Associations between immune-suppressive and stimulating drugs and novel COVID-19—a systematic review of current evidence
Beth Russell1,*, Charlotte Moss1,*, Gincy George1,*, Aida Santaolalla1,*, Andrew Cope2,3, Sophie Papa2,4,** and Mieke Van Hemelrijck1,**
1Translational Oncology and Urology Research, King’s College London, London, UK
2Guy’s and St. Thomas NHS Foundation Trust, London, UK
3Centre for Rheumatic Diseases, King’s College London, London, UK
4School of Cancer and Pharmaceutical Sciences, King’s College London, London, UK
*All authors contributed equally.
**Both senior authors contributed equally.
Abstract
Background: Cancer and transplant patients with COVID-19 have a higher risk of developing severe and even fatal respiratory diseases, especially as they may be treated with immune-suppressive or immune-stimulating drugs. This review focuses on the effects of these drugs on host immunity against COVID-19.
Methods: Using Ovid MEDLINE, we reviewed current evidence for immune-suppressing or -stimulating drugs: cytotoxic chemotherapy, low-dose steroids, tumour necrosis factorα (TNFα) blockers, interlukin-6 (IL-6) blockade, Janus kinase (JAK) inhibitors, IL-1 blockade, mycophenolate, tacrolimus, anti-CD20 and CTLA4-Ig.
Results: 89 studies were included. Cytotoxic chemotherapy has been shown to be a specific inhibitor for severe acute respiratory syndrome coronavirus in in vitro studies, but no specific studies exist as of yet for COVID-19. No conclusive evidence for or against the use of non-steroidal anti-inflammatory drugs (NSAIDs) in the treatment of COVID-19 patients is available, nor is there evidence indicating that TNFα blockade is harmful to patients in the context of COVID-19. COVID-19 has been observed to induce a pro-inflammatory cytokine generation and secretion of cytokines, such as IL-6, but there is no evidence of the beneficial impact of IL-6 inhibitors on the modulation of COVID-19. Although there are potential targets in the JAK-STAT pathway that can be manipulated in treatment for coronaviruses and it is evident that IL-1 is elevated in patients with a coronavirus, there is currently no evidence for a role of these drugs in treatment of COVID-19.
Conclusion: The COVID-19 pandemic has led to challenging decision-making about treatment of critically unwell patients. Low-dose prednisolone and tacrolimus may have beneficial impacts on COVID-19. The mycophenolate mofetil picture is less clear, with conflicting data from pre-clinical studies. There is no definitive evidence that specific cytotoxic drugs, low-dose methotrexate for auto-immune disease, NSAIDs, JAK kinase inhibitors or anti-TNFα agents are contraindicated. There is clear evidence that IL-6 peak levels are associated with severity of pulmonary complications.
Keywords: immune modulation, immune suppression, cancer, COVID-19, adverse events
Correspondence to: Mieke Van Hemelrijck
Email: mieke.vanhemelrijck@kcl.ac.uk
Published: 27/03/2020
Received: 20/03/2020
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Since the outbreak of severe acute respiratory syndrome (SARS)-CoV-2 or COVID-19 coronavirus started in China in December 2019, there is increasing evidence that those with existing comorbidities, older age or a compromised immune system are at higher risk of developing severe and even fatal respiratory diseases [1]. Cancer and transplant patients are also considered to be in this risk group [2, 3], especially as they may be treated with immune-suppressive or immune-stimulating drugs.
The current review focuses on the effects of immune-suppressive or immune-stimulating drugs on the host immunity against COVID-19. Here, we report a short introduction to each drug, followed by a summary of the results from the identified studies for each relative drug.
We hypothesise that the drugs selected will likely be categorised into one of two groups: 1) those that may be harmful for patients and put them at risk of increased morbidity/mortality associated with COVID-19 infection and 2) those that may be used to treat the immunopathology associated with severe persistent viral infection. The clinical impact of this review is, thus, twofold. It aims to identify which drugs clinicians should be thinking about taking patients off to protect them from increased harm from COVID-19 and also which drugs could be potentially beneficial in the fight against the disease.
This review covers the information available today. As the COVID-19 pandemic progresses, there is an opportunity and responsibility to collect prospective data using established randomised controlled trial involving drugs of interest and cohort-based translational studies.
1) All cytotoxic chemotherapy
2) Low-dose steroids and non-steroidal anti-inflammatory drugs (NSAIDs)
3) Any tumour necrosis factor (TNF) blocker
4) interlukin-6 (IL-6) blockade
5) Janus kinase (JAK) inhibitors
6) IL-1 blockade
7) Mycophenolate
8) Tacrolimus
9) Anti-CD20
10) CTLA4-Ig
Methods
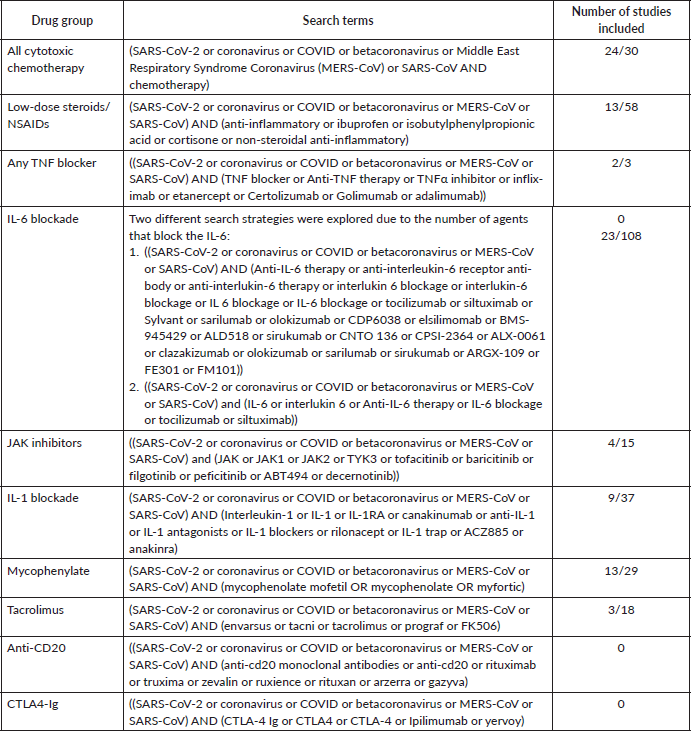
A search was conducted using Ovid MEDLINE and a total of 89 studies were included in this review for 10 different types of immune-suppressing or immune-stimulating drug groups. Table 1 shows the search terms and the number of studies included in the review.
Table 1. Search terms and the number of studies included for each investigated drug group.
Results
All cytotoxic chemotherapy
Cytotoxic chemotherapy inhibits cell division through multiple mechanisms. It may have therapeutic activity as compounds against corona viral strains. Moreover, cancer patients who have undergone chemotherapy may be at increased risk of developing symptoms of SARS.
Results
The search yielded 24 results, of which 18 were included (Supplementary Table 1). Below are key case-reports of interest to highlight:
• Coronavirus was one of the most common viral pathogens identified in paediatric cancer patients undergoing chemotherapy, second to human rhinovirus [4]. Viral co-detection was frequent in patients with cancer and acute respiratory infections.
• A brain biopsy was HCoV-OC43-positive by metagenomic next-generation sequencing in the case of a 1-year-old child with pre-B acute lymphoblastic leukaemia [5].
• A case of a young woman with stage IIIA breast cancer from 1999 reported that diagnosis of coronavirus following treatment for cancer using a high-dose chemotherapy regimen and autologous bone marrow and stem cell transplantation. The electron microscopy revealed coronavirus pneumonia [6]. Coronavirus should be potentially considered in the differential diagnoses of respiratory failure in patients who have undergone high-dose chemotherapy and autologous bone marrow transplantation.
In addition, some further observations have been made in relation to specific treatments:
• Although HIV protease inhibitors such as lopinavir has been suggested as a low-micromolar inhibitor of MERS-CoV, the different mechanistic classes that HIV and coronavirus fall under meant that there was low affinity for coronavirus strains compared to HIV [7].
• The following compounds have been shown to be active in vitro against the SARS-CoV virus: TNFα-converting enzyme inhibitor (TAPI-2); IFN-α (B/D, mDEF201 by adenovirus 5 vector, CR3014 humanized monoclonal antibody (a neutralising antibody specific for SARS-CoV), recombinant IFN-α2b and type I IFN-β); Interferon inducers (Ampligen and polyinosinic–polycytidylic); therapeutic antibodies (2978/10, equine anti-SARS-CoV F[ab’] and monoclonal antibody 201); attachment inhibitors (Urtica Dioica lectin and griffithsin); host immune system [8].
• 6-mercaptopurine (6MP) and 6-thioguanine (6TG) have been used in cancer chemotherapy for treatment of acute lymphoblastic or myeloblastic leukaemia and were found to be specific inhibitors for the SARS coronavirus [9].
• Carbohydrate-binding agents (CBA) may be able to block enveloped viruses other than HIV in their entry process and coronaviruses and influenza viruses are other examples of enveloped viruses that may be highly susceptible to the antiviral action of CBAs [10].
