Two cases of gastric mucosa-associated lymphoid tissue (MALT) lymphoma masquerading as follicular gastritis
Masaya Iwamuro1, Takehiro Tanaka2, Kenji Nishida3, Hiromitsu Kanzaki1, Seiji Kawano1, Yoshiro Kawahara4, Tadashi Yoshino3 and Hiroyuki Okada1
1Department of Gastroenterology and Hepatology, Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences, Okayama 700-8558, Japan
2Department of Pathology, Okayama University Hospital, Okayama 700-8558, Japan
3Department of Pathology, Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences, Okayama 700-8558, Japan
4Department of Endoscopy, Okayama University Hospital, Okayama 700-8558, Japan
Abstract
In this report, we describe two cases of extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma) of the stomach, which presented with multiple small, whitish nodules in the gastric body. The endoscopic appearance was similar to that of lymphoid follicular hyperplasia found in follicular gastritis or nodular gastritis. Both patients were positive for Helicobacter pylori, and the eradication treatment resulted in complete remission of the lymphoma. However, recurrence was noted in one patient. These cases indicate that, although infrequent, gastric MALT lymphoma can show a nodular appearance resembling that of follicular gastritis.
Keywords: mucosa-associated lymphoid tissue lymphoma, gastrointestinal endoscope, gastric neoplasms, follicular gastritis, nodular gastritis
Correspondence to: Masaya Iwamuro
Email: iwamuromasaya@yahoo.co.jp
Published: 03/06/2019
Received: 27/02/2019
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Helicobacter pylori (H. pylori) infection in the stomach leads to inflammation of gastric mucosa, which in turn leads to various types of alterations of the microscopic morphology. The process includes regenerative changes, intestinal metaplasia and foveolar hyperplasia [1]. In some patients, H. pylori infection causes the formation of prominent lymphoid follicles and mononuclear cell infiltration. This form of gastric inflammation has been endoscopically described as a nodular or diffuse miliary pattern of small elevations, identified typically in the antrum and occasionally extending to the whole gastric body [2–9]. This unique macroscopic feature was first reported as ‘gooseflesh skin-like’ gastric mucosa [2, 10] and is being increasingly called follicular gastritis or nodular gastritis in endoscopic practice. Although the term is not formally cited in the Sydney System for the classification of gastritis [2, 11], ‘nodularity’ is included as one of the features associated with active H. pylori infection in the Kyoto classification [2–14].
In this paper, we report two cases of extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma) of the stomach, which presented multiple small, whitish nodules resembling follicular gastritis. It was noteworthy that the nodularity was predominantly observed in the gastric body, rather than in the antrum. Biopsy examination revealed monomorphic proliferation of B-cells that were positive for CD20 and negative for CD3, CD10 and Cyclin D1. Lymphoepithelial lesions were also present, leading to the definitive diagnosis of gastric MALT lymphoma.
Case report
Case 1
A 41-year-old Japanese man underwent esophagogastroduodenoscopy screening. He had diabetes mellitus, hypertension and dyslipidaemia, and was receiving metformin, amlodipine and pravastatin. He had no history of gastrointestinal disease. The physical examination revealed no abnormalities and no evidence of peripheral lymphadenopathy. Laboratory findings demonstrated elevated levels of choline esterase (475 U/L), aspartate aminotransferase (54 U/L) and alanine aminotransferase (89 U/L), probably related to non-alcoholic fatty liver. The white blood cells count (9,090/μL) and eosinophil fraction (11.3%) were increased, but no atypical lymphocytes were identified in the peripheral blood. The patient’s serum was positive for anti-H. pylori immunoglobulin G antibodies, and his urea breath test was also positive.
