Clinical outcomes of adding vincristine to procarbazine-lomustine in patients with gliomas: insights from current evidence
Viviana Pinzón-Ramírez1a, Luis E Cueva-Cañola2,3,4b, Dilmareth E Natera5c, Helder Edgar Aldo-Chávez Olivera2d, Oscar Eduardo Camacho-Hernández2,6e, Andrea C Beltran-De la Fuente2,7f, Sergio Alexis Ramirez-Alvarez2,8g, Einstein Yhair Gallardo Cubas2h, Giomar Vilca Flores2,9i, Mauricio E Gamez2,10j and Leonardo Rangel Castilla2,11k
1School of Medicine, Universidad Pedagógica y Tecnológica de Colombia (UPTC), Tunja, Boyacá 150003, Colombia
2Neurology Research Club, Universidad Nacional de Piura (UNP), Piura 20000, Peru
3Department of Neurosurgery, Instituto Nacional de Ciencias Neurológicas (INCN), Lima 15003, Peru
4Department of Neurosurgery, Hospital III-1 EsSalud José Cayetano Heredia, Piura 20007, Peru
5Department of Neurosurgery, University of Minnesota, Minneapolis, MN 55455, USA
6Department of Medicine and Nutrition, Health Sciences Division, University of Guanajuato, Leon Campus, Leon, Guanajuato 37544, Mexico
7School of Medicine, Universidad México Americana Del Norte (UMAN), Reynosa, Tamaulipas 88500, México
8School of Medicine, Universidad Autonoma de Guadalajara (UAG), Guadalajara, Jalisco 45129, Mexico
9School of Medicine, Universidad Católica de Santa María, Umacollo, Arequipa 04017, Peru
10Creo Cancer Center, Universidad Autonoma de San Luis Potosi (UASLP), San Luis Potosi 78210, Mexico
11Department of Neurosurgery, Hospital Lomas International, San Luis Potosí 78218, Mexico
a https://orcid.org/0009-0008-6525-6468
b https://orcid.org/0009-0003-6320-5767
c https://orcid.org/0009-0001-5256-9798
d https://orcid.org/0009-0000-2294-5038
e https://orcid.org/0009-0000-3141-3041
f https://orcid.org/0009-0009-9895-0824
g https://orcid.org/0009-0009-0024-288X
h https://orcid.org/0009-0000-3785-6249
i https://orcid.org/0000-0001-8232-0806
j https://orcid.org/0009-0009-0277-5698
k https://orcid.org/0000-0002-7915-6205
Key points
-
Procarbazine–lomustine combination (PC) shows superior progression-free survival and overall survival versus procarbazine, lomustine and vincristine (PCV) in gliomas.
-
Omitting vincristine may improve efficacy and reduce neurotoxicity.
-
Findings suggest PC could be a safer alternative, pending confirmation by randomised controlled trials.
Importance of the study
This meta-analysis is the first to directly evaluate whether vincristine is necessary within the procarbazine, lomustine and vincristine (PCV) regimen for gliomas. By synthesising data from three retrospective cohorts, it demonstrates that the procarbazine–lomustine combination (PC) achieved significantly longer progression-free and overall survival compared with PCV, while markedly reducing neurotoxicity and treatment discontinuation. These findings challenge the traditional view that vincristine is an indispensable component of chemotherapy for diffuse gliomas. Given that temozolomide has become the preferred agent in contemporary practice, clarifying the relative contributions of PC, PCV and TMZ remains essential for optimising therapeutic strategies. Although limited by the small number and retrospective design of included studies, this analysis suggests that PC may represent a more effective and tolerable alternative, underscoring the need for prospective randomised trials in the modern molecular era of glioma classification.
Abstract
Background Temozolomide (TMZ) is the standard chemotherapy for gliomas due to good tolerability. Clinical studies suggest a combination of procarbazine, lomustine (CCNU) and vincristine (PCV) provides superior survival and progression delay, but toxicities limit use. Given the ongoing debate over the role of vincristine in glioma chemotherapy amid the dominance of TMZ, simplified procarbazine and CCNU (PC) regimens warrant renewed evaluation.
Methods Three databases were searched up to August 2025 to identify studies that directly compared PC and PCV in patients with low-grade and high-grade gliomas. Outcomes included progression-free survival (PFS), overall survival (OS) and treatment-related toxicities. Hazard ratios (HRS) and relative risks (RRS) were then pooled using a random-effects model.
Results In a total population of 301 patients with low- and high-grade gliomas, PC significantly reduced the risk of disease progression compared with PCV (HR = 0.72, 95% confidence interval (CI): 0.53–0.98;
Conclusion With the limited evidence available to date, we found that PC achieved significantly longer PFS and OS than PCV, while maintaining a better safety profile. We recommend further well-designed and adequately powered randomised clinical trials directly comparing TMZ, PC and PCV to determine which patient populations and glioma subtypes – according to the current World Health Organisation 2021 classification – derive the greatest survival benefit and tolerability from each regimen.
Keywords: glioma, procarbazine, lomustine, vincristine, meta-analysis
Correspondence to: Leonardo Rangel Castilla.
