95% CI 0.7–1.52; p = 0.95). Haematological TRAEs were comparable except for neutropenia, which was less common with capecitabine (33% versus 52%, RR 0.63, 95% CI 0.43–0.91; p = 0.01). Severe anaemia occurred more frequently in the capecitabine group compared to the 5-FU group, with a RR of 2.6 (95% CI 1.02–6.55). Hypokalemia was also lower with capecitabine (6% versus 17%, RR 0.37, 95% CI 0.14–0.97; p = 0.04). Hospitalisation, G-CSF use and red blood cell transfusions were similar between groups, but treatment interruptions were less frequent with capecitabine (16% versus 50%, RR 0.33, 95% CI 0.19–0.56; p < 0.0001). Treatment completion rates were 60% for 5-FU and 54% for capecitabine (p = 0.10).
Conclusion: Both infusional 5-FU and capecitabine are generally well tolerated among African patients with gastrointestinal cancer. However, capecitabine is associated with a significantly lower risk of neutropenia and hypokalemia but a higher RR of severe anaemia with overall fewer incidents of treatment interruption.
Keywords: fluoropyrimidine toxicity, treatment side effects, cancer complication, African cancer
Correspondence to: Tinashe Adrian Mazhindu
Email: atmazhindu@gmail.com
Published: 07/05/2026
Received: 26/09/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Fluoropyrimidines such as 5-fluorouracil (5-FU) or capecitabine are commonly employed in the management of gastrointestinal tract (GIT) cancers, either as monotherapy or in combination regimens [1]. Capecitabine is an oral pro-drug that is enzymatically converted to 5-FU in-vivo via the activity of the enzymes carboxylesterase, cytidine deaminase and thymidine phosphorylase, which are encoded by the CES1, CDA and TYMS genes, respectively [2]. About 80% to 90% of administered or in-vivo formed 5-FU is rapidly metabolised into an inactive form by dihydropyrimidine dehydrogenase (DPD), an enzyme encoded by the DPYD gene [3]. The DPD-catalysed step is the rate-limiting stage in the catabolic pathway for 5-FU. Reduced or absent expression of DPD, caused by DPYD gene polymorphism, may increase the risk of toxicity or death. Therefore, pre-emptive DPYD pharmacogenomic testing is recommended for patients who are to receive fluoropyrimidines [4]. At present, genotyping for CES1, CDA or TYMS is not recommended for patients scheduled to receive capecitabine [5]. Currently, fluoropyrimidine recommendation guidelines are primarily derived from studies involving patients of European and Asian ancestry [6].
In the past 20 years, capecitabine has been widely utilised as a substitute for 5-FU in numerous chemotherapy regimens [7–9]. Toxicity comparison studies, mostly involving Europeans, have noted differences in risk for diarrhea, hand and foot syndrome and peripheral neuropathy side effects between infusional 5-FU and capecitabine [10, 11]. Based on these observations, numerous comparative studies into GIT, dermatological and neurological side effects incidence, severity and associated factors of capecitabine have been reported [11–13]. Studies comparing African Americans and Europeans have found inconsistent results and genetic associations regarding racial and ethnic differences in the incidence and severity of dermatological and neurological side effects from capecitabine [14, 15]. Limited studies reviewed the question of 5-FU versus capecitabine in patients of African ancestry, a population that has the widest genetic diversity [16]. Similarly, studies showing capecitabine to be cost-effective compared to infusional 5-FU, due to lower total patient costs like reduced hospitalisation or central vein catheter insertion costs, were conducted in non-African settings [17]. In Africa, much like the rest of the world fluoropyrimidines are recommended in the National Comprehensive Cancer Network® harmonised guidelines for sub-Saharan Africa [18, 19].
There is a lack of specific studies comparing the incidence and severity of haematological, hepatic and renal treatment-related adverse events (TRAEs) between infusional 5-FU and capecitabine specifically in African patients. The distinct toxicity profiles of 5-FU and capecitabine are attributable to differences in their administration routes, metabolic processing – capecitabine being an oral prodrug requiring in vivo biotransformation – and variations in tissue distribution. Research on DPYD genetic polymorphism in Africans is ongoing, but studies focusing on genetic polymorphisms in the capecitabine metabolic pathway remain limited, despite their potential impact on drug response. More investigation is needed into the less-studied toxicities of fluoropyrimidine therapy in African populations, particularly comparing 5-FU and capecitabine, as well as evaluating the healthcare burden related to hospitalisations, blood transfusions and use ofgranulocyte colon-stimulating factor (G-CSF) – all of which are costly and scarce in Africa.
Materials and methods
Study design and setting
We conducted a retrospective cohort study of patients treated for GIT cancers from 2013 to 2022 at the Parirenyatwa Group of Hospitals, Radiotherapy & Oncology Centre in Harare, Zimbabwe.
Inclusion and exclusion criteria
Adult black Africans over 18 years of age with confirmed esophageal, gastric, gall bladder, biliary tree, pancreatic, colon, rectal or anal cancer who received at least one cycle of infusional 5-FU or capecitabine as first line (or, if second line, after more than 6 months from the previous treatment) with adequate laboratory results for study endpoints were included. Exclusion criteria were incomplete records, receiving second-line therapy within 6 months or documented, multiple cancer diagnoses.
Data collection
Demographic and clinicopathologic information, treatment details and laboratory results obtained prior to and during therapy were systematically collected using an enhanced data collection tool. Haematological adverse events were assessed by extracting data on haemoglobin (Hb) levels, as well as leucocyte, neutrophil, lymphocyte and platelet counts, both before and during treatments. For hepatic adverse events, parameters such as alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), albumin and total and direct bilirubin were extracted. Renal function was evaluated through creatinine levels and the electrolytes sodium and potassium. TRAEs were graded for occurrence and severity according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 5, based on the serial laboratory findings.