Esophagogastroduodenoscopy showed a diffuse miliary pattern with slightly whitish, small elevations in the gastric body (Figure 1a and b). The multiple granular elevations were emphasised on narrow-band imaging (Figure 1c) and after indigo carmine spraying (Figure 1d). A mild atrophic change was noted in the gastric antrum compared with the gastric body, but the granular appearance was not evident (Figure 1e). A biopsy specimen from part of the small elevations of the gastric body revealed follicle formation (Figure 2a). Infiltrating lymphocytes within the follicle were monomorphic (Figure 2b) and positive for CD20 (Figure 2c), but they were negative for CD3 (Figure 2d), CD10 (Figure 2e) and Cyclin D1 (Figure 2f). Staining with haematoxylin and eosin (Figure 2g) and anti-human cytokeratin clone CAM5.2 showed lymphoepithelial lesions (Figure 2h). Fluorescence in situ hybridisation (FISH) analysis for t(11;18)(q21;q21) translocation revealed no fusion genes of baculoviral IAP repeat-containing protein 3 (BIRC3)-MALT1. On 18F-fluorodeoxyglucose positron emission tomography, no tracer uptake was noted. Colonoscopy and bone marrow biopsy revealed no lymphoma lesions as well. Consequently, the gastric lesion was diagnosed as stage I MALT lymphoma of the stomach.
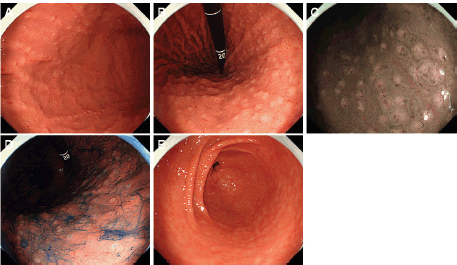
Figure 1. Esophagogastroduodenoscopy images of Case 1. (a) and (b): A diffuse miliary pattern of slightly whitish, small elevations was identified in the gastric body. (c): The elevations were emphasised on narrow-band imaging. (d): After indigo carmine spraying. (e): Granular appearance was not evident in the antrum.
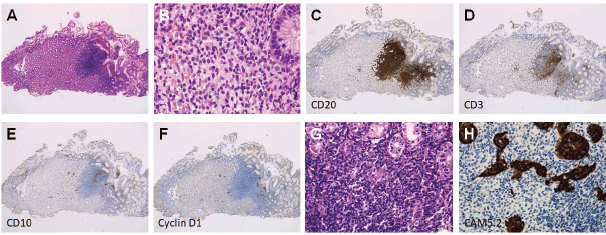
Figure 2. Pathological images of Case 1. (a): Follicle formation in a biopsy specimen. (b): Monomorphic lymphocytes. (c): Cells positive for CD20. (d): Negative for CD3. (e): CD10. (f): Cyclin D1. (g) and (h): Lymphoepithelial lesions.
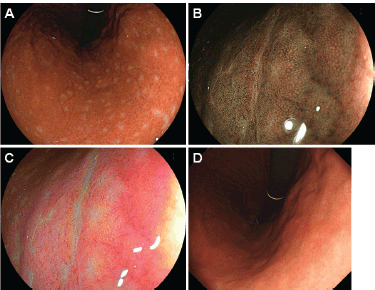
Figure 3. Esophagogastroduodenoscopy images of Case 1. (a)–(c): 3 months. (d): 45 months after eradication of H. pylori. Small elevations disappeared and whitish spots could be partly observed in the gastric body. (b): Magnifying observation with narrow-band imaging and (c) linked colour imaging also showed that the elevations regressed. (d): Esophagogastroduodenoscopy performed 45 months after eradication of H. pylori showed vague whitish spots in the gastric body.
Because the patient tested positive for H. pylori, eradication treatment was attempted. Esophagogastroduodenoscopy performed 3 months after the completion of eradication treatment showed disappearance of the small elevations, while whitish spots were partly observed in the gastric body (Figure 3a–c). Elimination of neoplastic cells was pathologically confirmed on the biopsy specimens. No recurrence was documented for 45 months since complete remission was achieved. Esophagogastroduodenoscopy performed 45 months after achieving complete remission showed that the whitish spots had almost disappeared (Figure 3d).
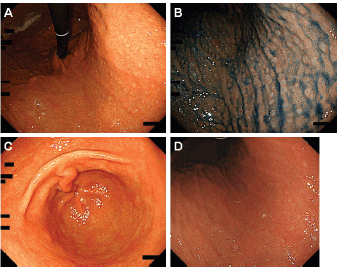
Figure 4. Esophagogastroduodenoscopy images of Case 2. [(a) and (b): After indigo carmine spraying] Slightly whitish, small, multiple elevations can be seen in the lesser curvature of the gastric body. (c) and (d): The small elevations regressed 5 months after H. pylori eradication.