• The genome of SARS-CoV encodes five major proteins: the spike protein (S), the envelope protein (E), the membrane glycoprotein (M), and the nucleocapsid protein (N). M and E may help host cells to induce the production of protective IFN-α to fight against the virus. Bananin 1-[3-hydroxy-5-(hydroxymethyl)-2-methyl-4-pyridinyl]-2,8,9-trioxaadamantane-3,5,7-triol acts as zinc (Zn2 ) chelator and is therefore of interest to target and inhibit immunodeficiency virus type 1 (HIV-1) zinc finger HIV-1 RNA-binding nucleocapsid protein p7 (NCp7). Bananin is converted to bananin 5’-monophosphate (BNP) which together with B6RA (vitamin A-vitamin B6 conjugate) and could inhibit infectious virion encapsidation. Targets of BNP and B6RA has shown to be present also in SARS-associated coronavirus making them possible therapeutic candidates [11].
Conclusion
Coronavirus strains were one of the most common viral pathogens identified in paediatric cancer patients undergoing chemotherapy. Patients with pre-B acute lymphoblastic leukaemia and breast cancer who have undergone chemotherapy have reported cases of coronavirus infection. Cytotoxic therapies used in cancer chemotherapy such as 6MP and 6TG have shown to be specific inhibitors for SARS coronavirus in in vitro studies. However, further in vitro and in vivo studies are required to confirm this, especially in COVID-19. Currently, there is no scientific evidence of the interaction between methotrexate and COVID-19.
Low-dose steroids and NSAIDs
Since the outbreak of the novel COVID-19 infection, various contradictory information has been circulated regarding the potentially negative effect of treating patients with NSAIDs, non-NSAIDs and corticosteroids. NSAIDs work through inhibition of the cyclooxygenase enzymes (COX-1/COX-2), which are involved in the synthesis of key biological mediators. These mediators in turn control inflammation. Corticosteroids are involved in a number of key physiological processes including the immune response and inflammation and low-dose steroids are often prescribed to cancer patients with suppressed immune systems to prevent the development of related auto-immune diseases.
Results
A total of 58 studies were identified from the search terms, of which 13 were deemed suitable for inclusion (Supplementary Table 2). Our search did not identify any strong evidence for or against the use of ibuprofen for treatment of COVID-19 specifically. One study did however link SARS-CoV to the downregulation of angiotensin converting enzyme-2 (ACE2) which is upregulated by ibuprofen [12]. The authors of this study were investigating the link between the severity of COVID-19 symptoms in patients with asthma and hypertension.
The only other study to investigate a non-steroidal anti-inflammatory drug was one which looked at indomethacin [13]. This study suggested that indomethacin exhibited potent antiviral activity against canine coronavirus (dramatically inhibiting virus replication and protecting the host cell from virus-induced damage). This activity was also observed against human SARS-CoV at a concentration dose of 1 mg/kg.
In general, there appeared to be a few positive results for the use of corticosteroids in viral infections such as SARS-CoV [14-19]. Corticosteroids were widely used during the SARS-CoV outbreak due to their known ability to modulate a variety of involved cytokines (including IL-1, IL-6, IL-8, IL-12 and TNFα) [14, 16]. Various studies in humans noted that corticosteroids appeared effective in reducing immunopathological damage but concerns centred around the promotion of viral rebound and association with adverse events (including acute respiratory distress syndrome) [14]. One laboratory study which treated porcine respiratory coronavirus infected pigs with dexamethasone suggested that one or two doses of the corticosteroid in the acute phase of infection may effectively alleviate early pro-inflammatory response, but prolonged administration may play a role in enhancing viral replication [18]. A separate Chinese study, which separated SARS-CoV patients into 4 treatment groups, identified early high-dose steroids in combination with a quinolone as producing the best patient outcomes [19]. Nevertheless, one review stated that the WHO does not currently recommend corticosteroids in other viral diseases such as Dengue as the ‘glucocorticoid-mediated stimulation of the hypothalamic-pituitaryadrenal axis can also drive lymphocytopenia, or it may promote exaggerated pro-inflammatory responses that eventually cause a worsening of the pathogenic condition’ [20].
Conclusion
The current literature does not give conclusive evidence for or against the use of NSAIDs in the treatment of COVID-19 patients, though there appears to be some evidence that corticosteroids may be beneficial in the treatment of SARS-CoV. However, it is important to note this is not specific to COVID-19.
TNFa blocker
TNF family of receptors and cytokines is very large and are often the targets for drugs. One example are TNFa inhibitors, which act by supressing the physiologic response to TNFa. TNFa is a pro-inflammatory cytokine involved in autoimmune and immune-mediated disorders such as rheumatoid arthritis, ankylosing spondylitis, inflammatory bowel disease, psoriasis, hidradenitis suppurativa and refractory asthma. Inhibitors of TNFa may be used in their treatment.
Inhibition of TNFa can be achieved with a monoclonal antibody, such as infliximab (Remicade), adalimumab (Humira), certolizumab pegol (Cimzia) and golimumab (Simponi), or with the receptor fusion protein etanercept (Enbrel).
Results
A total of three studies were identified, of which two were deemed suitable for inclusion (Supplementary Table 3). The first study was a research letter that suggested that TNFa has been implicated in the severe immune-based pulmonary injury caused by SARS coronavirus, suggesting that TNFa inhibitors could be a potential treatment for the acute respiratory disease syndrome caused by coronavirus [21]
The second study utilised 22 piglets to assess the efficacy of an anti-TNFa)therapy for endotoxin respiratory diseases and observed that TNFa blockade was not associated with decrease in disease severity [22].
Conclusion
Currently, there is no evidence indicating that TNFa blockade is harmful to patients in the context of COVID-19.
IL-6 blockade
IL-6, promptly and transiently produced in response to infections and tissue injuries, contributes to host defence through the stimulation of acute phase responses, hematopoiesis, and immune reactions. Although its expression is strictly controlled by transcriptional and posttranscriptional mechanisms, dysregulated continual synthesis of IL-6 plays a pathological effect on chronic inflammation and autoimmunity [23]. For this reason, tocilizumab, a humanized anti-IL-6 receptor antibody was developed. Other approved anti IL-6 drugs are siltuximab (Sylvant) and sarilumab. Several agents are in clinical trials:olokizumab (CDP6038), elsilimomab, BMS945429(ALD518), sirukumab (CNTO 136), CPSI-2364, ALX-0061, clazakizumab, olokizumab, sarilumab, sirukumab, ARGX-109, FE301 and FM101.
Results
A total of 108 studies were identified from the second search strategy, 23 were suitable for inclusion (Supplementary Table 4). The first search strategy found no hits.
IL6 an actor in the pathogenetic mechanisms of the coronavirus infection
• COVID-19 induces a pro-inflammatory generation and secretion of cytokines including IL-1b and IL-6 via the toll like receptors (TLR) that causes the production of active mature IL-1b which is a mediator of lung inflammation, fever and fibrosis. Anti-inflammatory cytokines, such as IL1-Ra, IL-37 or IL-38 could potentially provide relief in both systemic inflammation and fever occurring after infection [24].
• Cytokine profiles in patients diagnosed with SARS showed marked elevation of T-helper lymphocyte type 1 (Th1) cytokine interferon-gamma (IFN-γ), inflammatory cytokines IL-1β, IL-6 and IL-12 for at least two weeks after disease onset. Children however presented a much milder cytokine and chemokine storm [16].
• The high levels of IL-6 in the acute stage associated with lung lesions found in SARS patients are activated by the viral nucleocapsid SARS-CoV N protein [25].
• Over induction of inflammatory cytokine and dysregulation of cytokine signalling has been observed in patients with SARS in comparison with other respiratory viruses including respiratory syncytial virus (RSV), influenza A virus (FluAV), and human parainfluenza virus type 2 (hPIV2). SARS-CoV and RSV induced high levels of IL-6 and RANTES compared with FluAV and hPIV2 [26].
• The N-protein of SARS-CoV induces pulmonary inflammatory reaction and acute lung injury, which were related to the increase and imbalance of pro-inflammatory and anti-inflammatory cytokines. Glucocorticoids could effectively alleviate the pulmonary inflammatory reaction induced by N-protein of SARS-CoV [15].
• SARS-CoV does not productively infect human macrophages (Mphi) or dentritic cells (DCs), however it modulates a massive release of IL-6 and IL-12 and compromises the endocytic capacity (e.g., antigen capture capture) of Mphi was significantly compromised [27].
• Changes in plasma T helper (Th) cell cytokines, inflammatory cytokines and chemokines in 20 patients diagnosed with SARS were assessed. The elevation of Th1 cytokine IFN-γ, inflammatory cytokines IL-1, IL-6 and IL-12 and chemokines IL-8, MCP-1 and IP-10 confirmed the activation of Th1 cell-mediated immunity and hyper-innate inflammatory response in SARS through the accumulation of monocytes/macrophages and neutrophils [17].
IL-6 as a potential marker of disease severity in coronavirus infected patients
• IL-6 blood measurements seem useful to diagnose severe COVID-19 cases. The findings suggest that IL-6 and D-Dimer level can be used to estimate the severity of COVID-19. The optimum critical point of IL-6 in the group was 24.3 pg/ml, which was the upper limit of no severe pneumonia [28].