Email:Lrcastilla@hotmail.com
Published: 01/05/2026
Received: 08/10/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Diffuse gliomas account for 77%–80% of malignant primary brain tumours and nearly 30% of all primary brain tumours worldwide. These include both low-grade (World Health Organisation (WHO) grade II) and high-grade (WHO grade III–IV) gliomas [1]. Since the 1990s, adjuvant chemotherapy has become an essential component of glioma management [2, 3]. The combination of procarbazine, lomustine (CCNU) and vincristine (PCV), when added to radiotherapy, has demonstrated significant improvements in overall survival (OS) and progression-free survival (PFS) in randomised controlled trials (RCTs), particularly among patients with anaplastic oligodendrogliomas harboring 1p/19q–codeleted, in whom PCV plus radiotherapy outperformed radiotherapy alone [4, 5].
In current practice, however, temozolomide (TMZ) has largely replaced PCV because of its oral administration, favourable toxicity profile and improved tolerability [6]. Nonetheless, evidence indicates that PCV offers superior efficacy compared to TMZ, especially in WHO grade III isocitrate dehydrogenase (IDH)-mutant oligodendroglioma 1p/19q–codeleted [7, 8]. The major limitation of PCV is its toxicity: grade 3–4 hematologic toxicities, reported in up to 40% of patients, often necessitating early treatment discontinuation [9–11].
Within the PCV regimen, the therapeutic role of vincristine has increasingly been questioned for several reasons, including concerns related to its tolerability, pharmacological properties and overall contribution to treatment efficacy [12]. Emerging data suggest that omitting vincristine, using procarbazine and CCNU (PC) alone, may maintain the survival benefit while reducing toxicity [13].
Comparative data evaluating modified regimens, such as PC without vincristine or TMZ-based approaches, remain limited and largely retrospective [6, 14]. Previous systematic reviews have focused broadly on chemotherapy in gliomas but have not specifically addressed the necessity of vincristine in oligodendroglioma treatment [6]. Given the long survival trajectories of these patients, clarifying the role of vincristine is crucial for balancing efficacy with toxicity.
The objective of this meta-analysis is to synthesize the available evidence from three retrospective cohort studies comparing procarbazine–CCNU regimens with and without vincristine in patients with low- and high-grade gliomas.
Methods
This systematic review and meta-analysis were conducted in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), which establish a structured approach to enhance clarity and reliability in reporting [15]. In addition, to ensure methodological transparency and minimise the possibility of duplication or selective outcome reporting, the protocol was prospectively registered in the International Prospective Register of Systematic Reviews on 25 August 2025 (registration ID: CRD420251134032) [16].
Eligibility criteria
We included studies that enrolled patients with histologically confirmed low- or high-grade gliomas, directly compared PC with PCV and reported at least one relevant clinical outcome. After a comprehensive screening, only three retrospective cohort studies meeting these criteria were identified and included in the synthesis. Studies were excluded if they were preclinical reports, reviews, meta-analyses, case reports, case series, lacked a direct PC versus PCV comparison or were published in languages other than English.
Search strategy
We systematically searched PubMed, Embase and Web of Science for studies published up to 13 August 2025. The search strategy combined controlled vocabulary and free-text terms related to glioma, procarbazine, lomustine and vincristine. The detailed search strategies for each database are provided in Table S1.
Screening of studies
All records retrieved from the database search were imported into Zotero for reference management, where duplicates were removed. The remaining studies were screened in Rayyan through a two-step process. In the first stage, two independent reviewers (SRA and HCO), who were blinded to each other’s decisions, screened titles and abstracts to identify potentially eligible studies. In the second stage, the same reviewers independently assessed the full texts for final inclusion. Any disagreements were resolved through discussion, and if consensus could not be achieved, a third reviewer served as arbitrator (LCC).
Data extraction
Two reviewers (SRA and HCO) independently extracted data using a standardised form, with disagreements resolved by a third reviewer (LCC). Extracted information included study characteristics, treatment details, baseline patient and tumour features and reported clinical outcomes. When necessary, corresponding authors were contacted for clarification and all data were cross-checked for accuracy before analysis.
Outcomes
The predefined outcomes of interest were categorised into efficacy and safety endpoints. Efficacy was evaluated through PFS and OS, which represent the length of time patients lived without disease progression and the survival time from treatment initiation, respectively [6]. Safety outcomes included the occurrence of adverse events, as well as treatment modifications, dose reductions and dose delays, which reflect treatment tolerability in clinical practice [14]. These endpoints were consistently extracted from the included studies, using the longest follow-up available when multiple time points were reported.
Statistical analysis
For time-to-event outcomes (PFS and OS), hazard ratios (HR) with 95% confidence intervals (CIs) were either extracted directly or calculated from available data. The pooled analyses were performed using adjusted HR. For OS, HR were adjusted for age, tumour type, IDH mutation status, 1p/19q–codeleted status, O⁶-methylguanine-DNA methyltransferase (MGMT) promoter methylation status, prior history of chemotherapy and Karnofsky performance status (KPS) [10, 11, 13]. For PFS, HR was adjusted for age, extent of resection before chemotherapy, tumour type and KPS [10, 11, 13].
