Treatment outcomes of paediatric chest wall Ewing’s sarcoma; report from tertiary care cancer center
Rabia Wali1a, Sadia Anjum2, Sumera Abdul Kareem3 and Najma Shaheen1
1Department of Paediatric Oncology, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore 54000, Pakistan
2Department of Radiation Oncology, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore 54000, Pakistan
3Department of Paediatric Hematology and Oncology, University of Child Health Sciences, Lahore 54600, Pakistan
ahttps://orcid.org/0000-0002-8252-3977
Abstract
Introduction: Chest wall Ewing sarcoma (EWS) is a common paediatric malignant tumour affecting the chest wall, accounting for about 10% of all EWS patients. Treatment typically involves intense chemotherapy, complete surgical resection when possible and/or radiation. The EWS of the chest wall has a reported 5-year survival ranging between 40% and 60% in most studies. The following study evaluated the effect of different treatment modalities on survival outcomes in paediatric patients.
Methodology: This is a single-center retrospective cohort study that included 46 patients diagnosed with chest wall EWS between January 2011 and December 2023 at Shaukat Khanum Memorial Cancer Hospital and Research Center. Approval was obtained from the Institutional Review Board for this study. The 5-year follow up period and outcomes were analysed using the SPSS.
Results: The cohort included 46 patients (56% female, 44% male) with a median age of 10 years. Non-metastatic disease was observed in 94% of the patients. The compressed vincristine, doxorubicin, cyclophosphamide, ifosfamide and etoposide chemotherapy regimen showed better outcomes, with a 48% survival rate compared with 38% for vincristine, doxorubicin, actinomycin, ifosfamide and etoposide. Negative surgical margins were associated with a 56% survival rate. 5-year overall survival (OS) of 42.5% and event-free survival (EFS) of 39%. Both the OS and EFS were better in patients who had surgery alone as local control (LC).
Conclusion: Outcomes in patients who had surgery as LC did better in this study. Early recognition and referral may help in reducing the tumour burden so that good LC with surgical R-zero resection is possible.
Keywords:Ewing sarcoma, chest wall, paediatric oncology, chemotherapy, survival analysis, surgical margins
Correspondence to: Rabia Wali
Email: rwali@skm.org.pk
Published: 01/05/2026
Received: 22/09/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Ewing sarcoma (EWS) is an aggressive malignant tumour that mainly affects children and adolescents, with the chest wall being one of the common sites where it can develop [1]. This tumour type arises from primitive neuroectodermal tissue and is characterised by translocation involving the EWSR1 gene, which results in the production of a fusion protein that promotes tumour growth [2]. EWS accounts for about 10% of all paediatric chest wall cancers [3]. Managing paediatric EWS of the chest wall presents significant challenges due to the tumour’s location and its potential for metastasis [4].
Chemotherapy followed by surgical resection and radiotherapy (RT) is the primary treatment for EWS, aiming to achieve the best optimal tumour response while minimising complications and preserving function [5]. Surgical approaches often involve complex multidisciplinary planning, including thoracic, spinal surgery and orthopedic oncology, to ensure complete excision of the tumour while maintaining chest wall integrity [6]. Chemotherapy plays a critical role in managing systemic diseases, improving survival rates and reducing the risk of local recurrences. Despite advancements in treatment strategies, the reported 5-year overall survival (OS) rate for patients with chest wall EWS remains between 40% and 60% [7]. Managing EWS is further complicated by its aggressive clinical behaviour, rapid metastatic potential and high recurrence rate [8]. Tumours with associated malignant pleural effusion and nodules can add to the complexities in achieving local control (LC). Since chest wall involvement is less common, clinical outcome data are limited. Given the long-term side effects of treatment, including cardiac and pulmonary complications, growth disturbances and secondary malignancies associated with chemotherapy or radiation, long-term follow-up is essential to monitor for recurrence and late complications [9]. In recent years, advances in imaging techniques, surgical approaches and chemotherapy protocols have significantly improved the outcomes of EWS [10].
Given the rarity of this condition, this study aimed to evaluate the clinical outcomes of paediatric patients diagnosed with EWS of the chest wall who were treated with chemotherapy, surgical resection where possible and RT in the setting of a low-income country. Outcomes based on primary treatment modalities, including chemotherapy regimens and LC, along with tumour size, were examined.