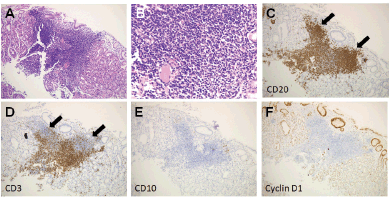
Figure 5. Pathological images in Case 2. (a) and (b): Haematoxylin and eosin staining of a biopsy specimen. (c): Cells positive for CD20. (d): Negative for CD3. (e): CD10. (f): Cyclin D1.
Case 2
A 54-year-old Japanese woman underwent esophagogastroduodenoscopy for screening purposes. She had been receiving amlodipine for the treatment of hypertension. The physical examination revealed no abnormalities. The laboratory findings demonstrated elevated levels of glutamyl transpeptidase (111 U/L). Other blood chemistry and complete blood count were within the normal ranges. The test for serum anti-H. pylori immunoglobulin G antibodies showed positive results.
Esophagogastroduodenoscopy showed multiple slightly whitish, small elevations in the lesser curvature of the gastric body (Figure 4a and b). The granular appearance was not evident in the gastric antrum (Figure 4c). Biopsy from the elevation revealed diffuse infiltration of monomorphic lymphocytes, which were predominantly positive for CD20 on immunochemical analysis (Figure 5). FISH analysis for t(11;18)(q21;q21) translocation revealed no fusion genes of BIRC3-MALT1. Gastric MALT lymphoma was highly suspected, but definitive diagnosis could not be established, because no prominent lymphoepithelial lesion was identified. The patient underwent eradication treatment for H. pylori. Esophagogastroduodenoscopy performed 5 months after H. pylori eradication revealed regression of small elevations (Figure 4c). Monomorphic lymphocytes and H. pylori disappeared from the biopsy specimens. However, esophagogastroduodenoscopy performed 24 months after the initial examination showed re-emergence of miliary appearance in the gastric body. Infiltration of monomorphic B-cells was noted in the biopsy specimens, and the lesion was pathologically diagnosed as probable MALT lymphoma of the stomach. Five months later, multiple granular elevations remained on the lesser curvature of the gastric body (Figure 6). Pathological analysis revealed infiltration of small- to medium-sized monomorphic B-cells (Figure 7a–c) showing prominent lymphoepithelial lesions (Figure 7d–f). Thus, the definitive diagnosis of gastric MALT lymphoma was established. The patient underwent computed tomography, colonoscopy and bone marrow biopsy, and no lymphoma lesions were noted. Radiotherapy was planned for the treatment of gastric MALT lymphoma.
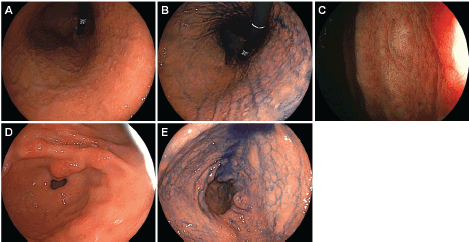
Figure 6. Esophagogastroduodenoscopy images of Case 2 performed 29 months after the initial examination. [(a) and (b): After indigo carmine spraying] Miliary appearance re-emerged in the gastric body. (c): Magnifying observation with narrow-band imaging showed small elevations. [(d) and (e): After indigo carmine spraying] Granular appearance was not evident in the antrum.
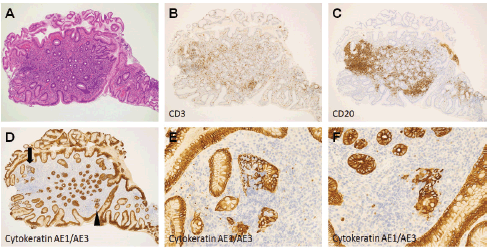
Figure 7. Pathological images of Case 2. (a) Haematoxylin and eosin staining of a biopsy specimen. (b): Cells negative for CD3. (c) Positive for CD20. [(d) arrow and arrowhead]: Lymphoepithelial lesions were noted in cytokeratin AE1/AE3 staining. (e): Magnified views of arrow. (f): Arrowhead.