For safety outcomes, relative risks (RR) with 95% CIs were used to compare adverse events, dose reductions and dose delays between treatment groups. All statistical analyses were performed using Review Manager version 5.3.5 [17].
A random-effects model was applied to account for between-study variability and pooled results were presented in forest plots. Heterogeneity was assessed using the Chi-squared test and quantified with the I² statistic, with I² ≥ 50% or p < 0.10 were considered indicative of substantial heterogeneity [18].
Given that only three retrospective cohort studies met the inclusion criteria, the scope of the statistical analysis was limited. With such a small number of studies, advanced approaches such as meta-regression, subgroup analyses or formal assessments of publication bias (funnel plots and Egger’s test) were not feasible, as they would provide unreliable or uninterpretable results [19].
Risk-of-bias assessment
The risk of bias was assessed using the Risk Of Bias In Non-randomised Studies of Interventions tool, which covers confounding factors, participant selection, classification of interventions, deviations from planned interventions, missing data, outcome measurement and selective reporting [20]. Two reviewers performed the assessment blindly and a third reviewer resolved any disagreements.
Results
Study selection
A total of 838 records were identified through the database search. After removal of duplicates and screening of titles and abstracts, 41 full-text articles were reviewed in detail to evaluate their eligibility. Ultimately, only three studies met all eligibility criteria and were included in the final synthesis. All three were retrospective cohort studies conducted in Germany [10], South Korea [13] and the United States [11]. The study selection process is depicted in the PRISMA flow diagram (Figure 1).
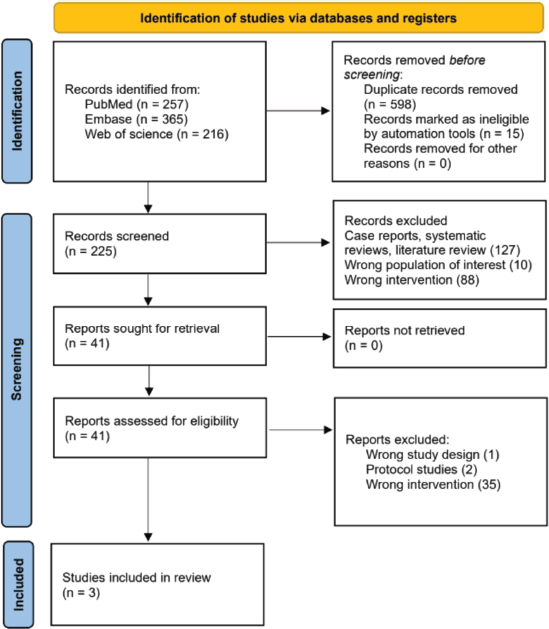
Figure 1. PRISMA 2020 flow diagram.
Included population
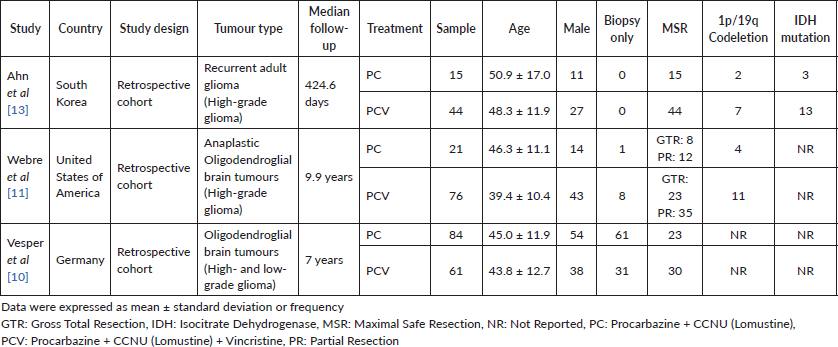
In our study, we analysed 301 patients (187 men and 114 women) with a mean age of 44.2 ± 12.3 years. Of these, 88 (29.2%) had low-grade and 213 (70.8%) high-grade gliomas. Regarding surgical management, 190 (63.1%) patients underwent maximal safe resection at initial surgery. Baseline clinical and molecular characteristics are shown in Table 1, while detailed treatment regimens are summarised in Table S2.
Table 1. Summary of included studies.
Efficacy of chemotherapy regimens
Patients receiving PC had a significantly lower risk of disease progression compared with those receiving PCV (HR = 0.72, 95% CI: 0.53–0.98, p = 0.04). Similarly, OS was significantly longer in the PC group than in the PCV group (HR = 0.59, 95% CI: 0.37–0.95, p = 0.03). No heterogeneity was detected across studies for either outcome. These analyses are shown in Figure 2.
Treatment-related toxicities and tolerability
The pooled analyses revealed that neurotoxicity was significantly less frequent in patients treated with PC compared with PCV (RR = 0.12, 95% CI: 0.02–0.63, p = 0.01; I² = 0%). Similarly, treatment discontinuation due to adverse events was markedly lower with PC (RR = 0.11, 95% CI: 0.02–0.80, p = 0.03; I² = 0%). These analyses are shown in Figure 3.
For hematologic toxicities, no statistically significant differences were observed between groups. Anemia, neutropenia, thrombocytopenia and leukopenia occurred at comparable rates. Likewise, no significant differences were found in allergic reactions, dose reductions or dose delays.
