Breast cancer clinical trials in low- and middle-income countries (LMICs)
Adnan-Mustafiz Chowdhury1, Sattam A Halaseh1, Debora Joseph1, Abel I Abel1, Nikita Jha1, Amro Al-Karadsheh2, Hakam Al-Karadsheh3, Omar A Salem4, Sama F Salaitah5, Noor M Arman6 and Georges Rizkallah7
1General Surgery Department, Dorset County Hospital NHS Foundation Trust, Williams Avenue, Dorchester, Dorset DT1 2JY, UK
2University Hospitals Sussex NHS Foundation Trust, Worthing Hospital, Worthing, West Sussex BN11 2DH, UK
3United Lincolnshire Hospitals NHS Trust, Lincoln County Hospital, Lincoln, Lincolnshire LN2 4AX, UK
4East Sussex Healthcare NHS Trust, Conquest Hospital, St Leonards-on-Sea, East Sussex TN37 7PT, UK
5The Hashemite University, Damascus Hwy, Zarqa 13133, Jordan
6University of Jordan, Amman 11942, Jordan
7Salisbury District Hospital, Salisbury, Wiltshire, Salisbury SP2 8BJ, UK
Abstract
Background: Breast cancer is the most diagnosed malignancy worldwide and a leading cause of cancer-related mortality in women. Clinical trials underpin advances in prevention, diagnosis and treatment; however, the landscape of breast cancer trials in low- and middle-income countries (LMICs) remains insufficiently characterised. This study aimed to describe the scope, design, interventions and funding of breast cancer clinical trials conducted in LMICs between January 2010 and December 2020.
Methods: We searched the Cochrane Central Register of Controlled Trials on 15 November 2023 for trials using the keywords ‘breast cancer’, ‘breast adenocarcinoma’ and ‘breast tumour’. Trials were classified by World Bank income category. Extracted variables included study design, primary purpose, time perspective, observational model, trial phase, intervention type, sample size, status, reason for termination, funding source and centre type. Descriptive statistics summarised trial characteristics.
Results: A total of 333 breast cancer trials were identified; Two trials (0.6%) were conducted in low-income countries and 331 (99.4%) in lower-middle-income countries; no trials conducted exclusively in upper-middle-income countries were identified within the registry during the study period. Most trials were interventional (85.3%), prospective (95.8%) and treatment-focused (71.5%). Drug-based interventions predominated (64.9%). Funding was primarily from pharmaceutical or biotechnology companies (53.2%), followed by academic or institutional (university-based) sources (39.6%). Only 7.5% of trials addressed prevention, screening and supportive care. Multicentre trials accounted for 53.8%, single-centre 46.3%.
Conclusion: Breast cancer trials in LMICs are predominantly interventional, pharmaceutically-funded and concentrated in lower-middle-income settings. There remains a substantial paucity of trials from low-income countries and in non-treatment domains such as prevention, screening and supportive care.
Keywords: breast cancer, clinical trials, low- and middle-income countries, global health disparities, pharmaceutical funding
Correspondence to: Adnan-Mustafiz Chowdhury
Email: adnanchowdhury@live.com
Published: 14/04/2026
Received: 01/09/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Background
Breast cancer is the most commonly diagnosed malignancy worldwide, with an estimated 2.3 million new cases and 670 000 deaths reported in 2020, accounting for nearly one quarter of all cancer diagnoses in women and 16% of cancer-related mortality [1]. While high-income countries (HICs) have witnessed substantial improvements in early detection, systemic therapies and survivorship care, these gains have not been uniformly realised in low- and middle-income countries (LMICs) [2, 3]. In many LMIC settings, patients present with more advanced disease, access to standard-of-care diagnostics and treatments is limited and health systems face significant infrastructural and financial constraints [4]. According to data from GLOBOCAN, although breast cancer incidence is highest in HICs, such as those in North America and Europe, mortality rates are disproportionately elevated in LMICs, particularly in sub-Saharan Africa and parts of South America [4]. This disparity is not only due to differences in risk factors like lifestyle and genetics but also reflects systemic barriers to early detection and effective treatment. Consequently, incidence-to-mortality ratios are significantly lower in LMICs (e.g., between 1.77 and 1.94 in sub-Saharan Africa) compared to 6.39 in Northern Europe and 6.73 in North America [5].
Clinical trials are essential for generating high-quality evidence to inform prevention, diagnosis and treatment strategies in breast cancer care. However, the trial landscape in LMICs remains poorly characterised. Despite the growing disease burden, the number and variety of clinical trials conducted in these regions are limited. Most trials are concentrated in a few upper-middle-income countries and are often funded or led by institutions based in HICs [6].