• The increased expression of IL-2R and IL-6 in serum is expected to predict the severity of the 2019-nCoV pneumonia and the prognosis of patients [29].
• The serum levels of IL-6 and CXCL-10 were significantly elevated in MERS-CoV patients who developed severe diseases [30].
• A new lethal animal model was characterised for SARS-CoV. Strain v2163 had nine mutations that increased levels of IL-1alpha and IL-6 in mice. The high IL-6 expression was correlated with mortality [31].
• SARS vaccination was tested in a murine SARS model. A high level of IL-6 and on days 2 and 3 after SARS-CoV infection was closely linked to the virus replication and disease severity [32]
• Interleukin-6 (IL-6) and IL-8 are key SARS-CoV-induced epithelial cytokines capable of inhibiting the T-cell-priming ability of dendritic cells, a cellular element of the host innate defenses against respiratory infections, leading to an exacerbated inflammatory cascades and severe tissue damage in SARS patients [33].
• In patients with a diagnosis of SARS-associated coronavirus infection, there were no significant differences in peak levels of IL-6, IL-8 and TNFα between patients with and without acute respiratory distress syndrome. However , CRP and TNFα were associated with worse outcomes and might be used as prognostic markers of SARS [34].
• IFN-gamma, IL-18, TGF-beta, IL-6, IP-10, MCP-1, MIG were highly elevated in the acute phase sera of Taiwan SARS patients, being IL-18, IP-10, MIG, and MCP-1 were significantly higher in the death group than in the survival group. It suggests that an interferon-gamma-related cytokine storm was induced post SARS coronavirus infection [35].
• Eight patients with SARS [36] were treated with ribavirin, which was not effective in reducing the SARS coronavirus load in three of eight. Elevated levels of interleukin (IL)-6 and IL-8 subsequent to the peak viral load were found in eight and six cases [37].
• A cytokine profiling was performed for 110 serum from healthy donors, patients with SARS, patients with severe SARS, and patients with SARS in convalescence. IL6 concentrations were significantly elevated in severe SARS patients, but the IL-6 concentrations were similar in convalescent patients and control subjects which suggested that IL6 is associated with SARS severity [38].
• The authors of this study set out to study the inflammatory cytokine profile in children with SARS. They found that the plasma concentrations’ key proinflammatory cytokines, including IL-6, were not substantially increased in any of the patients throughout the course of illness. From this, the authors stated that the cytokine results cast doubt on the liberal use of corticosteroids in paediatric SARS patients [39].
Potential targets for therapeutic intervention related to IL6 regulation during infection
• ADAM-17 regulates IL-6 class switching as a mediator between pro- and anti-inflammatory responses to viral antigenic stimuli in Ebola, SARS-CoV and dengue infections in humans. Therefore, ADAM-17 should be considered as a potential target molecule for novel antiviral drug discovery for infections, such as SARS-CoV [20]
• SARS-CoV ssRNA is a new therapeutic target given its capacity to cause acute lung injury in mice with a high mortality rate in vivo experiment suggesting that SARS-CoV specific GU-rich ssRNA plays a very important role in the cytokine storm associated with a dysregulation of the innate immunity [40].
• DUSP1 and p38 MAPK are potential therapeutic targets for coronavirus infectious bronchitis virus, given their capability to reduce the production of an excessive amount of IL-6 and IL-8 in the infected cells [41].
Potential therapeutic agents that inhibit the infection-induced production of IL6
• Pretreatment of primary cultures of human nasal and tracheal epithelial cells with glycopyrronium or formoterol decreased viral RNA levels and/or titers, the expression of the HCoV-229E and the infection-induced production of cytokines, including IL-6, IL-8 and IFN-beta. Treatment of the cells with the CD13 inhibitor 2’2’-dipyridyl decreased viral titers. Pretreatment of the cells with a combination of three drugs (glycopyrronium, formoterol and budesonide) exerted additive inhibitory effects on viral titers and cytokine production [42].
Conclusion
It has been suggested that the pathogenesis of SARS-CoV is mediated by disproportional immune responses and the ability of the virus to circumvent innate immunity. The COVID-19 infection has also been observed to induce pro-inflammatory cytokine generation and secretion of cytokines, such as IL-6, which dysregulates the local inflammatory responses that have been suggested as partially responsible for the devastating acute respiratory distress syndrome.
Moreover, it has been observed that COVID-19 induces high levels of IL-6 for at least 2 weeks after disease onset. Children presented lower levels of cytokine production. IL-6 has been suggested as a potential prognostic marker of COVID-19 disease severity. Different molecules related with the IL6 pathway have been suggested as potential therapeutic targets such as ADAM-17, SARS-CoV ssRNA, DUSP1 and p38 MAPK.
Currently, there is no scientific evidence of the beneficial impact of IL-6 inhibitors in the modulation of the COVID-19 infection. Further understanding of the role of IL-6 reduction will be forthcoming as the pandemic progresses and further clinical data becomes available. In vitro treatment with glycopyrronium, formoterol and budesonide exerted additive inhibitory effects on viral titers and cytokine production human nasal and tracheal epithelial cells.
JAK inhibitors
JAK inhibitors work by inhibiting the activity of one or more of the JAK family of enzymes, including, JAK1, JAK2, JAK3 and TYK3. JAKs interact with signal transducer and activator of transcription proteins (STATs) and the JAK-STAT pathway is central to cellular response to exogenous signals in the immune system. The JAK family of enzymes are responsible for signal transduction and JAK inhibitors play a major role in inhibiting and blocking cytokine release that can contribute to growth of malignant cells in cancer. JAK inhibitors are used in the treatment of cancer and inflammatory diseases such as rheumatoid arthritis [43].
This review focuses on how the JAK-STAT pathway can be manipulated to reduce viral entry and inflammation in patients with coronavirus. The main targets that the review highlighted were baricitinib (JAK inhibitor), IRE1α (an endoplasmic reticulum stress sensor, leading to an increased expression of negative regulators of JAK-STAT SOCS1 and SOCS3) and combination therapies using tylophorine-based compounds with JAK2 inhibitors.
Results
Fifteen studies were yielded from the search of which 4 were deemed suitable for inclusion (Supplementary Table 5).
Baricitinib
Baricitinib (Olumiant) is a JAK inhibitor that was approved by the European Medicines Agency in February 2017 for the treatment of moderate-to-severe active rheumatoid arthritis in adults with an inadequate response to one or more disease-modifying anti-rheumatic drugs [44]. One study suggested the use of Baricitinib to reduce the viral entry and inflammation caused by 2019-nCoV. Most viruses enter cells through receptor-mediated endocytosis. ACE2, a cell-surface protein expressed on cells in the kidney, blood vessels, heart, and alveola type 2 (AT2) cells in the pulmonary epithelia, may be the receptor that 2019-nCoV uses to infect lung cells. The authors of Richardson et al [45] suggest that by inhibiting adaptor associated protein kinase 1 (AAK1) receptor that promotes endocytosis involved in ACE2, Baricitinib may reduce both the viral entry and the inflammation in 2019-nCoV patients.
IRE1α
Inositol-requiring transmembrane kinase/endoribonuclease 1α (IRE1α) is an endoplasmic reticulum stress sensor that leads to increased expression of negative regulators of JAK-STAT, suppressor of cytokine signalling (SOCS)-1 and SOCS-3 [46]. Therefore, IRE1α may be a novel target against coronavirus infection requiring further exploration.
Tylophorine-based compounds
Tylophorine-based compounds are isolated from plants and exert potent anti-coronaviral activities against SARS-CoV and MERS-CoV [47]. Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) activation is a common pro-inflammatory response of host cells to viral infection. Following in vitro analysis, Yang et al [48] suggests the use of a combination therapy for SARS-CoV or MERS-CoV, wherein a tylophorine compound known to target transmissible gastroenteritis virus and a JAK2 inhibitor synergise to block the alternative dominant NF-κB activation mediated by JAK2. Therefore, the combination treatment for the inhibition of coronavirus per se, e.g. viral genome replication, and blocking cellular NF-κB activation by coronaviruses, is a promising approach for the development of anti-coronavirals.
Conclusion
Current studies suggest that although there are potential targets in the JAK-STAT pathway that can be manipulated in the treatment for coronaviruses, they are all in early stages and require further in vitro and in vivo studies to confirm their therapeutic effects.
IL-1 blockade
IL-1 is a pro-inflammatory cytokine and an important mediator of local and systemic inflammation. Excessive IL-1 release during viral infections can cause lung and tissue inflammation, fever and fibrosis. IL-1 suppression has found to be effective in many inflammatory diseases including rheumatoid arthritis [24].
It is well established that an over-expression of interleukin-1 is a hallmark of SARS-CoV infection, probably through activation of transcription factor nuclear factor, activator protein 1 and activating factor 2. In COVID-19 specifically, the virus is thought to bind to TLRs which activate the formation of pro-IL-1 and activation of the inflammasome [24]. This inflammasome activation is important for the regulation of cells of both the innate and adaptive immune system paving the way for specific immune responses. As part of the inflammasome activation, IL1-b is subsequently produced which mediates the inflammation of the lungs, fever and fibrosis thus causing respiratory complications in the infected host.