Risk-of-bias assessment
All studies were judged to have a moderate overall risk of bias (Figure 4). The main sources of bias across the included studies were related to their retrospective, non-randomised design, which makes residual confounding likely and the absence of pre-registered protocols, raising the possibility of selective reporting. Additional concerns included treatment deviations in the PCV groups due to toxicity and the inherent limitations of retrospective case capture. However, because important clinical and molecular variables were reported and adjusted for, patient selection was generally appropriate. The key outcomes (OS, PFS, toxicity) were consistently presented, so the overall risk of bias was judged to be moderate rather than serious [10, 11, 13].
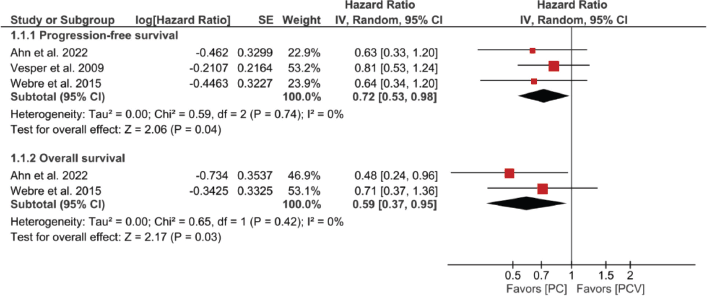
Figure 2. Efficacy of treatment regimens HR analysis.
Discussion
In contemporary clinical practice, TMZ has become the most commonly prescribed chemotherapy for IDH-mutant diffuse gliomas classified according to the WHO grading system, because of its more favorable tolerability profile [21]. However, several studies directly comparing TMZ with PCV have demonstrated that PCV, when combined with radiotherapy, achieves superior outcomes in terms of OS and PFS, albeit at the cost of greater treatment-related toxicity [7, 8, 22–25]. This trade-off between efficacy and tolerability has generated ongoing debate regarding the optimal chemotherapy backbone in this patient population.
To the best of our knowledge, this is the first meta-analysis to provide quantitative evidence that omitting vincristine from the PCV regimen not only preserves but may enhance therapeutic efficacy while substantially improving tolerability. Patients receiving PC had a 41% lower risk of death and a 28% lower risk of disease progression compared with those treated with PCV. These findings suggest that the inclusion of vincristine may not only fail to improve outcomes but could, under certain conditions, be counterproductive. While vincristine has long been considered an integral component of the PCV regimen, its actual contribution to intracranial tumour control has remained uncertain.
Procarbazine exerts its cytotoxicity primarily through the generation of O⁶-methylguanine (O⁶-MeG) adducts, lesions whose biological impact depends critically on deoxyribonucleic acid (DNA) replication and intact mismatch repair (MMR) [26]. During the first Synthesis phase (S phase) after exposure, O⁶-MeG mispairs with thymine, prompting futile MMR cycles that persist into the second cell cycle, when long single-stranded DNA stretches and double-strand breaks emerge and activate Ataxia Telangiectasia and Rad3-related/Ataxia Telangiectasia Mutated, Checkpoint Kinase 1/ Checkpoint Kinase 2, p53 and downstream apoptosis or senescence pathways [21, 27]. CCNU also relies on replication-associated lesion processing: its O⁶-chloroethylguanine adducts convert into interstrand crosslinks (ICLs), whose collapse during replication fork collision triggers double-strand break (DSB) formation, checkpoint activation and cell death [27]. Thus, although ICLs possess intrinsic cytotoxicity, their full lethal potential is amplified when cells progress through S phase, a feature shared with O⁶-alkylating agents.
Vincristine disrupts this replication-dependent cytotoxic cascade by binding β-tubulin, inhibiting microtubule polymerisation and inducing a durable Gap 2 / Mitosis phase arrest [28]. This mitotic blockade markedly reduces the proportion of cells entering successive S phases – the very intervals during which O⁶-MeG lesions and chloroethyl adducts undergo the replication-dependent processing that converts them into DSBs and ICL-derived fork collapse [21, 29, 30].
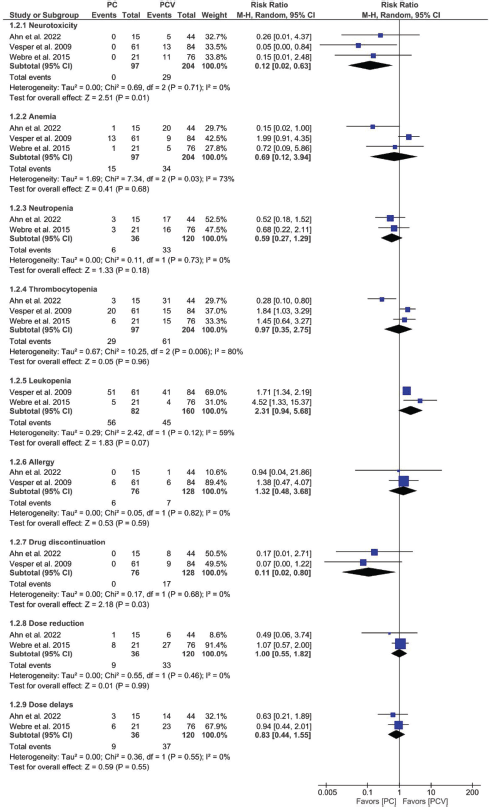
Figure 3. Tolerability of treatment regimens RR analysis.