Study endpoints
The primary objective the incidence of any or severe haematological, renal and hepatic TRAEs in GIT cancer patients treated with infusional 5-FU versus capecitabine. Secondary endpoints were trends in key blood and organ function markers during treatment, rates of treatment interruption, completion/discontinuations and the frequency of hospitalisations, blood transfusions and G-CSF requirements for TRAE management. Severe TRAEs were defined as NCI CTAE ≥3 grade events.
Statistical analysis
Patient characteristics were described using mean and range or median and interquartile range (IQR) for continuous variables, according to normality determined by the Shapiro-Wilk test. Percentages were used to present categorical variables. Incidence of TRAEs and treatment modifications were expressed as proportions with 95% confidence intervals (CIs), both overall and stratified by grade within each fluoropyrimidine group. For comparison between treatment-group we employed chi-square tests for categorical data and Mann-Whitney U-tests for continuous data. Criteria for blood transfusion, G-CSF administration and hospitalisation (defined as additional days other than necessary for standard care) due to TRAEs were based on local guidelines: hemoglobin below 8 g/dL warranted transfusion, absolute neutrophil count below 1.5 ×10³/µL required G-CSF and hospital admission was enumerated as it was. Relative risk (RR), along with its standard error and corresponding 95% CI, was calculated using the 5-FU group as the control cohort and the capecitabine group as the exposed cohort [20]. A forest plot was generated using unadjusted calculated RR data. Statistical significance in the study was defined as a p-value less than 0.05.
Results
Participant characteristics
Out of 804 patients diagnosed with GIT cancer during the review period, 179 fulfilled the study’s inclusion criteria (Figure 1).
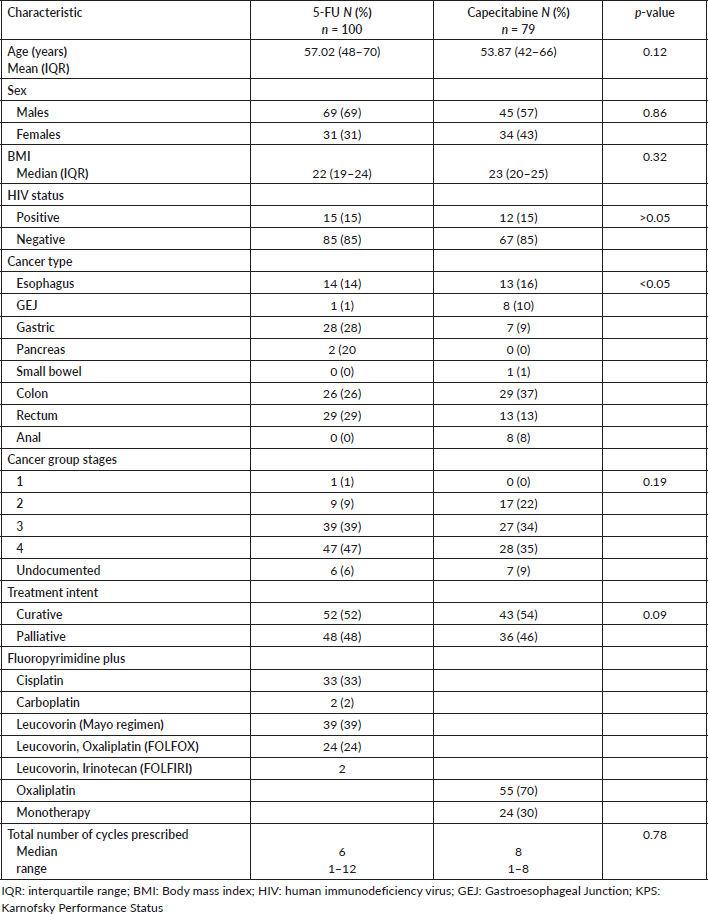
Of the participants, 100 received infusional 5-FU-based chemotherapy and 79 were treated with capecitabine. Both groups had comparable demographic, lifestyle, HIV status and cancer stage at diagnosis, but differed in cancer site/type. Slightly more than half of the participants received treatment with curative intent. Of those administered 5-FU, 59% were also given platinum analogues, 41% leucovorin and 2% irinotecan as combination therapy. In the capecitabine group, 70% received oxaliplatin in combination, while 30% underwent monotherapy (Table 1).
At baseline, the haematological, renal, electrolyte and hepatic function values for study participants were similar except for serum creatinine concentration were the difference between the 5-FU group and the creatinine group were statistically significant, p = 0.04. However, creatinine clearance calculated using the Cockcroft-Gault formula showed insignificant differences, p = 0.309. An analysis of the baseline values is detailed in Supplementary Table S1.
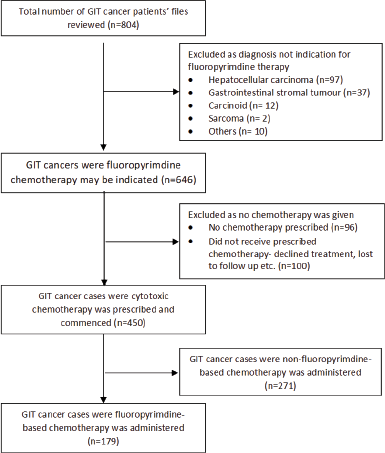
Figure 1. CONSORT flow chart illustrating the selection of eligible patients with gastrointestinal cancer for inclusion in the study.