Results
A total of 37 studies were identified from the search of which 9 were deemed suitable for inclusion (Supplementary Table 6). Many of the studies were in vitro studies and repeatedly demonstrated increased IL-1 levels in patients infected with a coronavirus. One study investigated the levels of various inflammatory cytokines in 29, COVID-19 patients in China and compared the levels between general, severe and critically ill groups [49]. The authors reported no significant differences in IL-1b levels between the three groups of patients. Another study involving 20 consecutive SARS patients admitted to a Hong Kong hospital identified significantly elevated levels of IL-1β within the first 12, 7 and 5 days following onset of infection [17]. Those patients with more severe disease were treated with pulsed methylprednisolone and IL-1β levels returned to normal after 7 days. The seven patients with less severe disease did not receive any dosage of corticosteroids and their cytokine levels returned to normal range levels over the same 7-day time period.
A further Chinese study also identified reduced levels of IL-1β following administration of corticosteroids- suggesting inhibition of pro-inflammatory cytokines such as IL-1 may be a beneficial treatment strategy for treatment of SARS [50]. A third study, which measured serum cytokine levels in four patient groups including controls, patients with SARS, patients with severe SARS and convalescent SARS patients suggested that longer term treatment (over a period of 7-10 days) with low-dose steroids can alter serum cytokine levels, including IL-1α [38].
One rat model showed promising results for an IL-1 receptor antagonist which reduced the chemokine expression in infected animals [51]. However, this result cannot be generalised for humans. Unfortunately, one study, in which the authors state the ‘demonstrate for the first time that inflammation by coronavirus may be inhibited by anti-inflammatory cytokines belonging to the IL-1 family’, was only available as an abstract [52]. Therefore, further evidence or information to back this claim up is not available.
Conclusion
Overall, this review demonstrated that although it is evident that IL-1 is elevated in patients infected with a coronavirus, there is not at present evidence for an established role for IL-1 blockers in the treatment of COVID-19 in humans. The literature did, however, suggest a potential role for low-dose corticosteroids to reduce levels of pro-inflammatory markers, such as IL-1, which are elevated as part of the immune response and may have a role in the severe lung damage associated with human coronaviruses.
Mycophenolate
Mycophenolate mofetil (which is a derivative of mycophenolic acid (MPA)) is an immune suppressant, antineoplastic and antiviral mediation. According to the British National Formulary, mycophenolate mofetil is used for the prophylaxis of acute rejection in renal transplantation and is usually used in combination with a corticosteroid and ciclosporin.
Results
Almost all of the studies investigated MPA as a potential therapy for MERS-CoV due to its anti-viral properties. Six of the 13 selected studies were in vitro studies, two were in vivo, one was a clinical example and four were reviews (therefore there was some overlapping of results) (Supplementary Table 7).
In-vitro studies
In general, the in vitro studies looked positive with MPA targeting the papain like proteases of both MERS-CoV and SARS-CoV [53, 54]. The studies found that MPA showed strong inhibition of the virus with a very low IC50 [55-57].
In vivo study
MPA can been used in combination with interferon-beta (IFN-b). One study, which applied this regime in marmosets exhibiting a severe disease resembling human MERS, reported high viral loads with more severe or even fatal disease [28]. The authors of this study state that MPA is likely to cause more harm than benefit to MERS patients.
Clinical studies
According to the review by Mo and Fisher [58], MPA monotherapy had not been tested in a clinical setting for the treatment of MERS-CoV. Al Ghamdi et al [59], presented an example where eight patients were treated with MPA for MERS-CoV, seven in combination with IFN-β. All eight of these patients survived, however the review by Mo and Fisher stated that this group of patients had lower Acute Physiology and Chronic Health Evaluation II scores compared with others in the cohort who received a variety of antiviral agents including ribavirin and IFN-a, steroids and antibiotics. Therefore, the results must be interpreted with caution.
Conclusion
Whilst the in vitro studies showed promising results for MPA against MERS, the in vivo studies suggest that its use is likely to cause more harm than benefit and hence is not likely to be useful against coronavirus infections. The clinical studies are too small to confirm or deny any beneficial use for MERS-CoV patients.
Tacrolimus
Tacroliumus, also known as fujimycin, envarsus or FK506, is an immunosuppressive drug which is mainly administered after allogeneic organ transplant to lower risk of organ rejection. It’s mechanism of action focusses on inhibition of calcineurin which is involved in the production of IL-2. IL-2 is a cytokine which promotes the development and proliferation of T cells which form a vital component of the human adaptive immune response.
Results
A total of 18 studies were identified from the search terms, of which three were deemed suitable for inclusion (Supplementary Table 8).
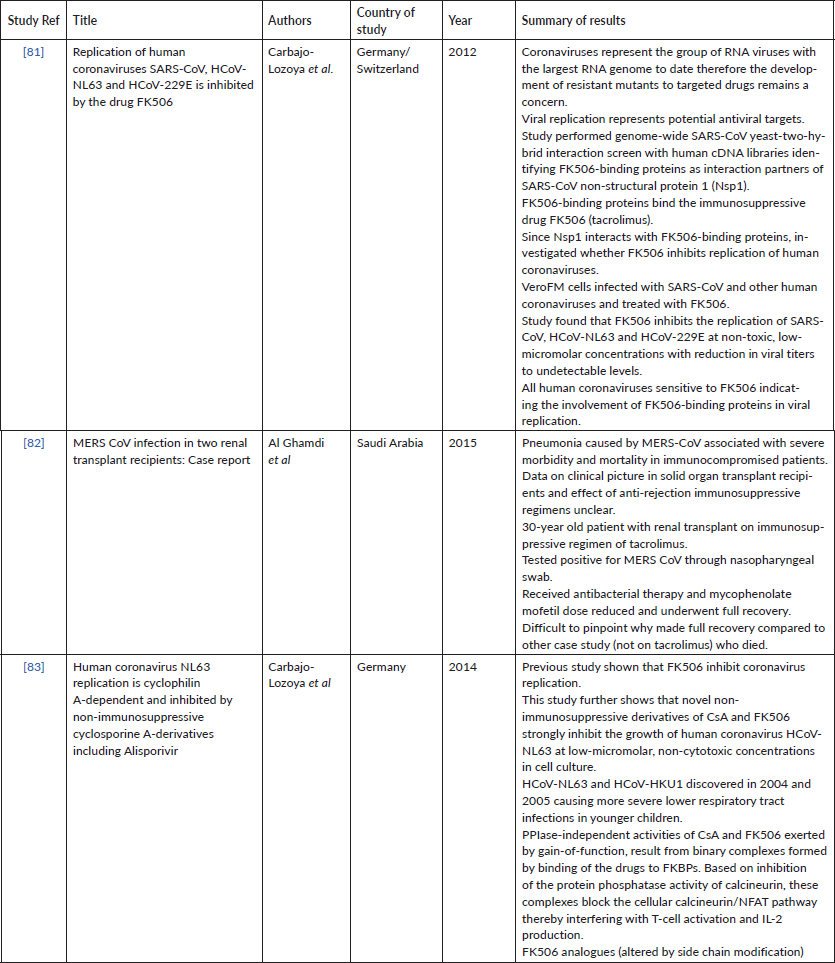
Overall, the literature appeared to suggest a potential role for tacrolimus in the treatment of human coronaviruses. In a case study of two renal transplant recipients who tested positive for MERS CoV, a patient who was being treated with an immunosuppressive regimen of tacrolimus underwent full recovery whilst the other patient (who was not on this treatment regimen) succumbed to the infection [60]. The patient who eventually made a full recovery was also treated with antibacterial therapy and a reduced dose of mycophenolate mofetil and it is therefore not possible to conclude that patient recovery due to tacrolimus.
The two other included studies were both laboratory studies involving cell line culture. The first investigated pathways of coronavirus viral replication as potential antiviral therapeutic targets [61]. Genome-wide SARS-CoV yeast-two-hybrid interaction screen with human cDNA libraries identified FK506-binding proteins as interaction partners of SARS-CoV non-structural protein 1. Subsequently, the authors investigated whether tacrolimus inhibits viral replication of human coronaviruses. VeroFM cells infected with SARS-CoV and other human coronaviruses were treated with FK506. Results showed that FK506 effectively inhibited viral replication of SARS-CoV, HCoV-NL63 and HCoV-229E at non-toxic, low-micromolar concentrations with a reduction in viral titers to undetectable levels. The second study further confirmed this inhibition using novel non-immunosuppressive derivatives of FK506 in the context of HCoV-NL63 at low-micromolar, non-cytotoxic concentrations in cell culture [62].
Conclusion
Overall the small amount of literature available suggests a potential role of FK506 (tacrolimus) as a potent antiviral in the treatment of human coronaviruses. It is important to note, however, that COVID-19 is a novel disease and may have different aetiology and mechanistic action compared to existing strains and to date, this immunosuppressive drug and its derivatives has not been tested in humans. Further study is warranted, both in the clinical setting and laboratory.
Anti-CD20
No studies were identified for inclusion.
CTLA-4 Ig
No studies were identified for inclusion.