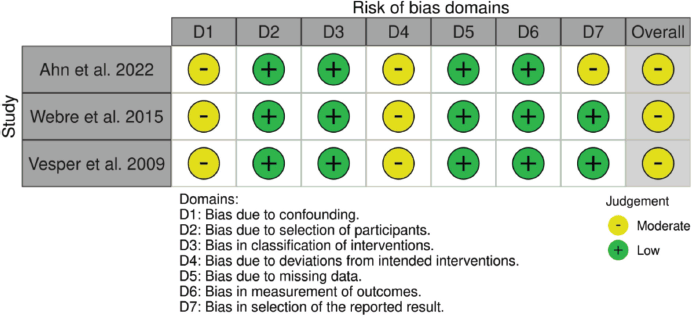
Figure 4. Risk of bias assessment.
Additionally, it is well known that vincristine exhibits low penetration across an intact blood–brain barrier, and although regions of tumour-associated barrier disruption may permit somewhat higher local exposure, its cytotoxic activity remains strictly dependent on active cell division [12, 31, 32].
The omission of vincristine would not only avoid the neurotoxicity related to its binding to neuronal β-tubulin, which presents as impaired axonal transport, peripheral neuropathy and autonomic dysfunction, but would also prevent the dose reductions and treatment interruptions that these toxicities often require [11, 33–35]. Removing this limiting factor may help support more consistent full-dose administration of procarbazine and CCNU, which in turn could facilitate the pharmacodynamic interactions they are expected to rely on through sustained exposure, progressive MGMT depletion and the replication-linked conversion of monoadducts into cytotoxic DNA lesions.
The absence of RCTs directly comparing PC and PCV regimens can be attributed to several practical and ethical barriers. Diffuse gliomas are relatively rare tumours, and demonstrating meaningful differences in OS or PFS would require very large patient cohorts and extended follow-up periods, making such trials logistically and financially challenging [36, 37]. Moreover, once PCV demonstrated survival benefit in earlier studies, it became established as part of the standard of care, reducing clinical equipoise and making it ethically difficult to randomise patients to modified regimens that omit vincristine [7, 8, 25]. Therefore, most available evidence comes from retrospective or observational studies, which are inherently vulnerable to selection bias and confounding by patient characteristics.
The therapeutic landscape of diffuse gliomas is increasingly shaped by molecular profiles, with treatment strategies varying according to grade and risk. For high-risk WHO grade 2 tumours, radiotherapy followed by PCV chemotherapy remains the standard of care, supported by level IA evidence [38], while the recent Food and Drug Administration approval of vorasidenib for IDH1/2-mutant gliomas underscores the role of targeted therapies [39]. In higher grades, management continues to rely on established regimens such as the Stupp protocol for glioblastoma and radiotherapy combined with either TMZ or PCV for anaplastic astrocytomas, particularly in IDH-mutant and 1p/19q–codeleted tumours [40]. Long-term data have shown that PCV provides a significant survival advantage and delays progression in IDH-mutant grade 3 diffuse gliomas compared with TMZ [7, 8]. Our results, however, suggest that this benefit may be driven primarily by the alkylating components of PCV, and that removal of vincristine not only preserves but enhances efficacy while substantially reducing toxicity.
While this study presents interesting findings, several limitations should be acknowledged. The small number of available studies, their retrospective design and variability in treatment details limit the strength of our conclusions; therefore, these results should be interpreted with caution. Our study population was broad and included Ahn et al [13], which evaluated recurrent gliomas, Webre et al [11], focused on anaplastic oligodendroglial brain tumours and Vesper et al [10], which included both low- and high-grade oligodendroglial tumours. Notably, these studies followed different WHO classifications – 2000 for Webre, 2000 for Vesper and 2016 for Ahn – further limiting comparability of results. In addition, the limited dataset prevented meaningful subgroup analyses.
Clinicians should interpret these findings with caution, given the absence of RCTs and the inherent limitations of retrospective data. However, the consistency of the observed survival benefit, together with the substantially improved tolerability profile and the strong mechanistic rationale supporting vincristine omission, suggests that the PC regimen represents a clinically meaningful and biologically plausible alternative to PCV.
Rather than prompting immediate changes to universal treatment guidelines, our results support the consideration of vincristine-free regimens in carefully selected clinical contexts, particularly for patients at increased risk of neurotoxicity or those unable to tolerate full-dose PCV. In this sense, the present meta-analysis provides a robust evidence-based framework that may assist clinicians and institutions in rationally evaluating and, where appropriate, implementing the PC protocol while awaiting confirmation from prospective studies. Future investigations should directly compare TMZ, PCV and PC across molecularly defined glioma subtypes, according to the WHO 2021 classification, to determine which patient populations derive the greatest survival benefit and best tolerability. These trials should also apply standardised chemotherapy dosing schedules, radiotherapy protocols and toxicity grading systems, while integrating patient-reported outcomes, neurocognitive assessments and long-term follow-up to capture delayed toxicities and survival benefit. Incorporation of translational endpoints evaluating cell cycle dynamics and DNA damage response may further clarify the biological impact of vincristine in alkylator-based regimens.