Methodology
This single-center retrospective cohort study was conducted at the Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, Pakistan. The study included all paediatric patients under 18 years of age diagnosed with EWS of the chest wall between January 2011 and December 2022. Newly diagnosed patients with chest wall Ewing confirmed on histology were included in the analysis. Data were gathered from the hospital’s electronic medical records, including the following parameters: (1) demographics: age, sex and socioeconomic status, (2) tumour characteristics: tumour size, location, histopathology and staging, (3) treatment details: chemotherapy regimens, surgical resection and RT and (4) outcomes: OS, event-free survival (EFS). Approval was obtained from the Institutional Review Board (EX-16-10-19-A2).
From March 2011 to February 2017, vincristine, doxorubicin, actinomycin, Ifosfamide and etoposide (VIDE/VAI) regimen was administered at a standard 3-week interval. In April 2017, the treatment regimen was shifted to a biweekly, intensified chemotherapy protocol – the vincristine, doxorubicin, cyclophosphamide, Ifosfamide and etoposide (VDC/IE) protocol – which showed improved survival outcomes [11]. The switch was done as delivered in an outpatient setting, reducing inpatient bed use and wait times. Three cycles of each VDC and IE were given 2 weeks apart. Reassessment scans were done after five cycles of neoadjuvant chemo for both protocols. LC was achieved through surgery, RT or both, done after six cycles of chemotherapy. A chest computed tomography (CT scan) was performed at baseline and at reassessment. A bone scan and bilateral bone marrow aspiration biopsy were done at baseline to identify metastatic disease. RT was given to patients with positive surgical margins and for whom surgery was not possible. The radiation dose ranged from 40 to 55.8 Gys, depending on surgical margins, resection completeness and resectability, for both chemo-regimens. The final follow up date for this study was 31st December 2023, and 5-year outcomes were performed. OS was defined as the time from diagnosis to last follow-up or death (due to disease or other causes), whichever came first. EFS is the time between a diagnosis and recurrence, progression or death. An Excel was designed to collect the variables and survival probabilities were performed by using Kaplan–Meier survival analysis for over-all outcomes, between different treatment modalities and two chemo-regimens. The rest of the analysis across groups is univariate and bivariate descriptive statistics. Statistical analyses were conducted using SPSS version 29.0, IBM Corp. (USA).
Results
A total of 46 patients with EWS of the chest wall were included in this study. The cohort was composed 56% females and 44% males and 56% were older than 10 years. The age range was from 0.7 to 16 years, with a median age of 10 years. Most tumours originated from the ribs (78%), followed by the thoracic spine (15%) and clavicle/shoulder (7%). Fluorescence in situ hybridization for EWSR1-FLI-1 was performed in 65% of patients after it was introduced by the pathology department in our institution in 2014. Most patients (94%) had non-metastatic disease. At the end of treatment, 39% of cases were in complete remission. At the end of the analysis, 45% are alive, while 55% had died. Disease progression was the main cause of death in 83% of patients, mainly occurring after local treatment and just before the completion of adjuvant chemotherapy.
Treatment-related deaths were observed in four patients (17%) caused by bacteremia-related sepsis and cardiogenic shock. One patient abandoned treatment and later died due to disease progression. Three patients developed myocardial dysfunction – one during adjuvant chemotherapy and two more developed it 2 years after treatment. Additionally, one patient developed secondary acute lymphoblastic leukaemia 2 years post-treatment 2 year and died from sepsis.
Table 1 shows the patient characteristics for both groups, VIDE/VAI and VDC/IE, while Table 2 shows outcomes for the two chemotherapy protocols. Among the 33 patients treated with VDC/IE, 48% survived at the end of the study and disease progression was noted in 39%. In contrast, only 38% of the 13 patients who received VIDE/VAI survived, with disease progression noted in 62%. However, the difference between the two groups is not statistically significant (
Table 1. Patient characteristics.
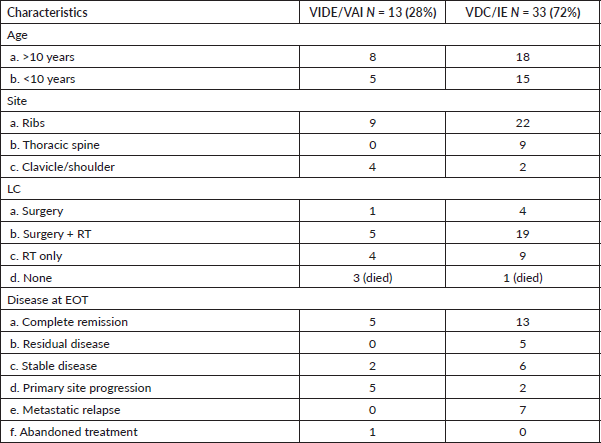
Table 2. Outcomes comparison between VDC/IE and VIDE/VAI chemo regimens (N = 46).