Discussion
In this report, we describe two cases of gastric MALT lymphoma presenting with a diffuse miliary appearance with slightly whitish and small elevations. The endoscopic features were similar to those of H. pylori-associated gastritis, known as follicular gastritis or nodular gastritis. It was noteworthy that the nodularity was predominant in the gastric body in the present two cases, whereas it is generally observed in the gastric antrum in follicular gastritis. As shown in Figures 2 and 5, the nodularity probably resulted from neoplastic follicle formation by MALT lymphoma cells.
Although gastric MALT lymphomas present a variety of macroscopic features, MALT lymphoma resembling follicular gastritis is infrequent. According to the previously reported classification of this disease [15], we retrospectively reviewed the data of 82 patients with gastric MALT lymphoma diagnosed at our institution. The endoscopic features of these patients included whitish lesions (N = 38, 46.3%), reddish or erosive lesions (N = 15, 18.3%), ulcers (N = 8, 9.8%), elevated lesions (N = 6, 7.3%), cobblestone appearance (N = 6, 7.3%), mucosal swelling or oedematous lesions (N = 4, 4.9%), depressed lesions resembling early gastric cancer (N = 3, 3.7%) and other lesions (N = 2, 2.4%). The last group comprised the two cases presented in this case report. Thus, at our institution, MALT lymphoma resembling follicular gastritis represented 2.4% of all gastric MALT lymphomas. Zullo et al [16, 17] proposed an updated endoscopic classification of gastric MALT lymphoma, as follows: ulcerative (single/multiple ulcerations or erosions), exophytic (irregular or polypoid mass), hypertrophic (large gastric fold or nodular mucosa), petechial haemorrhage (multiple mucosal petechial haemorrhages), normal (absence of macroscopic lesions) and mixed (a combination of more patterns). When whitish MALT lymphoma lesions without haemorrhages were subclassified as ‘normal,’ the endoscopic features of the 82 patients were normal (N = 32, 39.0%), ulcerative (N = 19, 23.2%), petechial haemorrhage (N = 13, 15.9%), hypertrophic (N = 11, 13.4%) and exophytic (N = 7, 8.4%). According to this classification, the two cases presented in this case report are classified as ‘hypertrophic.’
To our knowledge, only two cases of gastric MALT lymphoma showing similar endoscopic features to those of follicular gastritis have been reported (Table 1). Cheng et al [18] described a 49-year-old woman with gastric MALT lymphoma, presenting as fine granular mucosal change in the lower gastric body and antrum. The authors noted that the eradication of H. pylori resulted in complete remission. Lee et al [19] reported another case of a 12-year-old girl with gastric MALT lymphoma masquerading as follicular gastritis on endoscopy. Pathological features of MALT lymphoma improved after the eradication of H. pylori in this patient as well. Previously reported cases and the results of our review indicate that, although infrequent, gastric MALT lymphoma emerges with similar appearance to that of follicular gastritis. Owing to the rarity of this type of MALT lymphoma, the biological characteristics of gastric MALT lymphoma masquerading as follicular gastritis have not been elucidated. However, as the present two patients and previously reported patients were positive for H. pylori infection, we speculate the possible involvement of H. pylori in lymphoid follicle formation in the gastric body, which leads to a nodular or miliary pattern of small elevations.
Intact gastric mucosa is believed to inherently lack lymphocytic aggregates. Helicobacter pylori infection in the stomach induces chronic inflammation, leading to the development of lymphoid follicles [5, 20]. Emerged lymphoid follicles are called MALT. As described above, in some patients, lymphocytic aggregates and follicles in the gastric mucosa cause a granular appearance predominantly in the antrum, which is termed as follicular gastritis or nodular gastritis (Figure 8). The reported prevalence of follicular gastritis was 0.7%–2.9% [4, 6, 9, 21]. In contrast to the predominant involvement of the antrum in follicular gastritis, the present two cases with MALT lymphoma had prominent nodularity in the gastric body and nodularity was less obvious in the antrum. Therefore, we consider that endoscopists should take biopsy samples to differentiate the two entities when they observe a nodular or miliary pattern of small elevations in the gastric body.