Overall conclusion
The rapidly progressing SARS-CoV-2 pandemic has led to challenging decision-making about the treatment of critically unwell patients with the novel viral infection. In parallel, doctors across multiple specialties are making clinical decisions about the appropriate continuation of treatments for patients with chronic illnesses requiring immune-suppressive medication. This systematic review looks to provide guidance from the current available literature.
As the COVID-19 pandemic progresses, collective effort to capture data from prospective trials is required. Sponsors of randomized controlled trials recruiting patients randomized to receive immune modulatory drugs that may be affected by COVID-19 should collect data about the disease outcomes and consider interim analysis of potential advantages and disadvantages associated with using one of these medications.
Low-dose prednisolone and tacrolimus therapy may have beneficial impacts on the course of SARS-CoV-2. This observation requires further validation. The mycophenolate mofetil picture is less clear, with conflicting data from pre-clinical studies. There is no definitive evidence that specific cytotoxic drugs, low-dose methotrexate for auto-immune disease, NSAIDs, JAK kinase inhibitors or anti-TNFa biological agents are contraindicated. There is evidence that IL-6 peak levels are associated with severity of pulmonary complications. Ongoing studies of blockade of the IL-6 pathway are rational and will hopefully inform practice as the pandemic progresses.
Funding declaration
We would like to thank our various funders: Guy’s and St Thomas’ Charity, Cancer Research UK (C45074/A26553), and the CRUK King’s Health Partner Centre.
Conflicts of interest
The authors have no conflicts of interest to declare.
References
1. Chen N, Zhou M, and Dong X, et al (2020) Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study Lancet 395(10223) 507–513 https://doi.org/10.1016/S0140-6736(20)30211-7 PMID: 32007143
2. Wang H and Zhang L (2020) Risk of COVID-19 for patients with cancer Lancet Oncol https://doi.org/10.1016/S1470-2045(20)30149-2
3. Xia Y, Jin R, and Zhao J, et al (2020) Risk of COVID-19 for cancer patients Lancet Oncol https://doi.org/10.1016/S1470-2045(20)30150-9 PMID: 32142622
4. Benites EC, Cabrini DP, and Silva AC, et al (2014) Acute respiratory viral infections in pediatric cancer patients undergoing chemotherapy J Pediatr (Rio J) 90(4) 370–376 https://doi.org/10.1016/j.jped.2014.01.006
5. Nilsson A, Edner N, and Albert J, et al (2020) Fatal encephalitis associated with coronavirus OC43 in an immunocompromised child Infect Dis (Lond) 1–4
6. Folz RJ and Elkordy MA (1999) Coronavirus pneumonia following autologous bone marrow transplantation for breast cancer Chest 115(3) 901–905 https://doi.org/10.1378/chest.115.3.901 PMID: 10084516
7. LaFemina RL (2014) Alternative screening approaches for discovery of middle east respiratory syndrome coronavirus inhibitors Antimicrob Agents Chemother 58(8) 4251 https://doi.org/10.1128/AAC.03406-14 PMID: 24867994 PMCID: 4135998
8. Barnard DL and Kumaki Y (2011) Recent developments in anti-severe acute respiratory syndrome coronavirus chemotherapy Future Virol 6(5) 615–631 https://doi.org/10.2217/fvl.11.33 PMID: 21765859 PMCID: 3136164
9. Chen X, Chou CY, and Chang GG (2009) Thiopurine analogue inhibitors of severe acute respiratory syndrome-coronavirus papain-like protease, a deubiquitinating and deISGylating enzyme Antivir Chem Chemother 19(4) 151–156 https://doi.org/10.1177/095632020901900402 PMID: 19374142
10. Balzarini J (2007) Carbohydrate-binding agents: a potential future cornerstone for the chemotherapy of enveloped viruses? Antivir Chem Chemother 18(1) 1–11 https://doi.org/10.1177/095632020701800101 PMID: 17354647
11. Kesel AJ (2003) A system of protein target sequences for anti-RNA-viral chemotherapy by a vitamin B6-derived zinc-chelating trioxa-adamantane-triol Bioorg Med Chem 11(21) 4599–4613 https://doi.org/10.1016/S0968-0896(03)00500-5 PMID: 14527557
12. Fang L, Karakiulakis G, and Roth M (2020) Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med https://doi.org/10.1016/S2213-2600(20)30116-8 PMID: 32171062
13. Amici C, Di Caro A, and Ciucci A, et al (2006) Indomethacin has a potent antiviral activity against SARS coronavirus Antivir therapy 11(8) 1021–1030
14. Chihrin S and Loutfy MR (2005) Overview of antiviral and anti-inflammatory treatment for severe acute respiratory syndrome Expert Rev Anti Infect Ther 3(2) 251–262 https://doi.org/10.1586/14787210.3.2.251 PMID: 15918782
15. Hao D, He L-X, and Qu J-M, et al (2005) [A study of pulmonary inflammatory reaction induced by N-protein of SARS-CoV in rat models and effects of glucocorticoids on it] Zhonghua nei ke za zhi 44(12) 890–893
16. Lam CWK, Chan MHM, and Wong CK (2004) Severe acute respiratory syndrome: clinical and laboratory manifestations Clin Biochem Rev 25(2) 121–132
17. Wong CK, Lam CWK, and Wu AKL, et al (2004) Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome Clin Exp Immunol 136(1) 95–103 https://doi.org/10.1111/j.1365-2249.2004.02415.x PMID: 15030519 PMCID: 1808997
18. Zhang X, Alekseev K, and Jung K, et al (2008) Cytokine responses in porcine respiratory coronavirus-infected pigs treated with corticosteroids as a model for severe acute respiratory syndrome J Virol 82(9) 4420–4428 https://doi.org/10.1128/JVI.02190-07 PMID: 18287230 PMCID: 2293053
19. Zhao Z, Zhang F, and Xu M, et al (2003) Description and clinical treatment of an early outbreak of severe acute respiratory syndrome (SARS) in Guangzhou, PR China J Med Microbiol 52(Pt 8) 715–720 https://doi.org/10.1099/jmm.0.05320-0 PMID: 12867568
20. Mahmud-Al-Rafat A, Majumder A, and Taufiqur Rahman KM, et al (2019) Decoding the enigma of antiviral crisis: does one target molecule regulate all? Cytokine Cytokine 115 13–23 https://doi.org/10.1016/j.cyto.2018.12.008 PMID: 30616034
21. Tobinick E (2004) TNF-alpha inhibition for potential therapeutic modulation of SARS coronavirus infection Curr Med Res Opin 20(1) 39–40 https://doi.org/10.1185/030079903125002757 PMID: 14741070
22. Atanasova K, Van Gucht S, and Van Reeth K (2010) Anti-TNF-alpha therapy does not ameliorate disease in a model of acute virus-endotoxin mediated respiratory disease in pigs Vet Immunol Immunopathol 137(1–2) 12–9 https://doi.org/10.1016/j.vetimm.2010.04.003 PMID: 20466438 PMCID: 2922464
23. Tanaka T, Narazaki M, and Kishimoto T (2014) IL-6 in inflammation, immunity, and disease Cold Spring Harb Perspect Biol 6(10) a016295 https://doi.org/10.1101/cshperspect.a016295 PMID: 25190079 PMCID: 4176007
24. Conti P, Ronconi G, and Caraffa A, et al (2020) Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by COVID-19: anti-inflammatory strategies J Biol Regul Homeost Agents 34(2) pii:1 PMID: 32171193
25. Zhang X, Wu K, and Wang D, et al (2007) Nucleocapsid protein of SARS-CoV activates interleukin-6 expression through cellular transcription factor NF-kappaB Virology 365(2) 324–335 https://doi.org/10.1016/j.virol.2007.04.009 PMID: 17490702
26. Okabayashi T, Kariwa H, and Yokota S-I, et al (2006) Cytokine regulation in SARS coronavirus infection compared to other respiratory virus infections J Med Virol 78(4) 417–424 https://doi.org/10.1002/jmv.20556 PMID: 16482545
27. Tseng C-TK, Perrone LA, and Zhu H, et al (2005) Severe acute respiratory syndrome and the innate immune responses: modulation of effector cell function without productive infection J Immunol 174(12) 7977–7985 https://doi.org/10.4049/jimmunol.174.12.7977 PMID: 15944304
28. Chan JF, Yao Y, and Yeung ML, et al (2015) Treatment with lopinavir/ritonavir or interferon-beta1b improves outcome of MERS-CoV infection in a nonhuman primate model of common marmoset J Infect Dis 212(12) 1904–1913 https://doi.org/10.1093/infdis/jiv392 PMID: 26198719
29. Chen L, Liu HG, and Liu W, et al (2020) [Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia] Zhonghua jie he he hu xi za zhi = Zhonghua jiehe he huxi zazhi = Chin J Tuberc Respir Dis 43(3) 203–208