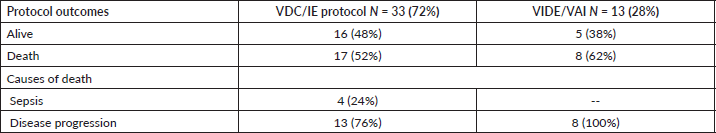
Table 3. Outcomes comparison with surgical margins, tumour sizes and age (N = 29).
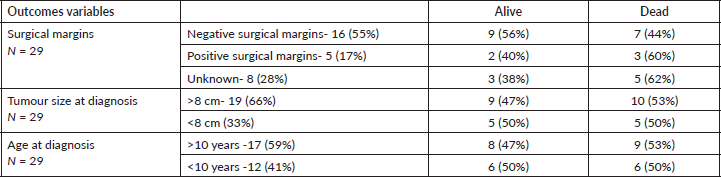
The outcomes of the 29 patients who underwent surgery (
RT alone as local treatment was given to 13(28%) patients because the chest tumour could not be surgically removed. Of these, seven (54%) are alive and in remission, while the remaining six (46%) died either from local progression or after developing metastatic disease at the end of treatment or on follow up.
The OS and EFS were analysed using Kaplan–Meier survival curves (Figure 1). The 5-year OS rate is 42.5%, and the 5-year EFS is 39%. OS for patients who had surgery alone for LC was 62%, compared to 52% with RT and 35% for with both surgery and radiation. Similarly, the EFS was better in those patients who had surgery alone, 65%, versus those who received RT alone, 52% and 35% in those with both surgery and RT as LC (Figure 2).
Discussion
Chest wall EWS is a rare and aggressive tumour that requires multimodal treatment, including surgery and RT, to improve patient outcomes [12]. It often metastasizes and has a higher risk of local recurrence. Reported outcomes of chest wall EWS in the literature vary widely, with 5-year LC, EFS and OS from72% to 97%, 34% to 74% and 34% to 81.6%, respectively [13]. This study presents outcomes of 46 patients with chest wall EWS treated with two chemotherapy regimens, surgery and RT in a lower-middle-income country. Our institution is one of the largest tertiary care cancer center, and we report our experience over the past 12 years.
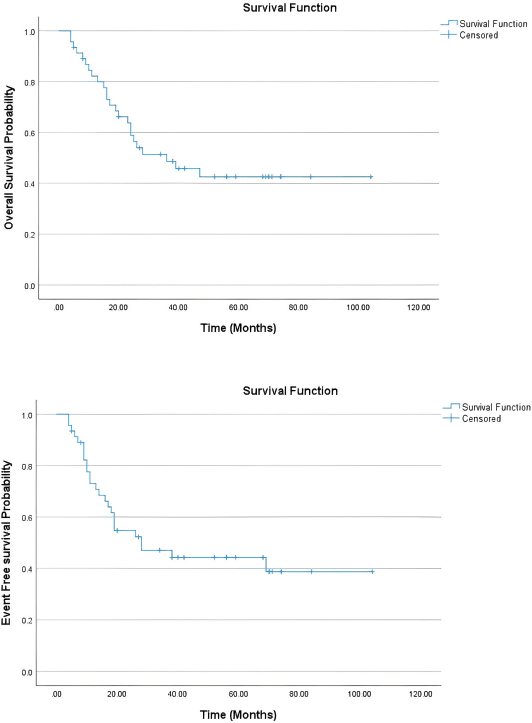
Figure 1. 5-year OS and EFS in chest wall EWS.
Included population
The peak incidence for EWS happens between 13 and 16 years old, with more male affected. In our study, 56% of patients were aged 10–18 years, with a nearly equal male-to-female ratio of 1:1.2 [14]. In 2017, the treatment protocol was switched to compressed cycles using the Children’s Oncology Group VDC/IE chemo regimen, replacing the Euro-Ewing protocol VIDE/VAI. This change aimed to improve survival outcomes, reduce hospital stays, and prevent delays in chemotherapy administration due to the long waiting list. A feasibility audit was performed to evaluate the outpatient administration of VDC/IE, it was found to be safe. We noted less toxicity and febrile neutropenic complications with this combination and patient tolerated it well [15].