The difficulty in differentiating gastric MALT lymphoma from follicular gastritis based on pathological features has been previously reported [18, 22–24]. Hummel et al [24] investigated B-cell clonality in cases with chronic gastritis (Wotherspoon scores 1 and 2, N = 53), overt gastric MALT lymphoma (Wotherspoon score 5, N = 26) and ambiguous histology (Wotherspoon scores 3 and 4, N = 18). The authors noted that B-cell clonality was found in 1/53 cases of chronic gastritis (1.9%), 24/26 cases of overt lymphoma (92.3%) and 4/18 cases with ambiguous histology (22.2%). Similarities and ambiguities in pathologic and molecular biologic features between the two disease entities indicate a possible transition from chronic gastritis to lymphoma during the pathogenesis of gastric MALT lymphoma [6, 9, 24–27]. The present two cases show morphological similarities of MALT lymphoma with follicular gastritis on endoscopy, which may be another evidence for this issue.
Table 1. Reported cases with gastric MALT lymphoma resembling follicular gastritis.

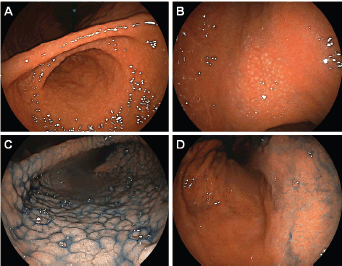
Figure 8. Typical endoscopic images of follicular gastritis. In a 21-year-old Japanese woman with H. pylori infection, (a) miliary appearance was noted in the gastric antrum (b) in the body. (c): After indigo carmine spraying, nodularity was predominantly observed in the antrum. (d): Rather than in the body.
Helicobacter pylori eradication is the appropriate first-line treatment strategy for follicular gastritis. Morphological regression of nodular appearance on endoscopy and decrease in number and size of the lymphoid follicles on pathology after a successful H. pylori eradication has been previously reported [2, 6, 9, 20, 21]. Even monoclonal B-cell populations, if they are detected in follicular gastritis, disappear after H. pylori eradication [9]. In the present two cases, nodularity of the gastric mucosa disappeared after H. pylori eradication and the gastric lesions turned to be whitish spots in both cases. However, although complete remission was obtained for 34 months in Case 1, re-emergence of miliary appearance was identified in Case 2 even after H. pylori eradication.
Different outcomes have been reported for several subsets of gastric MALT lymphoma. For example, it is well known that patients with t(11;18) translocation are resistant to H. pylori eradication [28, 29]. In our previous work, we have revealed that progression or relapse tended to be more frequent in patients with extra copies of MALT1 [30]. Conversely, MALT lymphoma with increased plasma cell differentiation shows favourable treatment response [31, 32]. Based on the outcome observed in Case 2 of the present report, careful follow-up may be necessary in patients with gastric MALT lymphoma resembling follicular gastritis to detect relapse. Further investigation is required to reveal treatment response and long-term outcomes of these patients.
Conclusion
We presented two cases of gastric MALT lymphoma showing a nodular appearance. Although presentation with such morphology is likely to be infrequent, the present two cases indicate that gastric MALT lymphoma may present a nodular appearance resembling that of follicular gastritis. Biopsy examination should be performed when a nodular or miliary pattern of small elevations is observed, particularly in the gastric body rather than in the antrum.
Conflict of interest
The authors declare that they have no conflict of interests regarding the publication of the paper.
Funding declaration
The authors received no specific funding for this work.