Treatment-related adverse events
In total, 75/100 participants (75%) in the 5-FU group experienced a TRAE, compared to 51/79 participants (65%) in the capecitabine group (RR 0.86; 95% CI, 0.70–1.05; p = 0.15). When considering the proportion within each group, 33% of participants experienced a severe TRAE during treatment (RR 0.99; 95% CI, 0.70–1.52; p = 0.95) Table 2.
Of those with severe TRAE, 17 of 33 participants (52%) in the 5-FU group and 6 of 26 (23%) in the capecitabine group had grade >3 events. In this small subgroup analysis of participants with severe TRAE, we found a RR 0.45, 95% CI 0.21–0.97, p = 0.004. Figure 2 presents the distribution of TRAE grades across the treatment groups, while Figure 3 provides detailed information regarding haematological, renal and electrolyte and hepatic TRAEs.
In this cohort, rates of haematological TRAEs for 5-FU versus capecitabine were: anaemia (62% versus 51%, p = 0.12), neutropenia (52% versus 33%, p = 0.01), leucopaenia (32% versus 25%, p = 0.33), lymphopenia (23% versus 20%, p = 0.66), febrile neutropenia (16% versus 10%, p = 0.24) and thrombocytopenia (6% versus 4%, p = 0.51). A comparison of haematological TRAEs revealed that only neutropenia differed significantly between the 5-FU and capecitabine groups, with capecitabine being associated with a lower risk (RR 0.63, 95% CI 0.43–0.91). No statistically significant differences were observed for other haematological TRAEs. In a subgroup analysis among participants who experienced haematological TRAE, the rates of severe TRAE for 5-FU versus capecitabine were as follows: anaemia 10% versus 25% (p = 0.04); neutropenia 40% versus 42% (p = 0.87); leucopaenia 31% versus 10% (p = 0.11); lymphopenia 9% versus 6% (p = 0.78); and thrombocytopenia 33% versus 33% (p = 1.0). Severe anaemia occurred more frequently in the capecitabine group compared to the 5-FU group, with a RR of 2.6 (95% CI 1.02–6.55).
Table 1. Demographic and clinical profiles of the study cohort.
Renal and electrolyte TRAEs were similar for 5-FU and capecitabine: increased creatinine (16% versus 15%, p = 0.43), hyponatremia (14% versus 15%, p = 0.84), hyperkalemia (8% versus 5%) and hypernatremia (3% versus 1%, p = 0.45). Hypokalemia was less frequent with capecitabine (17% versus 6%, p = 0.04; RR 0.37, 95% CI 0.14–0.97). Subgroup analysis showed no significant difference in renal or electrolyte TRAEs between groups. Hepatic TRAE rates were similar between 5-FU and capecitabine groups: hypoalbuminemia (20% versus 18%, p = 0.70), increased (14% versus 10%, p = 0.44), AST (8% versus 6%, p = 0.77), hyperbilirubinemia (6% versus 6%, p = 0.94) and ALT (6% versus 5%, p = 0.77). No significant differences in overall any or severe TRAE risk were found between the treatments. Figure 3 below shows the distribution of any and severe TRAE. Supplementary Table S2 details all the TRAE and the corresponding RR.
Table 2. Comparative RR of TRAE incidence and severity between 5-FU and capecitabine groups.
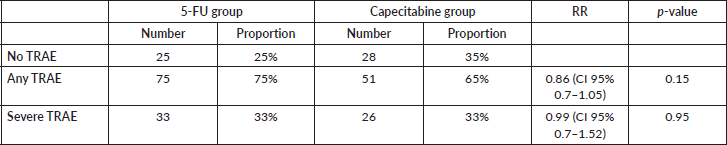
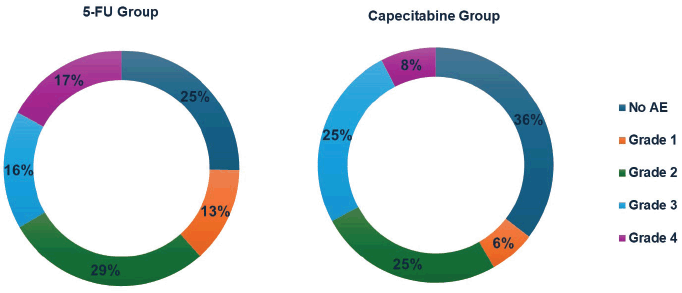
Figure 2. Illustrates the incidence and severity of TRAEs among gastrointestinal cancer patients treated with either 5-FU or capecitabine. The graph highlights that severe TRAEs (grades 3–4) were more frequent in the 5-FU group compared to the capecitabine group. Specifically, 52% of participants in the 5-FU group with severe TRAEs experienced grade >3 events versus 23% in the capecitabine group.
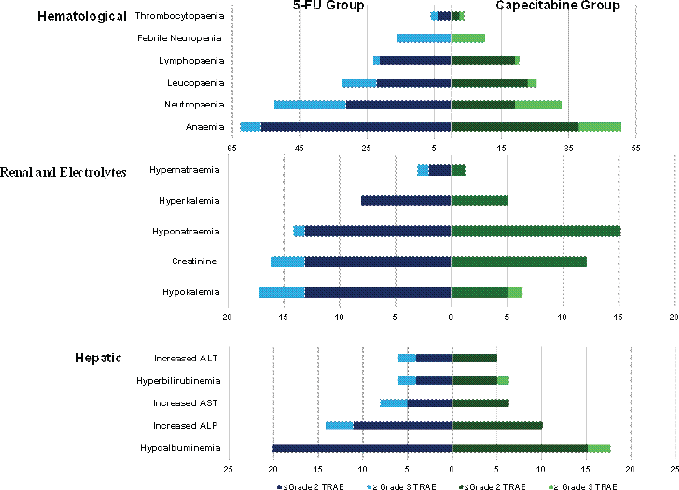
Figure 3. Distribution of participants experiencing any haematological, renal or hepatic TRAEs – including those classified as severe – within the 5-FU and capecitabine cohorts.