Collaborative, multicenter randomised designs and international registries will be essential to overcome the limited incidence and slow progression of these tumours. The generation of such high-quality prospective evidence will be critical to confirm the therapeutic contribution of vincristine and to optimise treatment strategies in the molecular era of glioma therapy.
Conclusion
Our meta-analysis suggests that omitting vincristine from the PCV regimen may be associated with improved PFS and OS, while reducing neurotoxicity and treatment discontinuation. However, given that these results derive from a small number of retrospective studies with heterogeneous designs and limited molecular characterisation, they should be interpreted with caution. The observed associations do not establish causality and should be considered hypothesis-generating. Future RCTs, ideally molecularly stratified according to the WHO 2021 classification, are essential to confirm whether vincristine provides any additional therapeutic benefit within combination chemotherapy in glioma treatment.
List of abbreviations
CCNU, lomustine; CI, confidence interval; DNA, deoxyribonucleic acid; DSB, double-strand break; HR, hazard ratio; ICL, interstrand crosslink; IDH, isocitrate dehydrogenase; KPS, Karnofsky performance status; MGMT, O⁶-Methylguanine-DNA Methyltransferase; MMR, mismatch repair; O⁶-MeG, O⁶-Methylguanine; OS, overall survival; PC, procarbazine and lomustine; PCV, procarbazine, lomustine and vincristine; PFS, progression-free survival; PRISMA, preferred reporting items for systematic reviews and meta-analyses; RCT, randomised controlled trial; RR, relative risk; S phase, synthesis phase; TMZ, temozolomide; WHO, World Health Organisation.
Acknowledgment
The authors express their sincere gratitude to their families and mentors for their unwavering support, encouragement, and guidance throughout the development of this work. Special acknowledgment is extended to Pavel PR, Luis CJ, Santos CC, Carmen CS, and Pamela ZM, for their valuable contributions, insightful feedback, and continuous support that significantly enriched this study.
Conflicts of interest
The authors declare that they have no competing interests. No financial, institutional or personal relationships influenced the design, conduct, analysis or reporting of this review.
Funding
This study did not receive funding from any public, commercial or non-profit organisation. All stages of the research, including study design, data collection, analysis, interpretation and manuscript preparation, were conducted without external financial support.
Ethical approval and patient consent
As this study is a systematic review and meta-analysis based solely on previously published studies, neither ethical approval nor informed consent was necessary.
Author contributions
Viviana Pinzon-Ramirez: Conceptualisation, Methodology, Investigation, Data curation, Writing – original draft, Project administration. Luis E. Cueva-Cañola: Conceptualisation, Methodology, Investigation, Software, Validation, Writing – original draft, Formal analysis, Writing – review & editing. Dilmareth E. Natera: Investigation, Resources, Data curation, Writing – original draft. Helder Edgar Aldo-Chávez Olivera: Investigation, Resources, Data curation, Writing – original draft. Oscar Eduardo Camacho-Hernández: Investigation, Resources, Data curation, Writing – original draft. Andrea C. Beltran-De la Fuente: Investigation, Resources, Data curation, Writing – original draft. Sergio Alexis Ramirez-Alvarez: Investigation, Resources, Data curation, Writing – original draft. Einstein Yhair-Gallardo Cubas: Investigation, Resources, Data curation, Writing – original draft. Giomar Vilca Flores: Investigation, Resources, Data curation, Writing – original draft. Mauricio E. Gamez: Writing, Review & editing. Leonardo Rangel Castilla: Writing, Review & editing.
Data transparency statement
The data extraction templates, extracted datasets and analysis codes are available from the authors upon reasonable request.