References
1. Whiting JL, Sigurdsson A, and Rowlands DC, et al (2002) The long term results of endoscopic surveillance of premalignant gastric lesions Gut 50(3) 378–381 https://doi.org/10.1136/gut.50.3.378 PMID: 11839718 PMCID: 1773155
2. Romero-Flores JL, Fernandez-Rivero JA, and Marroquín-Fabian E, et al (2016) Diagnostic accuracy of nodular gastritis for H. pylori infection Ther Clin Risk Manag 13 9–14 https://doi.org/10.2147/TCRM.S121735 PMID: 28031716 PMCID: 5179200
3. De Giacomo C, Fiocca R, and Villani L, et al (1990) Helicobacter pylori infection and chronic gastritis: clinical, serological, and histologic correlations in children treated with amoxicillin and colloidal bismuth subcitrate J Pediatr Gastroenterol Nutr 11(3) 310–316 https://doi.org/10.1097/00005176-199010000-00005 PMID: 2246711
4. Shimatani T, Inoue M, and Iwamoto K, et al (2005) Gastric acidity in patients with follicular gastritis is significantly reduced, but can be normalized after eradication for Helicobacter pylori Helicobacter 10(3) 256–265 https://doi.org/10.1111/j.1523-5378.2005.00318.x PMID: 15904484
5. Mejia CR, Vera CA, and Huiza-Espinoza L (2016) Association between follicular gastritis and Helicobacter pylori in children seen at a public hospital in Peru Rev Gastroenterol Mex 81(2) 80–85 PMID: 26993160
6. Choi HJ, Lee SY, and Lee JH, et al (2010) Two atypical cases of nodular gastritis: a poorly differentiated gastric adenocarcinoma and a pseudo-low grade gastric MALT lymphomama Gastroenterology Res 3(1) 41–45 PMID: 27956984 PMCID: 5139839
7. Sokmensuer C, Onal IK, and Yeniova O, et al (2009) What are the clinical implications of nodular gastritis? Clues from histopathology Dig Dis Sci 54(10) 2150–2154 https://doi.org/10.1007/s10620-009-0842-0 PMID: 19462235
8. Niknam R, Manafi A, and Maghbool M, et al (2015) Is endoscopic nodular gastritis associated with premalignant lesions? Neth J Med 73(5) 236–241 PMID: 26087803
9. Miyamoto M, Haruma K, and Hiyama T, et al (2002) High incidence of B-cell monoclonality in follicular gastritis: a possible association between follicular gastritis and MALT lymphoma Virchows Arch 440(4) 376–380 https://doi.org/10.1007/s00428-001-0575-8 PMID: 11956817
10. Miyagawa H, Takechi K, and Kato S, et al (1985) Clinical and immunohistological study on goose flesh-like mucosa of the stomach Gastroenterological Endoscopy 27(7) 1275–1280
11. Sipponen P and Price AB (2011) The Sydney system for classification of gastritis 20 years ago J Gastroenterol Hepatol 26(1) 31–34 https://doi.org/10.1111/j.1440-1746.2010.06536.x PMID: 21199511
12. Kamada T, Haruma K, and Inoue K, et al (2015) Helicobacter pylori infection and endoscopic gastritis-Kyoto classification of gastritis Nihon Shokakibyo Gakkai Zasshi 112(6) 982–993 PMID: 26050720
13. Shichijo S, Hirata Y, and Niikura R, et al (2017) Association between gastric cancer and the Kyoto classification of gastritis J Gastroenterol Hepatol 32(9) 1581–1586 https://doi.org/10.1111/jgh.13764 PMID: 28217843
14. Sugimoto M, Ban H, and Ichikawa H, et al (2017) Efficacy of the Kyoto classification of gastritis in identifying patients at high risk for gastric cancer Intern Med 56(6) 579–586 https://doi.org/10.2169/internalmedicine.56.7775 PMID: 28321054 PMCID: 5410464
15. Oda H (1999) Endoscopic therapy against low-grade gastric MALT lymphoma Stomach and Intestine (Tokyo) 34(11) 1381–1388
16. Zullo A, Hassan C, and Cristofari F, et al (2010) Gastric low-grade mucosal-associated lymphoid tissue-lymphoma: Helicobacter pylori and beyond World J Gastrointest Oncol 2(4) 181–186 https://doi.org/10.4251/wjgo.v2.i4.181 PMID: 21160595 PMCID: 2998825