30. Kim ES, Choe PG, and Park WB, et al (2016) Clinical progression and cytokine profiles of middle east respiratory syndrome coronavirus infection J Korean Medi Sci 31(11) 1717–1725 https://doi.org/10.3346/jkms.2016.31.11.1717
31. Barnard DL, Day CW, and Bailey K, et al (2006) Enhancement of the infectivity of SARS-CoV in BALB/c mice by IMP dehydrogenase inhibitors, including ribavirin Antiviral Res 71(1) 53–63 https://doi.org/10.1016/j.antiviral.2006.03.001 PMID: 16621037
32. Ishii K, Hasegawa H, and Nagata N, et al Neutralizing antibody against severe acute respiratory syndrome (SARS)-coronavirus spike is highly effective for the protection of mice in the murine SARS model Microbiol Immunol 53(2) 75–82 PMID: 19291090
33. Yoshikawa T, Hill T, and Li K, et al (2009) Severe acute respiratory syndrome (SARS) coronavirus-induced lung epithelial cytokines exacerbate SARS pathogenesis by modulating intrinsic functions of monocyte-derived macrophages and dendritic cells J Virol 83(7) 3039–3048 https://doi.org/10.1128/JVI.01792-08 PMCID: 2655569
34. Sheng W-H, Chiang B-L, and Chang S-C, et al (2005) Clinical manifestations and inflammatory cytokine responses in patients with severe acute respiratory syndrome J Formosan Med Assoc = Taiwan yi zhi 104(10) 715–723 PMID: 16385373
35. Huang K-J, Su I-J, and Theron M, et al (2005) An interferon-gamma-related cytokine storm in SARS patients J Med Virol 75(2) 185–194 https://doi.org/10.1002/jmv.20255
36. Wang C, Pang B-S, and National Research Project for Sars BG (2003) [Dynamic changes and the meanings of blood cytokines in severe acute respiratory syndrome] Zhonghua jie he he hu xi za zhi = Zhonghua jiehe he huxi zazhi = Chin J Tuberc Respir Dis 26(10) 586–589
37. Wang W-K, Chen S-Y, and Liu IJ, et al (2004) Temporal relationship of viral load, ribavirin, interleukin (IL)-6, IL-8, and clinical progression in patients with severe acute respiratory syndrome Clin Infect Dis 39(7) 1071–1075 https://doi.org/10.1086/423808 PMID: 15472864
38. Zhang Y, Li J, and Zhan Y, et al (2004) Analysis of serum cytokines in patients with severe acute respiratory syndrome Infect Immun 72(8) 4410–4415 https://doi.org/10.1128/IAI.72.8.4410-4415.2004 PMID: 15271897 PMCID: 470699
39. Ng PC, Lam CWK, and Li AM, et al (2004) Inflammatory cytokine profile in children with severe acute respiratory syndrome Pediatrics 113 e7–e14 https://doi.org/10.1542/peds.113.1.e7 PMID: 14702488
40. Li Y, Chen M, and Cao H, et al (2013) Extraordinary GU-rich single-strand RNA identified from SARS coronavirus contributes an excessive innate immune response Microb Infect 15(2) 88–95 https://doi.org/10.1016/j.micinf.2012.10.008
41. Liao Y, Wang X, and Huang M, et al (2011) Regulation of the p38 mitogen-activated protein kinase and dual-specificity phosphatase 1 feedback loop modulates the induction of interleukin 6 and 8 in cells infected with coronavirus infectious bronchitis virus Virology 420(2) 106–116 https://doi.org/10.1016/j.virol.2011.09.003 PMID: 21959016
42. Yamaya M, Nishimura H, and Deng X, et al (2020) Inhibitory effects of glycopyrronium, formoterol, and budesonide on coronavirus HCoV-229E replication and cytokine production by primary cultures of human nasal and tracheal epithelial cells Respir Investig https://doi.org/10.1016/j.resinv.2019.12.005 PMID: 32094077
43. Kontzias A, Kotlyar A, and Laurence A, et al (2012) Jakinibs: a new class of kinase inhibitors in cancer and autoimmune disease Curr Opin Pharmacol 12(4) 464–470 https://doi.org/10.1016/j.coph.2012.06.008 PMID: 22819198 PMCID: 3419278
44. Agency EM (2019) Olumiant (baricitinib) [https://www.ema.europa.eu/en/medicines/human/EPAR/olumiant]
45. Richardson P, Griffin I, and Tucker C, et al (2020) Baricitinib as potential treatment for 2019-nCoV acute respiratory disease Lancet 395(10223) e30–e1 https://doi.org/10.1016/S0140-6736(20)30304-4 PMID: 32032529
46. Ma Y, Wang C, and Xue M, et al (2018) The coronavirus transmissible gastroenteritis virus evades the type I interferon response through IRE1alpha-mediated manipulation of the microRNA miR-30a-5p/SOCS1/3 axis J Virol 92(22) https://doi.org/10.1128/JVI.00728-18
47. Yang CW, Lee YZ, and Kang IJ, et al (2010) Identification of phenanthroindolizines and phenanthroquinolizidines as novel potent anti-coronaviral agents for porcine enteropathogenic coronavirus transmissible gastroenteritis virus and human severe acute respiratory syndrome coronavirus Antivir Res 88(2) 160–168 https://doi.org/10.1016/j.antiviral.2010.08.009 PMID: 20727913
48. Yang C-W, Lee Y-Z, and Hsu H-Y, et al (2017) Targeting coronaviral replication and cellular JAK2 mediated dominant NF-κB activation for comprehensive and ultimate inhibition of coronaviral activity Sci Rep 7(1) 4105 https://doi.org/10.1038/s41598-017-04203-9 PMID: 28642467 PMCID: 5481340
49. Chen L, Liu HG, and Liu W, et al (2020) [Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia] Zhonghua jie he he hu xi za zhi = Zhonghua jiehe he huxi zazhi = Chin J Tuberc Respir Dis 43(0) E005
50. Zhu M (2004) SARS immunity and vaccination Cell Mol Immunol 1(3) 193–198
51. Miura TA, Wang J, and Holmes KV, et al (2007) Rat coronaviruses infect rat alveolar type I epithelial cells and induce expression of CXC chemokines Virology 369(2) 288–298 https://doi.org/10.1016/j.virol.2007.07.030 PMID: 17804032 PMCID: 2170429
52. Kritas SK, Ronconi G, and Caraffa A, et al (2020) Mast cells contribute to coronavirus-induced inflammation: new anti-inflammatory strategy J Biol Regul Homeost Agents 34(1)
53. Lin MH, Moses DC, and Hsieh CH, et al (2018) Disulfiram can inhibit MERS and SARS coronavirus papain-like proteases via different modes Antiviral Res 150 155–163 https://doi.org/10.1016/j.antiviral.2017.12.015 PMID: 29289665
54. Cheng KW, Cheng SC, and Chen WY, et al (2015) Thiopurine analogs and mycophenolic acid synergistically inhibit the papain-like protease of Middle East respiratory syndrome coronavirus Antiviral Res 115 9–16 https://doi.org/10.1016/j.antiviral.2014.12.011
55. Chan JF, Chan KH, and Kao RY, et al (2013) Broad-spectrum antivirals for the emerging Middle East respiratory syndrome coronavirus J Infect 67(6) 606–616 https://doi.org/10.1016/j.jinf.2013.09.029 PMID: 24096239
56. Shen L, Niu J, and Wang C, et al (2019) High-throughput screening and identification of potent broad-spectrum inhibitors of coronaviruses J Virol 93(12) https://doi.org/10.1128/JVI.00023-19
57. Hart BJ, Dyall J, and Postnikova E, et al (2014) Interferon-beta and mycophenolic acid are potent inhibitors of Middle East respiratory syndrome coronavirus in cell-based assays J Gen Virol 95(Pt 3) 571–577 https://doi.org/10.1099/vir.0.061911-0 PMCID: 3929173
58. Mo Y and Fisher D (2016) A review of treatment modalities for Middle East Respiratory Syndrome J Antimicrob Chemother 71(12) 3340–3350 https://doi.org/10.1093/jac/dkw338 PMID: 27585965
59. Al Ghamdi M, Alghamdi KM, and Ghandoora Y, et al (2016) Treatment outcomes for patients with Middle Eastern Respiratory Syndrome Coronavirus (MERS CoV) infection at a coronavirus referral center in the Kingdom of Saudi Arabia BMC Infect Dis 16(1) 174 https://doi.org/10.1186/s12879-016-1492-4 PMID: 27097824 PMCID: 4839124
60. AlGhamdi M, Mushtaq F, and Awn N, et al (2015) MERS CoV infection in two renal transplant recipients: case report Am J Transplant 15(4) 1101–1104 https://doi.org/10.1111/ajt.13085 PMID: 25716741
61. Carbajo-Lozoya J, Müller MA, and Kallies S, et al (2012) Replication of human coronaviruses SARS-CoV, HCoV-NL63 and HCoV-229E is inhibited by the drug FK506 Virus Res 165(1) 112–117 https://doi.org/10.1016/j.virusres.2012.02.002 PMID: 22349148
62. Carbajo-Lozoya J, Ma-Lauer Y, and Malesevic M, et al (2014) Human coronavirus NL63 replication is cyclophilin A-dependent and inhibited by non-immunosuppressive cyclosporine A-derivatives including Alisporivir Virus Res 184 44–53 https://doi.org/10.1016/j.virusres.2014.02.010 PMID: 24566223
Supplementary tables of summarised literature
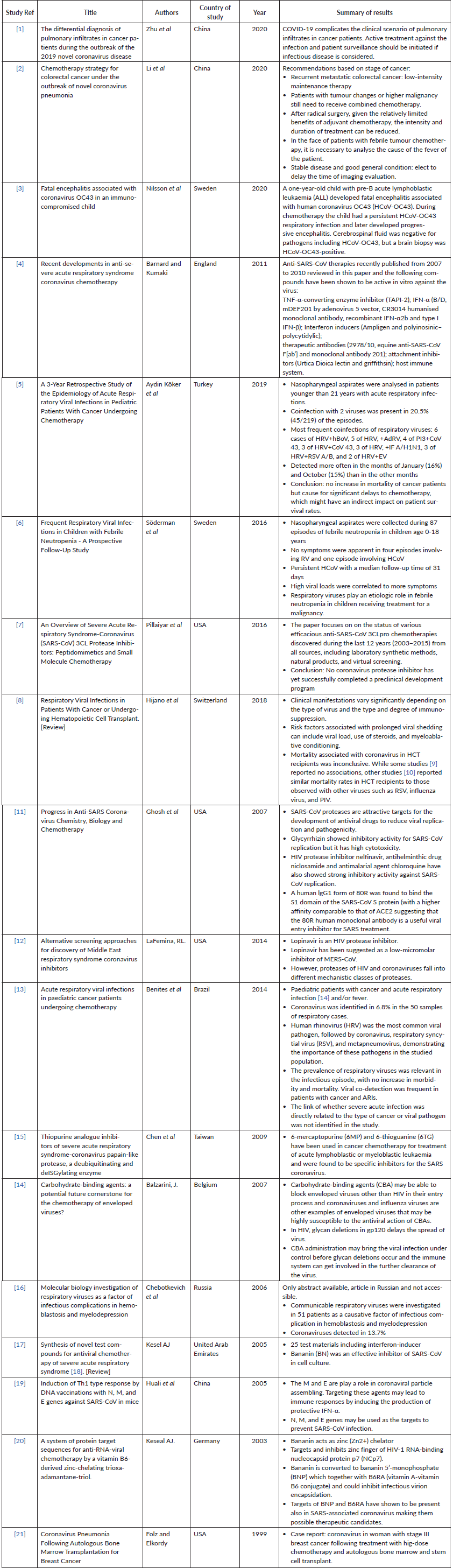
Supplementary Table 1. Current studies investigating COVID-19 and cytotoxic chemotherapy.