Reporting guidelines
This review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
References
1. Molinaro AM, Taylor JW, and Wiencke JK, et al (2019) Genetic and molecular epidemiology of adult diffuse glioma Nat Rev Neurol 15(7) 405–417 https://doi.org/10.1038/s41582-019-0220-2 PMID: 31227792 PMCID: 7286557
2. Zhu P, Du XL, and Lu G, et al (2017) Survival benefit of glioblastoma patients after FDA approval of temozolomide concomitant with radiation and bevacizumab: a population-based study Oncotarget 8(27) 44015–44031 https://doi.org/10.18632/oncotarget.17054 PMID: 28467795 PMCID: 5546458
3. Stupp R, Hegi ME, and Mason WP, et al (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial Lancet Oncol 10(5) 459–466 https://doi.org/10.1016/S1470-2045(09)70025-7 PMID: 19269895
4. Cairncross G, Wang M, and Shaw E, et al (2013) Phase III trial of chemoradiotherapy for anaplastic oligodendroglioma: long-term results of RTOG 9402 J Clin Oncol 31(3) 337–343 https://doi.org/10.1200/JCO.2012.43.2674
5. Van Den Bent MJ, Brandes AA, and Taphoorn MJB, et al (2013) Adjuvant procarbazine, lomustine, and vincristine chemotherapy in newly diagnosed anaplastic oligodendroglioma: long-term follow-up of EORTC brain tumor group study 26951 J Clin Oncol 31(3) 344–350 https://doi.org/10.1200/JCO.2012.43.2229
6. Hafazalla K, Sahgal A, and Jaja B, et al (2018) Procarbazine, CCNU and vincristine (PCV) versus temozolomide chemotherapy for patients with low-grade glioma: a systematic review Oncotarget 9(72) 33623–33633
7. Kacimi SEO, Dehais C, and Feuvret L, et al (2025) Survival outcomes associated with first-line procarbazine, CCNU, and vincristine or temozolomide in combination with radiotherapy in IDH-mutant 1p/19q-codeleted grade 3 oligodendroglioma J Clin Oncol 43(3) 329–338 https://doi.org/10.1200/JCO.24.00049
8. Gonzalez-Aguilar A, Reyes-Moreno I, and Peiro-Osuna RP, et al (2018) Radiotherapy plus temozolomide or PCV in patients with anaplastic oligodendroglioma 1p19q codeleted Rev Neurol 67(8) 293–297 PMID: 30289152
9. Brandes AA, Nicolardi L, and Tosoni A, et al (2006) Survival following adjuvant PCV or temozolomide for anaplastic astrocytoma Neuro Oncol 8(3) 253–260 https://doi.org/10.1215/15228517-2006-005 PMID: 16723632 PMCID: 1871946
10. Vesper J, Graf E, and Wille C, et al (2009) Retrospective analysis of treatment outcome in 315 patients with oligodendroglial brain tumors BMC Neurol 9 33 https://doi.org/10.1186/1471-2377-9-33 PMID: 19604414 PMCID: 2719586
11. Webre C, Shonka N, and Smith L, et al (2015) PC or PCV, that is the question: primary anaplastic oligodendroglial tumors treated with procarbazine and CCNU with and without vincristine Anticancer Res 35(10) 5467–5472 PMID: 26408710
12. Wang F, Zhou F, and Kruh GD, et al (2010) Influence of blood-brain barrier efflux pumps on the distribution of vincristine in brain and brain tumors Neuro Oncol 12(10) 1043–1049 https://doi.org/10.1093/neuonc/noq056 PMID: 20501632 PMCID: 3018922
13. Ahn S, Kim Y, and Shin J, et al (2022) Clinical feasibility of modified procarbazine and lomustine chemotherapy without vincristine as a salvage treatment for recurrent adult glioma Oncol Lett 23(4) 114 https://doi.org/10.3892/ol.2022.13234 PMID: 35251345 PMCID: 8850950
14. Qiu Y, Li Y, and Jiang C, et al (2025) Toxicity and efficacy of different target volume delineations of radiation therapy based on the updated Radiation Therapy Oncology Group/National Research Group and European Organization for Research and Treatment of Cancer guidelines in patients with grade 3-4 glioma: a randomized controlled clinical trial Int J Radiat Oncol Biol Phys 121(5) 1168–1181 https://doi.org/10.1016/j.ijrobp.2024.11.094
15. Page MJ, McKenzie JE, and Bossuyt PM, et al (2022) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews Declaración PRISMA 2020: una guía actualizada para la publicación de revisiones sistemáticas Rev Panam Salud Publica 46 112
16. Schiavo JH (2019) PROSPERO: an international register of systematic review protocols Med Ref Serv Q 38(2) 171–180 https://doi.org/10.1080/02763869.2019.1588072 PMID: 31173570
17. Schmidt L, Shokraneh F, and Steinhausen K, et al (2019) Introducing RAPTOR: revMan parsing tool for reviewers Syst Rev 8(1) 151 https://doi.org/10.1186/s13643-019-1070-0 PMID: 31242929 PMCID: 6595567
18. Brush PL, Sherman M, and Lambrechts MJ (2024) Interpreting meta-analyses: a guide to funnel and forest plots Clin Spine Surg 37(1) 40–42 https://doi.org/10.1097/BSD.0000000000001534
19. Calderon Martinez E, Ghattas Hasbun PE, and Salolin Vargas VP, et al (2025) A comprehensive guide to conduct a systematic review and meta-analysis in medical research Med (Baltimore) 104(33) e41868 https://doi.org/10.1097/MD.0000000000041868
20. Sterne JA, Hernán MA, and Reeves BC, et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions BMJ 355 4919 https://doi.org/10.1136/bmj.i4919