Mapping the current clinical trial landscape is essential for identifying context-specific research gaps and informing more equitable global health strategies. Understanding differences in clinical characteristics, such as age at diagnosis, stage of presentation and tumour subtypes, is crucial for improving outcomes in LMICs. Additionally, the focus of ongoing trials, whether on treatment, screening, prevention or supportive care must better reflect local health priorities and system capacities.
Currently, most breast cancer trials in LMICs focus on pharmacological treatments, with limited attention to screening, radiotherapy or supportive care [7]. Research efforts are often hindered by systemic challenges, including inadequate infrastructure, limited access to radiotherapy and constrained funding. Moreover, countries with the highest disease burden frequently host few trials, highlighting a disconnect between research activity and local health needs. Without intervention, LMICs may face a crisis point where the rising cost of treatment and increasing incidence collide with limited access and affordability. To avoid this, the development of affordable, simple and accessible alternatives is needed. Importantly, advancing global equity in breast cancer outcomes will require more robust, evidence-based research that specifically addresses the unique circumstances of LMICs [7].
This study aims to characterise the scope, design, focus of interventions and funding sources of breast cancer clinical trials in LMICs over a ten-year period, providing a foundation for more inclusive and context-appropriate research efforts.
Methods
Search strategy and selection criteria
We performed a registry-based analysis using the Cochrane Central Register of Controlled Trials (CENTRAL) on 15 November 2023. CENTRAL aggregates trial records from multiple sources, including ClinicalTrials.gov and several national and regional registries. WHO International Clinical Trials Registry Platform (ICTRP) was not searched independently.
Search terms included ‘breast cancer’, ‘breast adenocarcinoma’ and ‘breast tumour’. Eligible studies were clinical trials registered between January 2010 and December 2020 that enrolled adult participants with breast cancer and conducted in one or more LMICs, as defined by the World Bank Atlas (July 2023 classification [8]).
Phase 0 and pharmacokinetic-only studies, trials conducted exclusively in HICs and records lacking sufficient methodological information were excluded.
Data extraction and quality control
Data were extracted using a standardised proforma. Variables included country and income classification, study design (interventional or observational), primary purpose, time perspective, intervention type, trial phase, sample size, funding source and centre type. Retrospective studies were defined as those in which data had been collected prior to trial registration and subsequently analysed without prospective follow-up.
Trial completion status and dates were extracted as reported in registry entries at the time of data collection; subsequent updates were not captured.
Statistical analysis
Descriptive statistics were used to summarise trial characteristics. Categorical variables are reported as frequencies and percentages, and continuous variables as medians with interquartile ranges.
Results
Trial distribution
A total of 333 breast cancer clinical trials conducted in LMICs were identified. Two trials (0.6%) were conducted in low-income countries and 331 (99.4%) in LMICs (Table 1); No trials conducted exclusively in upper-middle-income countries were identified within the registry during the study period, which may reflect registry limitations or income classification at the time of trial registration.
Table 1. Trial distribution by income category. Number and proportion of breast cancer clinical trials conducted in low-income and lower-middle-income countries between 2010 and 2020.

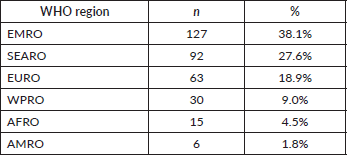
When grouped by WHO region, most trials were conducted in the Eastern Mediterranean (38.1%) and South-East Asia (27.6%) regions, with relatively few studies from Africa (4.5%) and the Americas (1.8%) (Table 2).
Table 2. Trial distribution by WHO region. Number and proportion of breast cancer clinical trials conducted in low- and middle-income countries between 2010 and 2020, stratified by World Health Organisation (WHO) region.
Study design and objectives
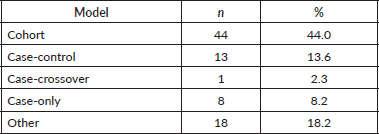
Most trials were interventional (85.3%), with observational studies accounting for 14.7% (Table 3). Treatment was the primary objective in 71.5% of trials, followed by supportive care (6.6%), prevention (6.0%) and screening (1.5%). The majority employed a prospective design (95.8%), with retrospective (1.5%) and cross-sectional (1.8%) designs less common. Among observational studies, cohort designs (44%) were the most common, followed by case-control (13.6%) and other models (Table 4).
Table 3. Study design and objectives. Characteristics of included trials according to study type, primary objective and time perspective.
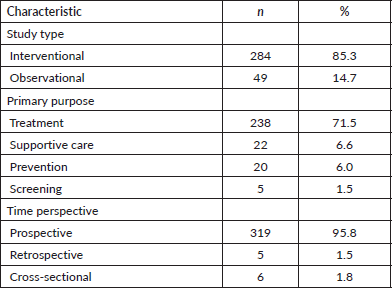
Table 4. Observational models. Distribution of observational study designs among included breast cancer clinical trials.