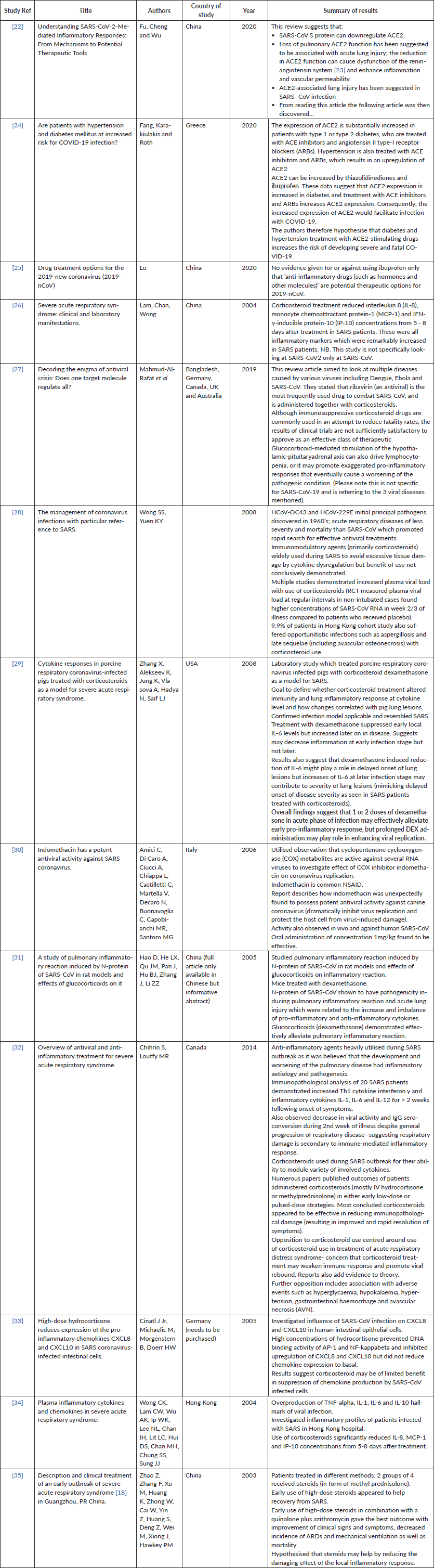
Supplementary Table 2. Current studies investigating COVID-19/ other coronavirus strains and low-dose steroids.
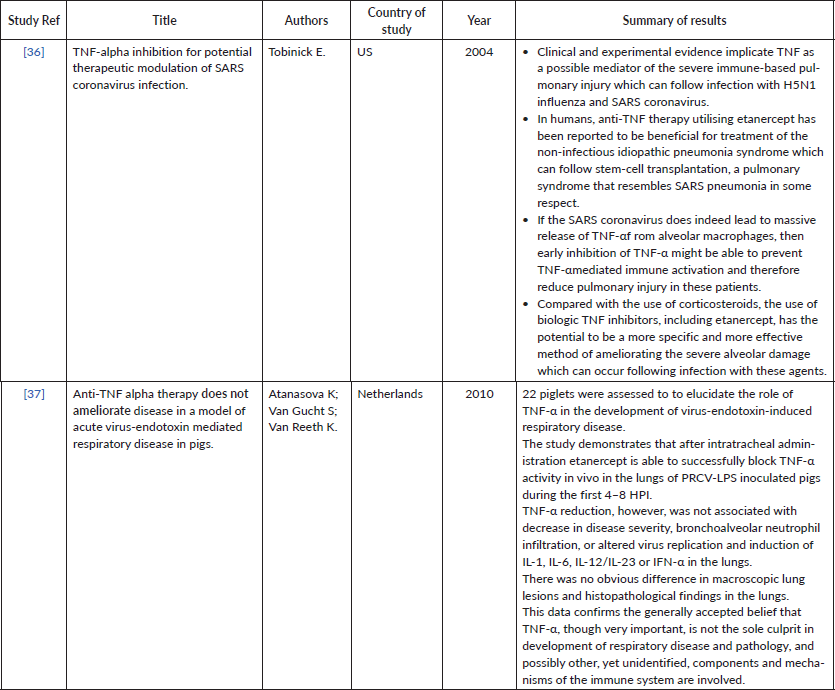
Supplementary Table 3. Current studies investigating COVID-19/other coronavirus strains and TNF blockers. (continued)
Supplementary Table 4. Current studies investigating COVID-19/ other coronavirus strains and IL-6. (continued)
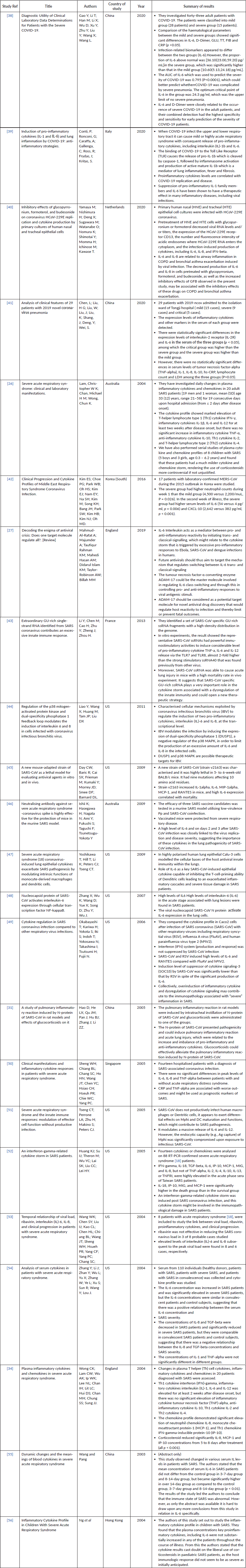
Supplementary Table 5. Current studies investigating COVID-19/ other coronavirus strains and JAK inhibitors
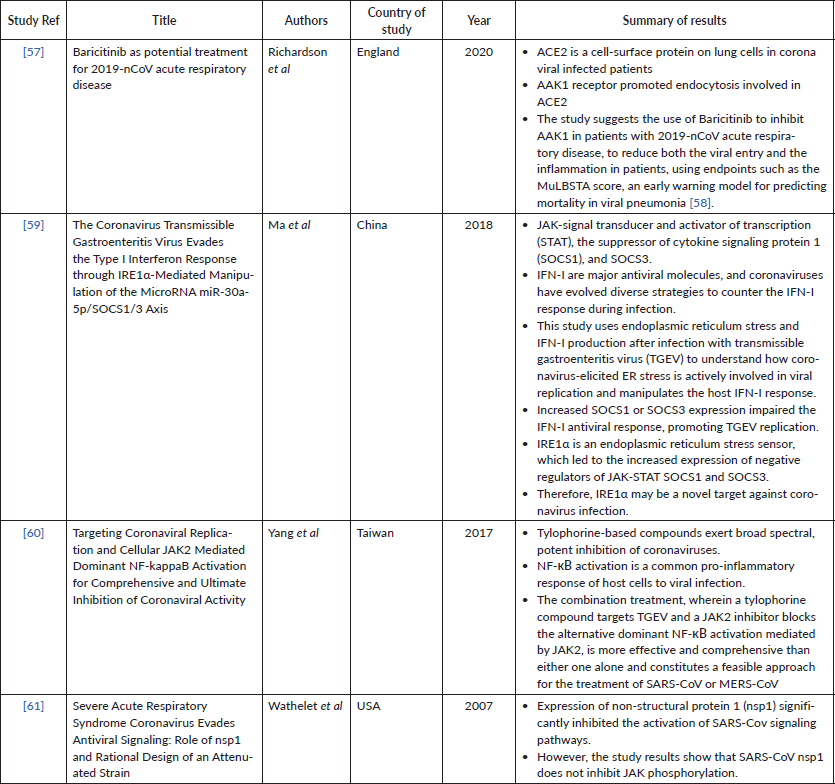
Supplementary Table 6. Current studies investigating COVID-19/ other coronavirus strains and IL-1 blockade.