17. Zullo A, Hassan C, and Ridola L, et al (2014) Gastric MALT lymphoma: old and new insights Ann Gastroenterol 27(1) 27–33 PMID: 24714739 PMCID: 3959547
18. Cheng TI, Tsou MH, and Tsai MP, et al (2004) Early gastric MALT lymphoma J Chin Med Assoc 67(3) 145–148 PMID: 15181968
19. Lee KS, Yang HR, and Ko JS, et al (2008) A case of gastric MALT lymphoma presenting as nodular gastritis in a child Korean J Pediatr Gastroenterol Nutr 11(2) 187–192 https://doi.org/10.5223/kjpgn.2008.11.2.187
20. Yang HJ, Lim SH, and Lee C, et al (2016) Management of suspicious mucosa-associated lymphoid tissue lymphoma in gastric biopsy specimens obtained during screening endoscopy J Korean Med Sci 31(7) 1075–1081
21. Chen MJ, Wang TE, and Chang WH, et al (2007) Nodular gastritis: an endoscopic indicator of Helicobacter pylori infection Dig Dis Sci 52(10) 2662–2666 https://doi.org/10.1007/s10620-006-9281-3 PMID: 17417730
22. Doglioni C, Ponzoni M, and Ferreri AJ, et al (2011) Gastric lymphoma: the histology report Dig Liver Dis 43(4) S310–S318 https://doi.org/10.1016/S1590-8658(11)60587-2 PMID: 21459337
23. Wotherspoon AC, Ortiz-Hidalgo C, and Falzon MR, et al (1991) Helicobacter pylori-associated gastritis and primary B-cell gastric lymphoma Lancet 338(8776) 1175–1176 https://doi.org/10.1016/0140-6736(91)92035-Z PMID: 1682595
24. Hummel M, Oeschger S, and Barth TF, et al (2006) Wotherspoon criteria combined with B cell clonality analysis by advanced polymerase chain reaction technology discriminates covert gastric marginal zone lymphoma from chronic gastritis Gut 55(6) 782–787 https://doi.org/10.1136/gut.2005.080523 PMID: 16423889 PMCID: 1856242
25. Ma ZQ, Tanizawa T, and Nihei Z, et al (2000) Follicular gastritis associated with Helicobacter pylori J Med Dent Sci 47(1) 39–47
26. Torlakovic E, Cherwitz DL, and Jessurun J, et al (1997) B-cell gene rearrangement in benign and malignant lymphoid proliferations of mucosa-associated lymphoid tissue and lymph nodes Hum Pathol 28(2) 166–173
27. Saxena A, Moshynska O, and Kanthan R, et al (2000) Distinct B-cell clonal bands in Helicobacter pylori gastritis with lymphoid hyperplasia J Pathol 190(1) 47–54 PMID: 10640991
28. Remstein ED, Kurtin PJ, and James CD, et al (2002) Mucosa-associated lymphoid tissue lymphomas with t(11;18)(q21;q21) and mucosa-associated lymphoid tissue lymphomas with aneuploidy develop along different pathogenetic pathways Am J Pathol 161 63–71 https://doi.org/10.1016/S0002-9440(10)64157-0 PMID: 12107090 PMCID: 1850705
29. Auer IA, Gascoyne RD, and Conners JM, et al (1997) t(11;18)(q21;q21) is the most common translocation in MALT lymphomas Ann Oncol 8 979–985 https://doi.org/10.1023/A:1008202303666 PMID: 9402171
30. Iwamuro M, Takenaka R, and Nakagawa M, et al (2017) Management of gastric mucosa-associated lymphoid tissue lymphoma in patients with extra copies of the MALT1 gene World J Gastroenterol 23(33) 6155–6163
31. Park S, Ahn S, and Hong M, et al (2017) Increased plasmacytic differentiation in gastric mucosa-associated lymphoid tissue lymphomas: Helicobacter pylori eradication response and IgG4 plasma cell association Hum Pathol 59 113–119 https://doi.org/10.1016/j.humpath.2016.09.013
32. Iwamuro M, Tanaka T, and Nishida K, et al (2018) Gastric MALT lymphoma with increased plasma cell differentiation showing unique endoscopic features Case Rep Gastrointest Med 2018 8054284 PMID: 29666720 PMCID: 5832122