Impact of TRAEs
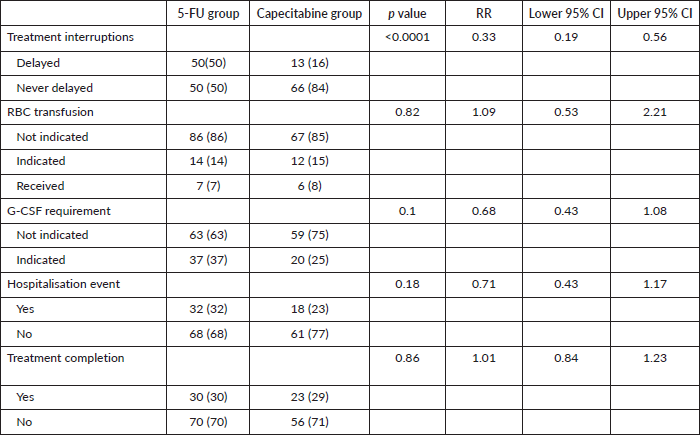
A total of 714 chemotherapy cycles were reviewed across the entire cohort of 179 patients. In comparison between the 5-FU and capecitabine treatment groups, treatment interruptions – defined as at least one chemotherapy cycle delayed by more than 1 day – were observed 4 in 50 patients (50%) versus 13 patients (16%), respectively (RR 0.33, 95% CI 0.19–0.56, p < 0.0001). Other treatment-related challenges, including red blood cell (RBC) transfusions, indications for G-CSF use, hospitalisations for management of TRAEs, and whether the prescribed number of chemotherapy cycles was completed, showed no significant differences between the 5-FU and capecitabine groups, as illustrated in Figure 4. Supplementary Table S3 shows details on the incidence of these events and calculated risk and CIs.
Not all participants who had an indication for RBC transfusion or G-CSF administration received these treatments; documentation shows that only 50% of both groups were given the prescribed transfusion or G-CSF therapy. The overall treatment completion rate was 60% (SD ± 34.63) for the 5-FU group and 54% (SD ± 36.23) for the capecitabine group (p = 0.10) and both cohorts had no recorded dose reduction.
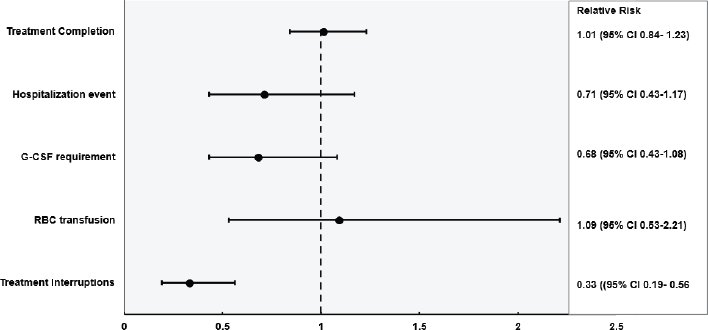
Figure 4. Presents a forest plot of RR and 95% CI comparing the 5-FU and capecitabine groups for treatment completion (p = 0.86), hospitalisations (p = 0.18), G-CSF use for neutropenia (p = 0.10), RBC transfusion (p = 0.82) and treatment interruption (p < 0.0001). Capecitabine-treated participants had fewer chemotherapy cycle delays than those treated with 5-FU.
Discussion
This study demonstrates that there is no significant difference in the overall risk of TRAEs or severe TRAE between gastrointestinal cancer patients treated with 5-FU and those receiving capecitabine. However, among patients who experienced severe TRAE, those on 5-FU exhibited a higher tendency for grade 4 TRAE. Review of system-specific TRAE revealed that capecitabine was associated with a reduced risk of neutropenia and hypokalemia within haematology parameters, while hepatic TRAE risks were comparable between groups. The management of TRAE – including hospitalisation rates, RBC transfusion needs, indications for G-CSF administration and treatment completion – was similar across both cohorts. Notably, patients receiving capecitabine had a lower risk of treatment interruption compared to those on infusional 5-FU.
The incidence of haematological TRAE among black African patients in this study matches findings from both clinical trials and real-world data [21–23]. Among haematological TRAE, anaemia was the most common. The incidence of anaemia in patients with GIT cancers is higher than in those with non-GIT cancers, which may be related to factors such as feeding difficulties, micronutrient deficiencies, malabsorption and advanced stage of cancer at diagnosis [24, 25]. The capecitabine group had significantly lower neutropenia risk, while febrile neutropenia rates were similar to infusional 5-FU in GIT cancer patients of African ancestry. Neutropenia incidence was 7% higher than reported in our earlier study at this site, which included non-GIT cancer patients [26]. Oral capecitabine provides greater convenience and, in this study, is associated with a lower risk of neutropenia. While these factors may be relevant in treatment considerations for African patients, there are limitations to its use, such as restrictions on oral intake or higher purchase costs for certain patient groups. Implementing neutropenia risk assessment and in high-risk individuals prophylactic G-CSF administration may further enhance the haematological safety profile of fluoropyrimidine therapy, as no such adverse events were observed in this cohort [27, 28]. Febrile neutropenia carries a reported mortality rate of up to 30%. Effective risk reduction strategies, prompt detection and timely, appropriate treatment are essential for improving patient outcomes and survival rates [29–31].