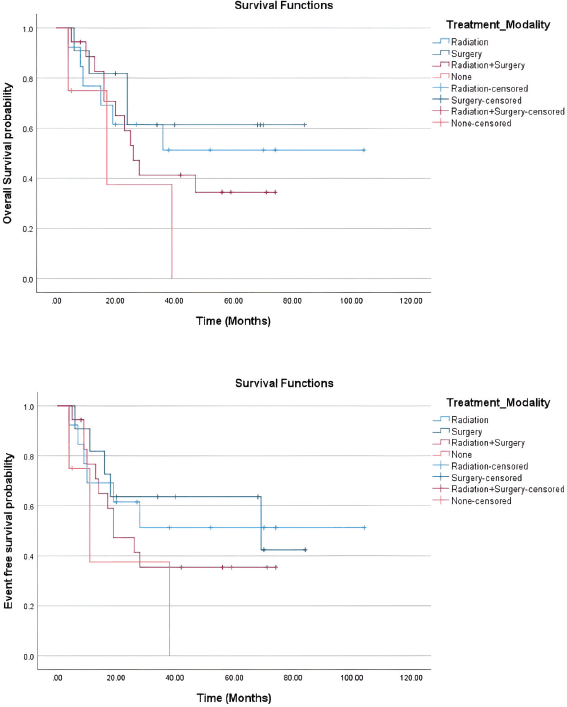
Figure 2. 5-year OS and EFS in patients treated with differential LC.
The comparison of outcomes between VDC/IE and VIDE/VAI chemotherapy regimens revealed notable differences in survival. Patients treated with VDC/IE demonstrated higher survival rates (48% alive) than those treated with VIDE/VAI (38% alive), but this difference was not statistically not significant (
Disease progression reached 100% in the VIDE/VAI group, likely due to a delay in starting this regimen on time due to a long waiting admission list and delays in count recovery. Patients tolerated VDC/IE regimen and it was changed to be given in the outpatient setting to prevent no delays.
Ewing’s sarcoma is chemo-radio sensitive tumour, so neo-adjuvant chemotherapy can help reduce the morbidity associated with of surgery, especially in the chest wall. If R zero (RO) resection is achieved, radiation can be avoided. However, removing the soft tissue chest wall mass along with the involved ribs to obtain negative margins is not feasible with large tumour size. In our study, we observed a 56% survival among patients with negative surgical margins. Reported outcomes in the literature range from 60% to 90% [16]. The lower survival rate in our cohort could be due to tumour size larger than 8 cm time, late diagnosis and in five cases, the margin status was unknown because surgeries were performed elsewhere, so no details were available. Shamberger
Our results showed an overall 5-year survival rate (OS) of 42.5%, with a median survival time of 36 months. The EFS was 39%. In a study by Bedetti, OS was 91% and EFS 65% at 5 years. In a review of 104 patients, Laskar
OS in patients who underwent surgery alone as LC was better than that of those who received RT or had both surgery and radiation. Similarly, the EFS was higher in patients who had surgery alone than in those who received RT alone or had both surgery and RT as LC.
Complete resection is essential, and studies have shown that patients with non-metastatic disease who undergo complete surgical resection and chemotherapy, with or without RT, experience favorable outcomes and long-term survival [21]. Neo adjuvant chemotherapy helps achieve complete tumour resection along with negative margins, as Ewing’s is very chemo sensitive. Denbo
The impact of RT on survival is unclear, as no randomised trials have directly compared local treatment (surgery plus RT) with surgery alone. Indelicato
Long-term follow-up is crucial for identifying late complications, monitoring recurrence and managing treatment-related toxicities, such as cardiotoxicity and secondary malignancies [2]. In our study, three patients developed myocardial dysfunction during disease progression and one patient had secondary acute lymphoblastic leukaemia 1 year after treatment and died from sepsis.
One of the limitations of this study was its retrospective, single-center design, small sample size, reliance on descriptive statistics and the omission of pleural involvement. Studies show that pleural involvement or effusion as an important prognostic factor and associated with worse survival outcomes, similar to those reported in metastatic Ewing’s Sarcoma [14].