Intervention types
Drug-based interventions predominated, accounting for 64.9% of trials. Other interventions included radiotherapy (4.8%), combined treatment modalities (3.0%), procedures, diagnostic tests, behavioural interventions and devices (Table 5).
Table 5. Intervention types. Distribution of breast cancer clinical trials in low- and middle-income countries by type of intervention.
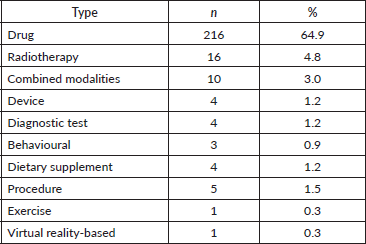
Among drug-focused trials (n = 216), 176 (81.4%) evaluated systemic anticancer therapies, including cytotoxic chemotherapy, endocrine agents and targeted treatments, either alone or in combination.
Sample size
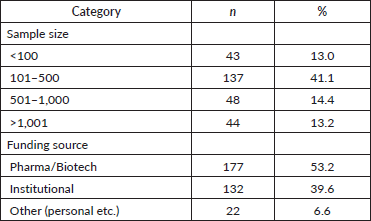
Over half of trials (54.1%) planned to recruit fewer than 500 participants (<100 in 13.0%, 101–500 in 41.1%) (Table 6).
Table 6. Sample size and funding. Distribution of included trials by planned sample size and reported funding source.
Funding, collaboration and completion status
Funding was primarily provided by pharmaceutical or biotechnology companies (53.2%), followed by academic or institutional (university-based) sources (39.6%) (Table 6). Other funding sources accounted for 6.6%.
Multicentre trials comprised 53.8% of studies (Table 7). Where collaboration data were available (n = 176), most multicentre trials involved partnerships spanning LMICs, UMICs and HICs (77.7%), while exclusively LMIC-based collaborations were uncommon (6.3%).
Table 7. Centre type. Distribution of included trials by centre type, comparing single-centre and multicentre studies.

Discussion
This analysis demonstrates that breast cancer clinical trial activity in LMICs between 2010 and 2020 was heavily concentrated in lower-middle-income countries (99.4%), with minimal representation from low-income settings (0.6%). Across the study period, trials were predominantly interventional (85.3%), prospective (95.8%) and randomised (72.5%), and largely focused on drug-based therapeutic interventions (64.9%).
The dominance of systemic therapies, including chemotherapy backbones, endocrine agents and targeted treatments, reflects a research agenda driven largely by pharmaceutical and biotechnology funding, which accounted for over half of all trials. In contrast, prevention, screening and supportive care were markedly under-represented, together comprising fewer than one-fifth of studies. Supportive care research, in particular, was sparse and largely observational, despite its potential to improve quality of life, treatment adherence and outcomes in resource-constrained settings.
These findings mirror observations from other malignancies in LMICs, including prostate cancer, where clinical trial activity is similarly skewed towards industry-sponsored, treatment-oriented studies conducted predominantly in middle-income countries [9]. Given that breast cancer carries a substantial burden in many LMICs – often presenting at more advanced stages and with distinct biological subtypes – the paucity of prevention, early detection and supportive-care research is particularly concerning. Evidence generated in HICs may not generalise to these contexts owing to differences in resource availability, treatment access, tumour genomics and patient comorbidities.
Several structural barriers likely contribute to these patterns. The predominance of pharmaceutical funding highlights the reliance on commercial interests to drive research in LMICs, which may bias trial design towards novel therapeutics rather than public-health interventions. The high costs of oncology trials frequently exceed the annual per-capita gross national income in many LMICs, constituting a formidable barrier to locally led research [10]. Moreover, regulatory complexity, limited research infrastructure and competing clinical duties further impede trial initiation and completion in resource-constrained settings. In addition, challenges in participant recruitment – including limited public awareness of clinical research and socioeconomic barriers to enrolment – may further hinder trial initiation and completion in these settings [11].
To address these gaps, a multifaceted approach is essential. International and local stakeholders must invest in research infrastructure, streamline regulatory pathways and foster equitable partnerships that prioritise capacity-building and local leadership. Diversification of funding – through governmental and philanthropic channels – would support studies of screening, prevention and supportive care. Strengthening training in research methodology and trial management for clinicians and trainees in LMICs is also critical. Finally, journals and professional societies should continue efforts to reduce publication barriers and promote the visibility of high-quality research from under-represented regions.
By broadening the scope of breast cancer clinical trials in LMICs and ensuring that research agendas align with local needs, the global oncology community can generate the context-relevant evidence necessary to improve outcomes and reduce disparities worldwide.