Renal and electrolyte TRAE generally exhibited comparable patterns, except for hypokalemia, which presented an increased risk in the 5-FU infusion group. In patients with GIT cancers receiving cytotoxic chemotherapy, hyponatremia and hypokalemia may occur because of various factors such as vomiting, diarrhea or the direct toxic effects of platinum-based agents. Cisplatin, known for its nephrotoxic potential, was administered alongside 5-FU in one-third of participants in the 5-FU cohort within this study. No participants were treated concurrently with both cisplatin and capecitabine. Cisplatin induces systemic hypomagnesemia by reducing magnesium reabsorption in the nephron’s loop of Henle and distal tubule, resulting in associated hypokalemia [32]. In hypokalemic conditions, muscle dysfunction can lead to cardiac arrhythmias and reduced intestinal peristalsis, which may further negatively affect patients’ nutritional and hydration status [33, 34]. The mechanism behind cisplatin-induced hyponatremia remains unclear. Acute kidney injury can occur due to factors such as the syndrome of inappropriate antidiuretic hormone secretion and renal salt wasting syndrome. In this study, the 5-FU group did not, however, experience a higher risk of hyponatremia [35]. In the context of this study, the improved hypokalemia outcome in the capecitabine group may be the result from combination therapy and might not persist if capecitabine is used with cisplatin instead of oxaliplatin.
All the elements analysed for chemotherapy-induced hepatic toxicity in this study showed a similar low risk between patients in infusional 5-FU and capecitabine. Both these observations between these two treatment regimens are consistent with the reported trend in previous clinical trials and real world studies [36, 37].
These findings highlight the advantages of using oral fluoropyrimidine therapy over infusional options for African patients. In resource-limited settings, an effective cancer treatment like capecitabine – which needs less routine hospitalisation and does not increase requirements for more supportive care in the form of RBC transfusion, G-CSF or TRAE hospitalisation – offers higher treatment completion rates and fewer interruptions. This is particularly important as the study highlights limited access to G-CSF and transfusions for cancer patients. While the exact cause remains unclear, high costs and low health insurance coverage may contribute and warrant further investigation [38–41].
Current pharmacogenomic testing guidelines for both fluoropyrimidine drugs recommend pre-emptive DPYD testing [3, 42]. However, there has been limited research into polymorphisms affecting the biotransformation of capecitabine to 5-FU within African populations [43]. The findings of this study indicate that, while further investigation is warranted, capecitabine appears to be well tolerated relative to 5-FU infusion. Additionally, observed differences may be attributable to variations in administration routes, impact of diet and adherence, factors that should ideally be evaluated in a prospective study.
This study had several limitations, including a limited sample size, a heterogeneous patient population, an inability to ascertain the cause for treatment completion failures – all of which affect the generalisability of the results and the inability to assess capecitabine adherence. Additionally, local G-CSF and RBC transfusion guidelines were used, which may differ across various settings, making the conclusion subject to setting-specific conclusions. The study was also conducted at a single institution. The research only reviewed haematological, renal and hepatic TRAE, limiting full assessment of the differences between the two fluoropyrimidines; however, assessing these TRAE retrospectively was viewed to be more objective because it depended purely on objective laboratory measurements allowing for interna validity. Nonetheless, this observational study on gastrointestinal cancer toxicity in an African context, comparing infusional 5-FU with capecitabine and evaluating supportive care requirements, provides further data relevant to the field and it was conducted at single institution but the largest of just two centres in the country and only objective measurements of toxicity were used, i.e., laboratory reports.
Conclusion
Both infusional 5-FU and capecitabine are generally well tolerated among African patients with gastrointestinal cancer. However, capecitabine is associated with a significantly lower risk of neutropenia and hypokalemia but higher RR of severe anaemia with overall fewer incidents of treatment interruption.
Acknowledgments
Many thanks to the SPARK at Stanford team from Stanford University (CA, USA) for their help in making their time and resources available for study. This work would have been impossible without the support, resources and encouragement from Skip Victor, who saw the work as important and supported it. Thank you to all our colleagues at the University of Zimbabwe, Department of Oncology Medical Physics and Imaging Sciences, and the staff and patients at Parirenyatwa Group of Hospitals Radiotherapy & Oncology Centre.
Conflicts of interest
No conflicts of interest.
Funding
Support for this work was provided through grant funding from the Bill and Melinda Gates Foundation (BMGF) (Grant Number ID INV-036801); study protocol design was partially supported by the National Center For Advancing Translational Sciences of the National Institutes of Health under Award Number UL1TR003142 through the Data Studio consultation service at the Stanford University and an award from the American Association for Cancer Research (Grant Number 24-15-75-MAZH).
Ethical approval and consent to participate
All elements of the study were performed in accordance with the Declaration of Helsinki and the relevant regulations and laws governing research in Zimbabwe. No direct patient contact or intervention were made during this study. The study was approved by University of Zimbabwe Faculty of Medicine & Health Sciences and Parirenyatwa Group of Hospitals Joint Research Ethics Committee (JREC/263/2023) and the Medical Research Council of Zimbabwe (MRCZ/A/3159). This was a retrospective study exempt from individual informed consent.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Artificial intelligence use
Microsoft® CoPilot was used to rephrase some sentences written by the authors in the manuscript. The majority of the authors are not native English speakers and this rephrasing assisted with word count reductions and grammatic correction.