Additionally, our center accepts patients from all across the country and even from across the border which can cause delays in presentation. Patients often have locally advance disease, which effects their treatment response, options for LC and ultimately their outcomes, even though they are treated according to standard of care protocols.
Conclusion
This study reports the outcomes of chest wall EWS from a resource-limited country. Patients who underwent surgery for LC showed better results. Early recognition and referral may help in reducing the tumour burden, allowing for effective LC with surgical RO resection, which can prevent long-term effects of RT. Access to a multidisciplinary team and close collaboration at the national level are essential to improve outcomes through the development of national treatment guidelines.
List of abbreviations
EFS, event-free survival; EWS, Ewing sarcoma; LC, local control; OS, overall survival; RT, radiation; VDC/IE, vincristine, doxorubicin, cyclophosphamide, ifosfamide and etoposide; VIDE/VAI, vincristine, doxorubicin, actinomycin, Ifosfamide and etoposide.
Acknowledgment
The authors would like to thank the patients and care givers and Ms. Rabia Anjum for data analysis.
Conflicts of interest
The authors declared no conflicts of interest during study and conduction and submission.
Funding
This study received no funding.
Ethical statement
This study was approved by the Institutional Review Board of the Shaukat Khanum Memorial Cancer Hospital & Research Centre (SKMCH), Lahore, Pakistan (Dated: July 2024).
Patient consent
No consent needed as retrospective data.
Author contributions
Conception and design: Najma Shaheen, Sadia Anjum, Sumera. A. Karim. Collection and assembly of data: all authors. Data analysis and interpretation: Rabia Wali, Sadia Anjum. Manuscript writing: Rabia Wali, Najma Shaheen, Sadia Anjum. Final approval of manuscript: All authors. Accountable for all aspects of the work: All authors.
Data availability
Available on request.
References
1. La Quaglia MP (2008) Chest wall tumors in childhood and adolescence Seminars in pediatric surgery (New York: Department of Surgery, Memorial Sloan–Kettering Cancer Center) p 173 https://doi.org/10.1053/j.sempedsurg.2008.03.007 PMID: 18582823
2. Anju TR and Jayanarayanan S (2021) Neurooncogenesis in the development of neuroectodermal cancers The Molecular Immunology of Neurological Diseases (Kerala: Newman College) p 85
3. Gupta AA, Pappo A, and Saunders N, et al (2010) Clinical outcome of children and adults with localized Ewing sarcoma: impact of chemotherapy dose and timing of local therapy Cancer 116(13) 3189–3194 https://doi.org/10.1002/cncr.25144 PMID: 20564643
4. Theodorou CM, Lawrence YS, and Brown EG (2023) Chest wall reconstruction in pediatric patients with chest wall tumors: a systematic review J Pediatr Surg 58(7) 1368–1374 https://doi.org/10.1016/j.jpedsurg.2022.11.008
5. Lin GQ, Li YQ, and Huang LJ, et al (2015) Chest wall tumors: diagnosis, treatment and reconstruction Exp Ther Med 9(5) 1807–1812 https://doi.org/10.3892/etm.2015.2353 PMID: 26136897 PMCID: 4471685
6. Rice D, Barrett S, and Lavan N, et al (2022) Evaluating the role of surgical resection and reconstruction in the management of ewing sarcoma of the chest wall in the paediatric population: a systematic review Surg Oncol 131(6) 1165–1187
7. Martin E, Senders JT, and Ter Wengel PV, et al (2019) Treatment and survival of osteosarcoma and Ewing sarcoma of the skull: a SEER database analysis Acta Neurochir (Wien) 161 317–325 https://doi.org/10.1007/s00701-018-3754-y PMCID: 6373276
8. Tseng WW, Swallow CJ, and Strauss DC, et al (2022) Management of locally recurrent retroperitoneal sarcoma in the adult: an updated consensus approach from the Transatlantic Australasian Retroperitoneal Sarcoma Working Group Ann Surg Oncol 29(12) 7335–7348 https://doi.org/10.1245/s10434-022-11864-y PMID: 35767103
