Recurrence of extramedullary plasmacytoma of the breast
Khaled Majadob, Wesam Al-Sakkaf, Farouk Rezk, Kris Zegocki and Faris Al-Refaie
Haematology Department, The Princess Alexandra Hospital, Harlow, Essex, UK
Correspondence to: Khaled Majadob. Email: knm405@yahoo.com
Abstract
We present a rare case of a multiple recurrent extramedullary plasmacytoma of the breast and spine that showed aggressive behaviour without bone marrow involvement. A 29-year-old woman initially presented with extramedullary plasmacytoma of the left breast that was treated with radiotherapy. Shortly after, a recurrence appeared in the right breast, spine, and in both breasts, despite treatment with local and systemic therapy.
Keywords: extramedullary plasmacytoma, breast plasmacytoma, aggressive plasmacytoma
Introduction
Extramedullary plasmacytoma (EMP) of the breast is a rare plasma cell neoplasm of the soft tissue that may occur with or without bone marrow involvement or other systemic features of multiple myeloma. Soft tissue extramedullary plasmacytoma (SEP) that occurs as a solitary tumour is extremely rare. SEP commonly involves the soft tissue of the upper respiratory tract, abdomen, mandible, maxilla, and gingiva [1].
In this case report, we present a patient with aggressive recurrent SEP of the breast with a six-month interval between each recurrence. We also aim to describe the response of the patient to the treatment with chemotherapy and radiotherapy at each stage of the disease.
Case presentation
A 29-year-old woman presented with swelling in the left breast near the nipple as early as October 2009, but this became prominent within one month, and she eventually went on to have fine needle aspiration cytology, which showed plasmacytoid cells in the background, followed by a core biopsy that showed infiltration by immature plasma cells, which were positive for CD138, MUM1, and CD56 and weakly positive for cyclin D1 and EMA. There was lambda light chain restriction. The cells were negative for CD20, CD19, PAX-5, and EBER. They were also negative for MNF 116 and CK7. This supported a diagnosis of plasmacytoma. There was no evidence of any secretory paraprotein in the patient’s blood or urine, and her serum free light chains were normal. An MRI of her spine was clear, as was her bone marrow, which only showed 5% plasma cells. Subsequently, she underwent radical radiotherapy (40.05 Gy in 15 fractions over three weeks).
Six months later, she re-presented with a lump in her right breast that was 2 cm in size felt clinically, which pointed to a recurrence in the contralateral breast (Figure 1). An ultrasound scan and core biopsy confirmed recurrent disease. Again, investigations, including a PET scan, failed to show myeloma or other lesions. The lump was PET positive (Figure 2). She commenced treatment with CTD (cyclophosphamide, thalidomide, and dexamethasone) in June 2010 and completed five cycles.
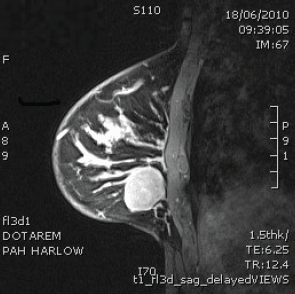
Figure 1: PET scan which shows metabolic activity only in the right breast.
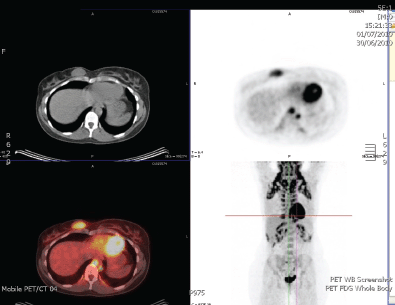
Figure 2: MRI scan of right breast shows well circumscribed solid mass.
Six months later, she presented with increasing back pain when she was due to start her sixth cycle of chemotherapy. She subsequently developed numbness in her feet and loss of power in her legs. An MRI scan showed a mildly enhanced soft tissue mass posterior to the T9, causing moderate cord compression (Figure 3). The patient was treated with urgent radiotherapy (five fractions). She subsequently had another MRI of her spine, which showed resolution of this mass. Her PET scan was clear, as was her bone marrow.
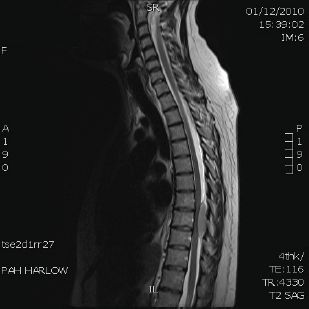
Figure 3: MRI scan of spine shows soft tissue mass posterior to T9 causing moderate cord compression.
Further chemotherapy (with velcade and dexamethasone) was given, also with consideration for sibling allogenic transplantation.
Three months later, while she was on velcade chemotherapy, she developed a lump in her right breast. A biopsy confirmed recurrent disease. This was the non-irradiated breast. A few days later, she developed two lumps in her left breast (the irradiated breast), although at a different site from her original disease. A biopsy again confirmed recurrence. At this stage, she was treated with PAD (bortezomib, doxorubicin, dexamethasone) followed with an allogenic bone marrow transplant from her HLA-identical match brother. The transplantation was sadly complicated with a neutropenic chest infection that lead to her death.
Discussion
Breast plasmacytoma is a rare type of EMP, which can occur in the context of multiple myeloma. EMP usually occurs in the head and neck area in 90% of cases, for example, the upper respiratory tract, nasal cavity, paranasal sinuses, oropharynx, and salivary glands [1–3].