21. Ortiz R (2021) Temozolomide: an updated overview of resistance mechanisms, nanotechnology advances and clinical applications Curr Neuropharmacol 19(4) 513–537 PMCID: 8206461
22. Rincon-Torroella J, Rakovec M, and Materi J, et al (2022) Current and future frontiers of molecularly defined oligodendrogliomas Front Oncol 12 934426 https://doi.org/10.3389/fonc.2022.934426 PMID: 35957904 PMCID: 9358027
23. Wick W, Roth P, and Hartmann C, et al (2016) Long-term analysis of the NOA-04 randomized phase III trial of sequential radiochemotherapy of anaplastic glioma with PCV or temozolomide Neuro Oncol 18(11) 1529–1537 PMID: 27370396 PMCID: 5063521
24. Lamba N, Mcavoy M, and Kavouridis VK, et al (2022) Short-term outcomes associated with temozolomide or PCV chemotherapy for 1p/19q-codeleted WHO grade 3 oligodendrogliomas: a national evaluation Neurooncol Pract 9(3) 201–207 PMID: 35601971 PMCID: 9113268
25. Lassman AB, Iwamoto FM, and Cloughesy TF, et al (2011) International retrospective study of over 1000 adults with anaplastic oligodendroglial tumors Neuro Oncol 13(6) 649–659 https://doi.org/10.1093/neuonc/nor040 PMID: 21636710 PMCID: 3107101
26. Vasquez JC, Bindra RS, and Gueble SE. (2025) Novel strategies for improved treatment of O6-methylguanine-DNA methyltransferase promoter-methylated glioma Med Res Arch 13(9) 6944 https://doi.org/10.18103/mra.v13i9.6944
27. Silber JR, Bobola MS, and Blank A, et al (2012) O(6)-methylguanine-DNA methyltransferase in glioma therapy: promise and problems Biochim Biophys Acta 1826(1) 71–82 PMID: 22244911 PMCID: 3340514
28. Calinescu AA and Castro MG (2016) Microtubule targeting agents in glioma Transl Cancer Res 5(Suppl 1) S54–S60 https://doi.org/10.21037/tcr.2016.06.12 PMID: 30680290 PMCID: 6342473
29. Kaina B (2023) Temozolomide, procarbazine and nitrosoureas in the therapy of malignant gliomas: update of mechanisms, drug resistance and therapeutic implications J Clin Med 12(23) 7442 https://doi.org/10.3390/jcm12237442 PMID: 38068493 PMCID: 10707404
30. Noonan EM, Shah D, and Yaffe MB, et al (2012) O6-Methylguanine DNA lesions induce an intra-S-phase arrest from which cells exit into apoptosis governed by early and late multi-pathway signaling network activation Integr Biol Quant Biosciences From Nano To Macro 4(10) 1237 https://doi.org/10.1039/c2ib20091k
31. Kellie SJ, Barbaric D, and Koopmans P, et al (2002) Cerebrospinal fluid concentrations of vincristine after bolus intravenous dosing: a surrogate marker of brain penetration Cancer 94(6) 1815–1820 https://doi.org/10.1002/cncr.10397 PMID: 11920545
32. Chen Z, Wang J, and He T, et al (2025) Vincristine exerts antiglioma effects by inhibiting the PI3K/AKT signaling pathway: a mechanistic study based on network pharmacology, bioinformatics analysis, and experimental validation Naunyn Schmiedebergs Arch Pharmacol 398(6) 7107–7120 https://doi.org/10.1007/s00210-024-03693-5
33. Ahmed HS, D’Souza L, and Suhas D, et al (2026) Vincristine-induced neurotoxicity in acute lymphoblastic leukemia: a comprehensive systematic review Indian J Hematology Blood Transfusion 42(1) 11–49 https://doi.org/10.1007/s12288-025-02047-2
34. Gomez-Deza J, Slavutsky AL, and Nebiyou M, et al (2023) Local production of reactive oxygen species drives vincristine-induced axon degeneration Cell Death Dis 14(12) 807 https://doi.org/10.1038/s41419-023-06227-8 PMID: 38065950 PMCID: 10709426
35. Triarico S, Romano A, and Attinà G, et al (2021) Vincristine-induced peripheral neuropathy (VIPN) in pediatric tumors: mechanisms, risk factors, strategies of prevention and treatment Int J Mol Sci 22(8) 4112 https://doi.org/10.3390/ijms22084112 PMID: 33923421 PMCID: 8073828
36. Fernandes RT, Teixeira GR, and Mamere EC, et al (2023) The 2021 World Health Organization classification of gliomas: an imaging approach Radiol Bras 56(3) 157–161 PMID: 37564083 PMCID: 10411771
37. Wen PY, Van Den Bent M, and Youssef G, et al (2023) RANO 2.0: update to the response assessment in neuro-oncology criteria for high- and low-grade gliomas in adults J Clin Oncol 41(33) 5187–5199 https://doi.org/10.1200/JCO.23.01059 PMID: 37774317 PMCID: 10860967
38. Vaz-Salgado MA, García BC, and Pérez IF, et al (2024) SEOM-GEINO clinical guidelines for grade 2 gliomas (2023) Clin Transl Oncol 26(11) 2856–2865 https://doi.org/10.1007/s12094-024-03456-x PMID: 38662171 PMCID: 11467015
39. Lamb YN (2024) Vorasidenib: first approval Drugs 84(10) 1325–1331 https://doi.org/10.1007/s40265-024-02097-2 PMID: 39375303 PMCID: 11628568
40. Kazda T, Dziacky A, and Burkon P, et al (2018) Radiotherapy of glioblastoma 15 years after the landmark Stupp’s trial: more controversies than standards? Radiol Oncol 52(2) 121–128w https://doi.org/10.2478/raon-2018-0023 PMID: 30018514 PMCID: 6043880
Supplymentry Tables
Table S1. Search strategy.

Table S2. Treatment regimen description.