References
1. NCCN Guidelines Detail [https://www.nccn.org/guidelines/guidelines-detail] Date Accessed: 15/01/2023
2. Ghafouri-Fard S, Abak A, and Tondro Anamag F, et al (2021) 5-fluorouracil: a narrative review on the role of regulatory mechanisms in driving resistance to this chemotherapeutic agent Front Oncol 11 658636 https://doi.org/10.3389/fonc.2021.658636 PMID: 33954114 PMCID: 8092118
3. Lunenburg CATC, Van Der Wouden CH, and Nijenhuis M, et al (2020) Dutch Pharmacogenetics Working Group (DPWG) guideline for the gene–drug interaction of DPYD and fluoropyrimidines Eur J Hum Genet 28(4) 508–517 https://doi.org/10.1038/s41431-019-0540-0 PMCID: 7080718
4. Henricks LM, Lunenburg CATC, and De Man FM, et al (2018) DPYD genotype-guided dose individualisation of fluoropyrimidine therapy in patients with cancer: a prospective safety analysis Lancet Oncol 19(11) https://doi.org/10.1016/S1470-2045(18)30686-7 PMID: 30348537
5. Joerger M, Huitema ADR, and Boot H, et al (2015) Germline TYMS genotype is highly predictive in patients with metastatic gastrointestinal malignancies receiving capecitabine-based chemotherapy Cancer Chemother Pharmacol 75(4) 763–772 https://doi.org/10.1007/s00280-015-2698-7 PMID: 25677447
6. Edem B, Onwuchekwa C, and Wariri O, et al (2021) Trends in clinical trial registration in sub-Saharan Africa between 2010 and 2020: a cross-sectional review of three clinical trial registries Trials 22(1) 472 https://doi.org/10.1186/s13063-021-05423-1 PMID: 34289892 PMCID: 8293494
7. Cutsem EV, Hoff PM, and Harper P, et al (2004) Oral capecitabine vs intravenous 5-fluorouracil and leucovorin: integrated efficacy data and novel analyses from two large, randomised, phase III trials Br J Cancer 90(6) 1190–1197 https://doi.org/10.1038/sj.bjc.6601676 PMID: 15026800 PMCID: 2409640
8. André T, Boni C, and Mounedji-Boudiaf L, et al (2004) Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer N Engl J Med 350(23) 2343–2351 https://doi.org/10.1056/NEJMoa032709 PMID: 15175436
9. Sapapsap B, Thongnoi P, and Pongpun A, et al (2024) The prevalence of 5-fluorouracil and capecitabine cardiotoxicity: a systematic review and meta-analysis World J Oncol 15(6) 902–921 https://doi.org/10.14740/wjon1920 PMID: 39697430 PMCID: 11650610
10. Rodrigues De Queiroz MV, Tardin Rodrigues De Medeiros AC, and Pires Toledo S, et al (2022) Hand-Foot Syndrome Caused by Capecitabine: Incidence, Risk Factors and the Role of Dermatological Evaluation ecancer 16 [https://ecancer.org/en/journal/article/1390-hand-foot-syndrome-caused-by-capecitabine-incidence-risk-factors-and-the-role-of-dermatological-evaluation]
11. Chintala L, Vaka S, and Baranda J, et al (2011) Capecitabine versus 5-fluorouracil in colorectal cancer: where are we now? Oncol Rev 5(2) 129–140 https://doi.org/10.1007/s12156-011-0074-3
12. Lal H (2014) Hand and foot syndrome secondary to capecitabine Indian J Dermatol Venereol Leprol 80(5) 427 https://doi.org/10.4103/0378-6323.140302 PMID: 25201844
13. Saif MW (2005) Capecitabine versus continuous-infusion 5-fluorouracil for colorectal cancer: a retrospective efficacy and safety comparison Clin Colorectal Cancer 5(2) 89–100 PMID: 16098249
14. Saif MW and Sandoval A (2008) Atypical hand-and-foot syndrome in an African American patient treated with capecitabine with normal DPD activity: is there an ethnic disparity? Cutaneous Ocular Toxicol 27(4) 311–315 https://doi.org/10.1080/15569520802355774
15. Brazelton A (2022) Racial and ethnic differences in capecitabine toxicity in patients with gastrointestinal tract cancers [http://www.annalsgastro.gr/files/journals/1/earlyview/2022/ev-02-2022-03-AG_5798-0688.pdf ] Date Accessed: 22/10/2024
16. Dower J, Salei Y, and Dumas B, et al (2023) Tolerability of capecitabine in different racial/ethnic groups JCO 41(16) e18833 https://doi.org/10.1200/JCO.2023.41.16_suppl.e18833
17. Colombo GL (2012) Economic comparison of capecitabine + oxaliplatin and 5-fluorouracil + oxaliplatin in the adjuvant treatment of colon cancer Cancer Manag Res 4 99–103 https://doi.org/10.2147/CMAR.S29267 PMID: 22500126 PMCID: 3324994
18. Anderson BO (2020) NCCN harmonized guidelines for sub-Saharan Africa: a collaborative methodology for translating resource-adapted guidelines into actionable in-country cancer control plans JCO Global Oncol 6 1419–1421 https://doi.org/10.1200/GO.20.00436
19. The ASCO Post (2023) Announces Additional NCCN Harmonized Guidelines for Sub-Saharan Africa [https://ascopost.com/issues/september-25-2019/announces-additional-nccn-harmonized-guidelines-for-sub-saharan-africa/] Date Accessed: 15/01/2023
20. Schoonjans F. MedCalc. MedCalc’s Relative Risk Calculator [https://www.medcalc.org/calc/relative_risk.php] Date Accessed: 14/07/2025
21. Liu Y, Ren Z, and Yuan L, et al (2016) Paclitaxel plus cisplatin vs. 5-fluorouracil plus cisplatin as first-line treatment for patients with advanced squamous cell esophageal cancer Am J Cancer Res 6(10) 2345–2350 PMID: 27822423 PMCID: 5088297
22. Moehler M, Maderer A, and Thuss-Patience PC, et al (2020) Cisplatin and 5-fluorouracil with or without epidermal growth factor receptor inhibition panitumumab for patients with non-resectable, advanced or metastatic oesophageal squamous cell cancer: a prospective,