Limitations
This study has several limitations. Reliance on CENTRAL without independent searching of WHO ICTRP or regional trial registries may have resulted in missed studies, particularly from upper-middle-income countries. Registry data did not permit systematic assessment of publication outcomes, outcome direction or the depth of international collaboration. Finally, trial characteristics were analysed as reported at the time of data extraction and may have changed subsequently.
Conclusion
Breast cancer clinical trials in LMICs are predominantly interventional, pharmaceutically funded, concentrated in lower-middle-income countries, with notable gaps in low-income settings and non-treatment research domains. Strengthening research capacity, diversifying funding sources and fostering equipments.
International collaborations are essential to align future trials with local health priorities and reduce global disparities in breast cancer outcomes.
Author contributions
Adnan-Mustafiz Chowdhury: Conceptualisation, Methodology, Formal Analysis, Writing – Original Draft Preparation, Writing – Original Draft Preparation, Writing – Review & Editing, Project Administration
Sattam A. Halaseh: Conceptualisation, Methodology, Formal Analysis, Writing – Original Draft Preparation, Writing – Original Draft Preparation, Writing – Review & Editing, Project Administration
Debora Joseph: Literature Review, Writing – Review & Editing
Abel I. Abel: Writing – Review & Editing
Nikita Jha: Writing – Review & Editing
Amro Al-karadsheh: Data Collection, Writing – Review & Editing
Hakam Al-karadsheh: Data Collection, Writing – Review & Editing
Omar A. Salem: Data Collection, Writing – Review & Editing
Noor M. Arman: Data Collection, Writing – Review & Editing
Georges Rizkallah: Data Collection, Writing – Review & Editing
Conflicts of interest
The authors declare no competing interests.
Funding
No external funding was received.
References
1. Arnold M, Morgan E, and Rumgay H, et al (2022) Current and future burden of breast cancer: global statistics for 2020 and 2040 Breast 66 15–23 https://doi.org/10.1016/j.breast.2022.08.010 PMID: 36084384 PMCID: 9465273
2. Berry DA, Cronin KA, and Plevritis SK, et al (2005) Effect of screening and adjuvant therapy on mortality from breast cancer N Engl J Med 353(17) 1784–1792 https://doi.org/10.1056/NEJMoa050518 PMID: 16251534
3. Coleman MP, Quaresma M, and Berrino F, et al (2008) Cancer survival in five continents: a worldwide population-based study (CONCORD) Lancet Oncol 9(8) 730–756 https://doi.org/10.1016/S1470-2045(08)70179-7 PMID: 18639491
4. Jedy-Agba E, Mccormack V, and Adebamowo C, et al (2016) Stage at diagnosis of breast cancer in sub-Saharan Africa: a systematic review and meta-analysis Lancet Glob Health 4(12) e923–e935 https://doi.org/10.1016/S2214-109X(16)30259-5 PMID: 27855871 PMCID: 5708541
5. Bray F, Ferlay J, and Soerjomataram I, et al (2018) Global cancer statistics 2018: gLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries CA Cancer J Clin 68(6) 394–424 https://doi.org/10.3322/caac.21492 PMID: 30207593
6. Tolentino-Rodriguez L, Chkeir M, and Pofagi V, et al (2024) Breast cancer characteristics in low- and middle-income countries: an umbrella review Cancer Epidemiol 96 102797 https://doi.org/10.1016/j.canep.2025.102797
7. Lynch-Kelly K, Singer M, and Williams NR (2017) The landscape of randomised controlled trials of therapies in breast cancer in low- and middle-income countries Int J Breast Cancer 2017 4259704 https://doi.org/10.1155/2017/4259704
8. World Bank (2025) World Bank country and lending groups [Internet] Washington (DC): World Bank; [cited 2025 Aug 31] [https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups]
9. Halaseh SA, Al-Karadsheh A, Mukherji D, et al (2023) Prostate cancer clinical trials in low- and middle-income countries Ecancermedicalscience 17 1629 https://doi.org/10.3332/ecancer.2023.1629
10. Monge C, Eldridge L, and Pearlman PC, et al (2025) Global perspectives on patient-centered outcomes: advancing patient-centered cancer clinical trials globally JNCI Monogr 68(68) 35–41 https://doi.org/10.1093/jncimonographs/lgae043
11. Ahram M, Farkouh A, and Haddad M, et al (2020) Knowledge of, attitudes to and participation in clinical trials in Jordan: a population-based survey East Mediterr Health J 26(5) 539–546 [10.26719/2020.26.5.539] https://doi.org/10.26719/2020.26.5.539 PMID: 32538447