9. Indelicato DJ, Keole SR, and Lagmay JP, et al (2011) Chest wall Ewing sarcoma family of tumors: long-term outcomes Int J Radiat Oncol Biol Phys 81(1) 158–166 https://doi.org/10.1016/j.ijrobp.2010.04.066
10. Jacobs AJ, Fishbein J, and Levy CF, et al (2016) Chest wall Ewing sarcoma: a population-based analysis J Surg Res 204(2) 475–480 https://doi.org/10.1016/j.jss.2016.05.033 PMID: 27565085
11. Womer RB, West DC, and Krailo MD, et al (2012) Randomized controlled trial of interval-compressed chemotherapy for the treatment of localized Ewing sarcoma: a report from the Children’s Oncology Group J Clin Oncol 30(33) 4148–4154 https://doi.org/10.1200/JCO.2011.41.5703 PMID: 23091096 PMCID: 3494838
12. Cardenas CA (2023) Ewing sarcoma: review of recent advances and directions Clin Oncol Case Rep 6(8) 2
13. Laughlin BS, Bogan A, and Allen-Rhoades WA, et al (2025) Comprehensive analysis of treatment approaches in chest wall ewing sarcoma: the impact of tumor volume on oncologic outcomes Adv Radiat Oncol 10(4) 101729
14. Zöllner SK, Amatruda JF, and Bauer S, et al (2021) Ewing sarcoma—diagnosis, treatment, clinical challenges and future perspectives J Clin Med 10(8) 1685 https://doi.org/10.3390/jcm10081685
15. Sarfraz S and Saeed H (2022) Feasibility and safety of outpatient administration of ifosfamide and etoposide for pediatric patients with Ewing sarcoma in a resource-limited setting amid the COVID-19 pandemic Pediatr Blood Cancer 69(5) e29595 https://doi.org/10.1002/pbc.29595 PMID: 35234343 PMCID: 9088619
16. Salimbene O, Viggiano D, and Muratori F, et al (2024) Primary chest wall Ewing sarcoma: treatment and long-term results Life (Basel) 14(6) 766 PMCID: 11204814
17. Van Roozendaal LM, Bosmans JWAM, and Daemen JHT, et al (2024) Management of soft tissue sarcomas of the chest wall: a comprehensive overview J Thorac Dis 16(5) 3484 https://doi.org/10.21037/jtd-23-1149 PMID: 38883634 PMCID: 11170428
18. Seitz G, Urla C, and Sparber‐Sauer M, et al (2019) Treatment and outcome of patients with thoracic tumors of the Ewing sarcoma family: a report from the Cooperative Weichteilsarkom Studiengruppe CWS‐81,‐86,‐91,‐96, and‐2002P trials Pediatr Blood Cancer 86 e27884 https://doi.org/10.1002/pbc.27884
19. Zogopoulos G, Teskey L, and Sung L, et al (2004) Ewing sarcoma: favourable results with combined modality therapy and conservative use of radiotherapy Pediatr Blood Cancer 43(1) 35–39 https://doi.org/10.1002/pbc.20037 PMID: 15170887
20. Provost B, Missenard G, and Pricopi C, et al (2018) Ewing sarcoma of the chest wall: prognostic factors of multimodal therapy including en bloc resection Ann Thorac Surg 106(1) 207–213 https://doi.org/10.1016/j.athoracsur.2018.02.031 PMID: 29551629
21. Denbo JW, Shannon Orr W, and Wu Y, et al (2012) Timing of surgery and the role of adjuvant radiotherapy in Ewing sarcoma of the chest wall: a single-institution experience Ann Surg Oncol 19(12) 3809–3815 https://doi.org/10.1245/s10434-012-2449-5 PMID: 22752372 PMCID: 3529468
22. Shamberger RC, LaQuaglia MP, and Krailo MD, et al (2000) Ewing sarcoma of the rib: results of an intergroup study with analysis of outcome by timing of resection J Thorac Cardiovasc Surg 119(6) 1154–1161 https://doi.org/10.1067/mtc.2000.106330 PMID: 10838532
23. Bottinor W and Chow EJ (2022) Mitigating, monitoring, and managing long-term chemotherapy-and radiation-induced cardiac toxicity Hematology 1(1) 251–258 https://doi.org/10.1182/hematology.2022000342
24. Curigliano G, Cardinale D, and Dent S, et al (2016) Cardiotoxicity of anticancer treatments: epidemiology, detection, and management CA Cancer J Clin 66(4) 309–325 PMID: 26919165
25. Laskar S, Nair C, and Mallik S, et al (2011) Prognostic factors and outcome in Askin-Rosai tumor: a review of 104 patients Int J Radiat Oncol Biol Phys 79(1) 202–207