The incidence of breast plasmacytomas is very low—only 63 cases were reported between 1928 and 2009. The reported cases were 66% unilateral and 77% associated with myeloma. Regarding these data, the number of reported cases of solitary breast plasmacytoma is about 15–16 cases over the last 80 years [4–12].
Breast plasmacytoma can be misdiagnosed as primary breast cancer. Usually it does not have specific radiological or clinical features [8]. According to 2009 British Committee for Standards in Haematology (BCSH) guidelines, the diagnostic criteria of SEP [13] are as follows:
No M-protein in serum and/or urine
Extramedullary tumour of clonal plasma cells
Normal bone marrow
Normal skeletal survey
No related organ or tissue impairment.
It is well known that plasmacytomas are radiosensitive, with success rates of 79%–90% and a 10-year survival rate of 50%–100% [5–12]. Solitary EMPs have better prognosis than solitary bony plasmacytomas and can be cured by local radiotherapy [5–7].The rate of local recurrence after radiotherapy is less than 5% [14]. In addition, the risk of distant relapse is more than 30%, which is less than that seen in solitary bony plasmacytoma [15].
According to the BCSH guidelines of 2009, recurrent solitary plasmacytoma beyond the original site of radiotherapy, in the continuing absence of systemic disease, may be treated with additional radiotherapy. Patients with more extensive disease or early relapse (as in our patient) may benefit from systemic therapy with or without autologous stem cell transplantation, as indicated for myeloma, with small case series, suggesting long-term disease control. Newer agents including thalidomide and bortezomib have also been used successfully, prior to transplantation, in small numbers of patients with relapsed plasmacytoma.
Conclusion
Primary breast plasmacytoma usually displays mild clinical behaviour and long patient survival, similar to other plasmacytomas originating from soft tissues [5–6]. The majority of cases can be cured by radiotherapy [7]. Breast plasmacytoma can be misdiagnosed as primary breast cancer. Usually, it does not have specific radiological or clinical features [8]. Careful follow-up is needed as some cases may develop multiple myeloma even after long-term remission [9].
References
1. Van Nieuwkoop C, Giard RW, Veen HF and Dees A (2001) Extramedullary plasmacytoma of the breast simulating breast cancer Neth J Med 58 174–6 DOI: 10.1016/S0300-2977(01)00091-2 PMID: 11325494
2. Brinch L, Hannisdal E, Abrahamsen AF, Kvaloy S and Langholm R (1990) Extramedullary plasmacytomas and solitary plasma cell tumors of bone Eur J Haematol 44 132–5 DOI: 10.1111/j.1600-0609.1990.tb00364.x PMID: 2318296
3. Wax MK, Yun KJ and Omar RA (1993) Extramedullary plasmacytomas of the head and neck Otolaryngol Head Neck Surg 109 877–85 PMID: 8247569
4. Vasiliu T and Popa R (1928) Forme gastrointestinale des tumeurs dites plasmacytomes Compr Rend Soc Biol 98 738–40
5. Kaviani A, Djamali-Zavareie M, Noparast M and Keyhani-Rofagha S (2004) Recurrence of primary extra medullary plasmacytoma in breast both simulating breast carcinoma World J Sur Oncol 2(1) 29 DOI: 10.1186/1477-7819-2-29
6. Wiltshaw E (1976) The natural history of extramedullary plasmacytoma and its relation to solitary myeloma of bone and myelomatosis Medicine 55 217–38 DOI: 10.1097/00005792-197605000-00002 PMID: 1272069
7. Dimopoulos MA, Kiamouris C and Moulopoulos LA (1999) Solitary plasmacytoma of bone and extramedullary plasmacytoma Hematol Oncol Clin North Am 13 1249–57 DOI: 10.1016/S0889-8588(05)70124-6
8. Surov A, Holzhausen HJ and Ruschke K (2010) Breast plasmacytoma 51(5) 498–504 DOI: 10.3109/02841851003712924
9. De Chiara A, Losito S, Terracciano L, Di Giacomo R, Laccarino G and Rubolotta M (2001) Primary plasmacytoma of the breast Arch Pathol Lab Med 125 1078–80 PMID: 11473462
10. Ben-Yehuda A, Steiner-Saltz D, Liboson E and Polliack A (1989) Plasmacytoma of the breast: unusual initial presentation of myeloma: report of two cases and review of the literature Blut 58 169–70 DOI: 10.1007/BF00320440 PMID: 2649190
11. Lamy O, Von Bremen K and Burckhardt P (2000) Breast plasmacytoma Leuk Lymphoma 37 611–5 DOI: 10.3109/10428190009058514 PMID: 11042522
12. Shui Cao, Kang H, Liu Y and Ren X (2009) Synchronous infiltrating ductal carcinoma and primary extramedullary plasmacytoma of the breast World Surg Oncol 7 43 DOI: 10.1186/1477-7819-7-43
13. United Kingdom Myeloma Forum (2009) Guidelines on the diagnosis and management of solitary plasmacytoma of bone, extramedullary plasmacytoma and multiple solitary plasmacytomas http://www.bloodmed.com/contentimage/guidelines/3454.pdf
14. Liebross RH, Ha CS, Cox JD, Weber D, Delasalle K and Alexanian R (1999) Clinical course of solitary extramedullary plasmacytoma Radiother Oncology 52 245–9 DOI: 10.1016/S0167-8140(99)00114-0 PMID: 10580871
15. Mayr NA, Wen BC, Hussey DH, Burns CP, Staples JJ, Doornbos JF and Vigliotti AP (1990) The role of radiation therapy in the treatment of solitary plasmacytomas Radiother Oncol 17 293–303 PMID: 2343147