open-label, randomised phase III AIO/EORTC trial (POWER) Ann Oncol 31(2) 228–235 https://doi.org/10.1016/j.annonc.2019.10.018 PMID: 31959339
23. Ajani JA, Abramov M, and Bondarenko I, et al (2017) A phase III trial comparing oral S-1/cisplatin and intravenous 5-fluorouracil/cisplatin in patients with untreated diffuse gastric cancer Ann Oncol 28(9) 2142–2148 https://doi.org/10.1093/annonc/mdx275 PMID: 28911091
24. Turkiewicz J, Garcia M, and Li Z, et al (2023) Micronutrient deficiencies in patients with gastrointestinal cancer: to treat or not to treat Nutr Clin Pract 38(4) 749–760 https://doi.org/10.1002/ncp.11025 PMID: 37280762
25. Dotan E, Tew WP, and Mohile SG, et al (2020) Associations between nutritional factors and chemotherapy toxicity in older adults with solid tumors Cancer 126(8) 1708–1716 https://doi.org/10.1002/cncr.32718 PMID: 31977084 PMCID: 7494013
26. Afolabi BL, Mazhindu T, and Zedias C, et al (2023) Pharmacogenetics and adverse events in the use of fluoropyrimidine in a cohort of cancer patients on standard of care treatment in Zimbabwe JPM 13(4) 588 https://doi.org/10.3390/jpm13040588 PMID: 37108974 PMCID: 10141018
27. Crawford J, Dale DC, and Lyman GH (2004) Chemotherapy-induced neutropenia Cancer 100(2) 228–237 https://doi.org/10.1002/cncr.11882 PMID: 14716755
28. Ba Y, Shi Y, and Jiang W, et al (2020) Current management of chemotherapy-induced neutropenia in adults: key points and new challenges: Committee of Neoplastic Supportive-Care (CONS), China Anti-Cancer Association Committee of Clinical Chemotherapy, China Anti-Cancer Association Cancer Biol & Med 17(4) 896–909 https://doi.org/10.20892/j.issn.2095-3941.2020.0069
29. Sereeaphinan C, Kanchanasuwan S, and Julamanee J (2021) Mortality-associated clinical risk factors in patients with febrile neutropenia: a retrospective study IJID Regions 1 5–11 https://doi.org/10.1016/j.ijregi.2021.09.002
30. Al-Tawfiq JA, Hinedi K, and Khairallah H, et al (2019) Epidemiology and source of infection in patients with febrile neutropenia: a ten-year longitudinal study J InfectPublic Health 12(3) 364–366
31. Parodi RL, Lagrutta M, and Tortolo M, et al (2019) A multicenter prospective study of 515 febrile neutropenia episodes in Argentina during a 5-year period PLoS One 14(10) 224299 https://doi.org/10.1371/journal.pone.0224299
32. Kalali D (2024) Potassium imbalances induced by systemic cancer therapy: pathophysiology and potential therapeutic strategies Oncol Clin Pract 20(1) 22–26 https://doi.org/10.5603/ocp.96314
33. Mohammadianpanah M, Omidvari S, and Mosalaei A, et al (2004) Cisplatin-induced hypokalemic paralysis Clin Therapeutics 26(8) 1320–1323 https://doi.org/10.1016/S0149-2918(04)80177-2
34. Oronsky B, Caroen S, and Oronsky A, et al (2017) Electrolyte disorders with platinum-based chemotherapy: mechanisms, manifestations and management Cancer Chemother Pharmacol 80(5) 895–907 https://doi.org/10.1007/s00280-017-3392-8 PMID: 28730291 PMCID: 5676816
35. Russo C (2021) Cisplatin-induced acute kidney injury and renal salt wasting syndrome Federal Practitioner [Internet] 38(12) 799 [https://www.mdedge.com/fedprac/article/249799/nephrology] https://doi.org/10.12788/fp.0198
36. Grothey A, Sobrero AF, and Shields AF, et al (2018) Duration of adjuvant chemotherapy for stage III colon cancer N Engl J Med 378(13) 1177–1188 https://doi.org/10.1056/NEJMoa1713709 PMID: 29590544 PMCID: 6426127
37. Degirmencioglu S, Tanrıverdi O, and Demiray AG, et al (2019) Retrospective comparison of efficacy and safety of CAPOX and FOLFOX regimens as adjuvant treatment in patients with stage III colon cancer J Int Med Res 47(6) 2507–2515 https://doi.org/10.1177/0300060519848258 PMID: 31099282 PMCID: 6567718
38. Zafar SY, Peppercorn JM, and Schrag D, et al (2013) The financial toxicity of cancer treatment: a pilot study assessing out-of-pocket expenses and the insured cancer patient’s experience Oncologist 18(4) 381–390 https://doi.org/10.1634/theoncologist.2012-0279 PMID: 23442307 PMCID: 3639525
39. Elmore SNC, Polo A, and Bourque JM, et al (2021) Radiotherapy resources in Africa: an International Atomic Energy Agency update and analysis of projected needs Lancet Oncol 22(9) e391–e399 https://doi.org/10.1016/S1470-2045(21)00351-X PMID: 34478675 PMCID: 8675892
40. Mafirakureva N, Nyoni H, and Nkomo SZ, et al (2016) The costs of producing a unit of blood in Zimbabwe Transfusion 56(3) 628–636 https://doi.org/10.1111/trf.13405
41. Rutendo Matowa P and Ndwabe H (2016) Allogeneic blood transfusion: the cost of blood transfusion at a large urban hospital in Zimbabwe OJBD 06(03) 33–43 https://doi.org/10.4236/ojbd.2016.63006
42. Muldoon M, Beck M, and Sebree N, et al (2024) Real‐world implementation of DPYD and UGT1A1 pharmacogenetic testing in a community‐based cancer center Clin Translational Sci 17(2) e13704 https://doi.org/10.1111/cts.13704
43. Mercier C, Dupuis C, and Blesius A, et al (2009) Early severe toxicities after capecitabine intake: possible implication of a cytidine deaminase extensive metabolizer profile Cancer Chemother Pharmacol 63(6) 1177–1180 https://doi.org/10.1007/s00280-008-0889-1
Supplementary Tables
Table S1. Initial haematological, renal, electrolyte and hepatic function measurements for participants receiving fluoropyrimidine therapy for gastrointestinal cancer.
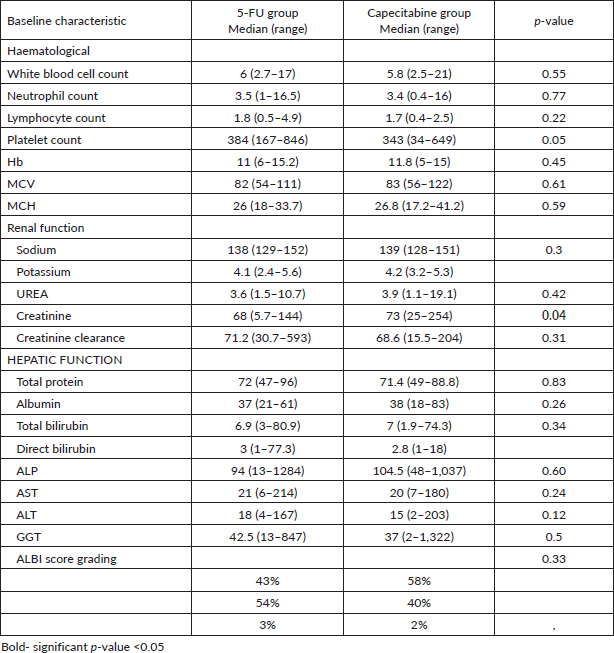
Table S2. Haematological, renal and hepatic TRAEs; incidence, RR and significance level for any TRAE and severe TRAE.
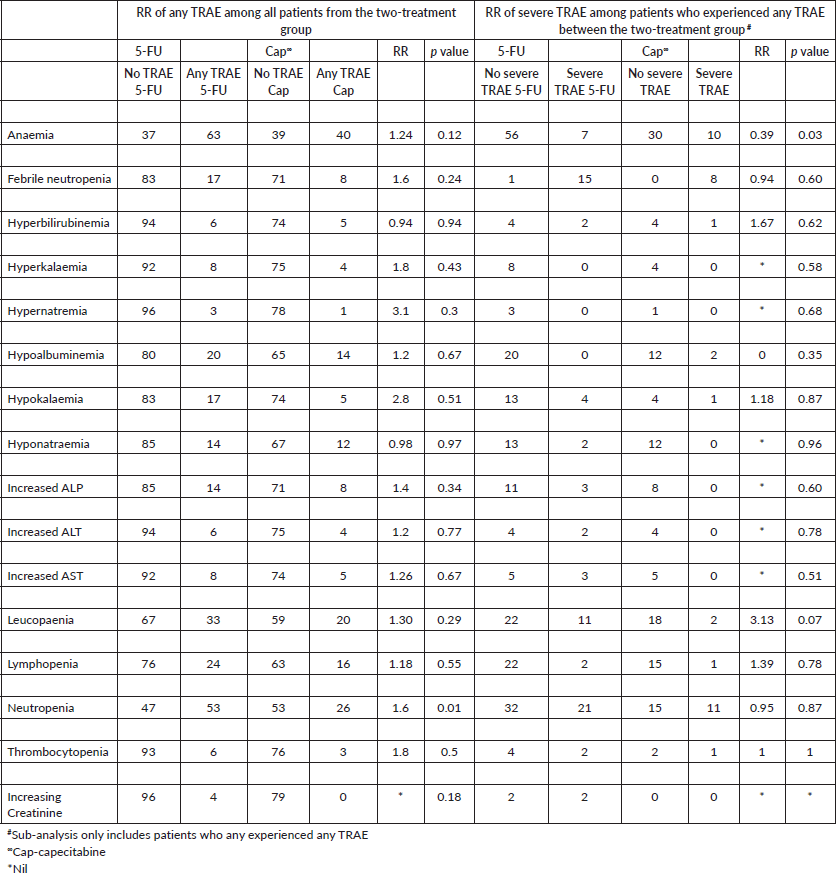
Table S3. RR and 95% CI for treatment completion, hospitalisation, G-CSF use for neutropenia, RBC transfusion and treatment interruption in 5-FU versus capecitabine groups.