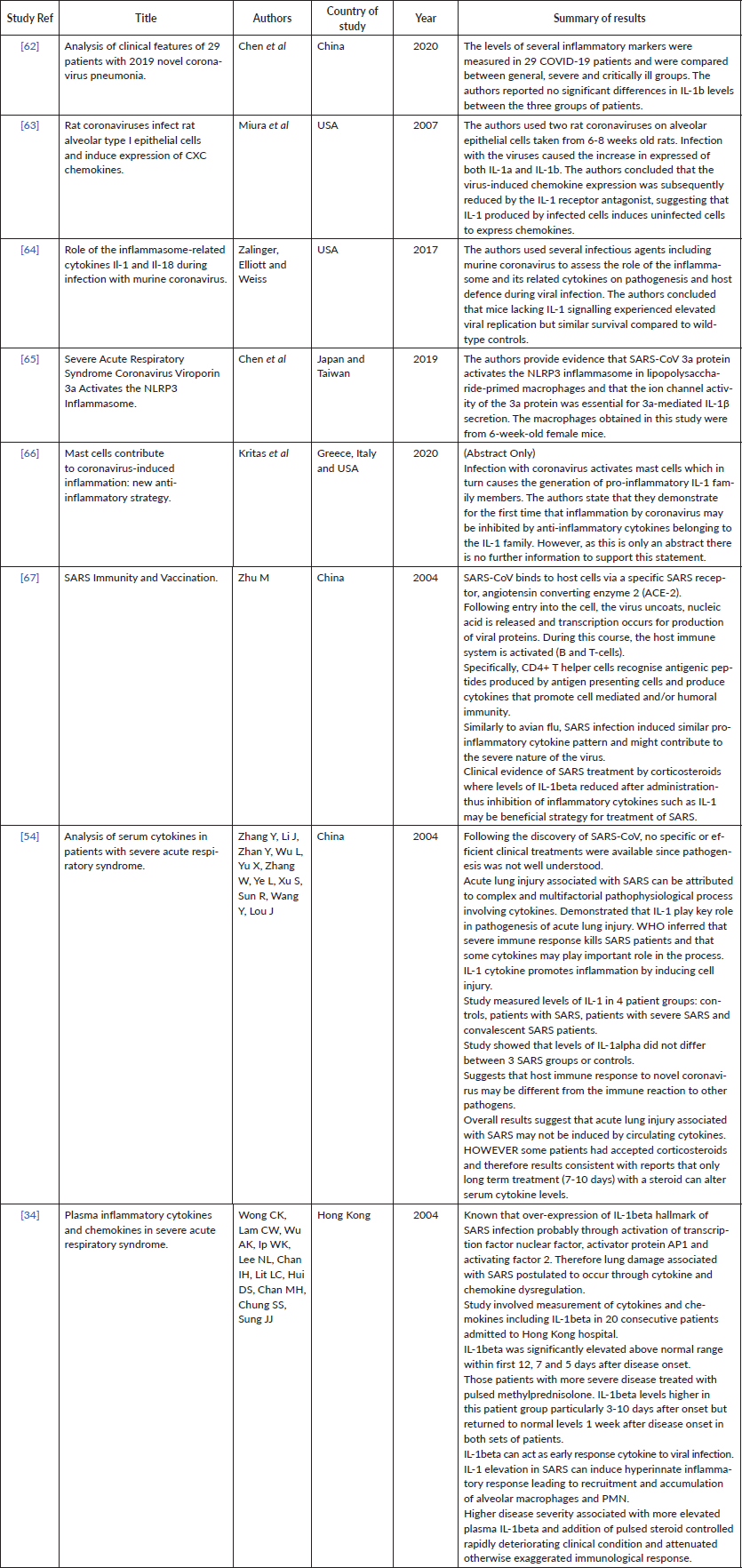
Supplementary Table 7. Current studies investigating COVID-19/ other coronavirus strains and Mycophenolate.
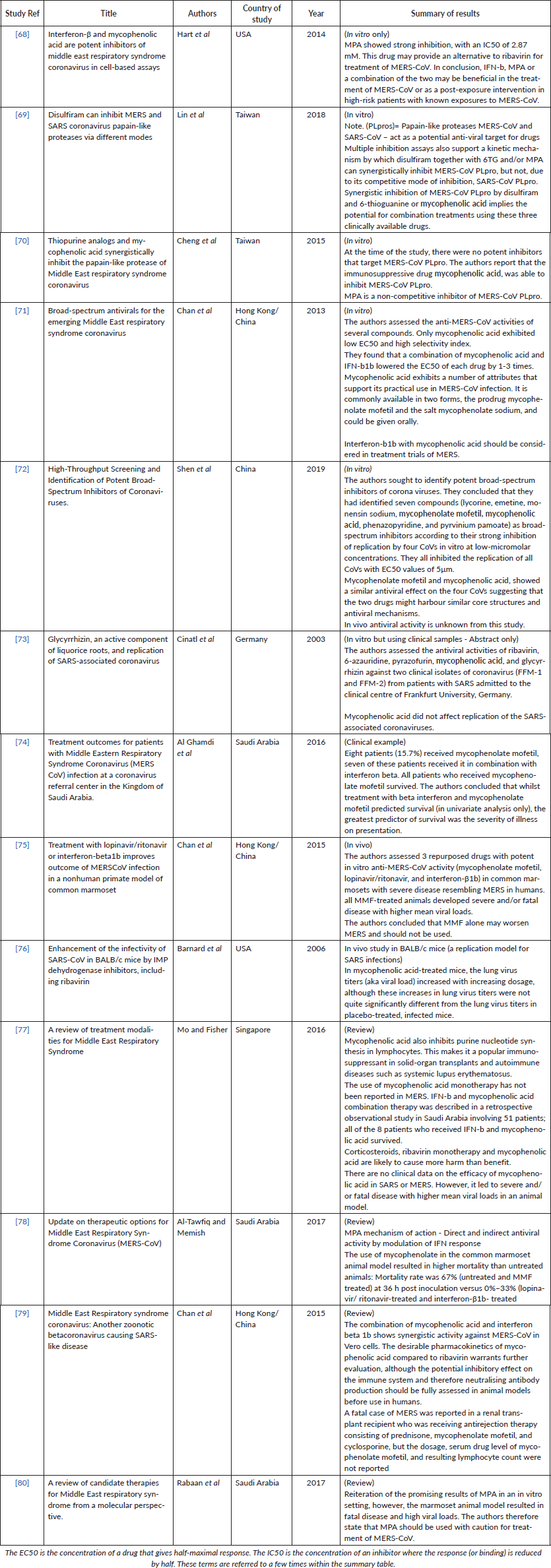
Supplementary Table 8. Current studies investigating COVID-19/ other coronavirus strains and Tacrolimus.
References
1. Zhu WJ, Wang J, and He XH, et al (2020) [The differential diagnosis of pulmonary infiltrates in cancer patients during the outbreak of the 2019 novel coronavirus disease] Zhonghua Zhong Liu Za Zhi 42(0) E008 PMID: 32133833
2. Li YH, Shen L, and Li J (2020) [Chemotherapy strategy for colorectal cancer under the outbreak of novel coronavirus pneumonia] Zhonghua Wei Chang Wai Ke Za Zhi 23(0) E004 PMID: 32100980
3. Nilsson A, Edner N, and Albert J, et al (2020) Fatal encephalitis associated with coronavirus OC43 in an immunocompromised child Infect Dis (Lond) 1–4
4. Barnard DL and Kumaki Y (2011) Recent developments in anti-severe acute respiratory syndrome coronavirus chemotherapy Future Virol 6(5) 615–631 https://doi.org/10.2217/fvl.11.33 PMID: 21765859 PMCID: 3136164
5. Aydin Köker S, Demirag B, and Tahta N, et al (2019) A 3-year retrospective study of the epidemiology of acute respiratory viral infections in pediatric patients with cancer undergoing chemotherapy J Pediatr Hematol Oncol 41(4) e242–e246 https://doi.org/10.1097/MPH.0000000000001418 PMID: 30688827
6. Soderman M, Rhedin S, and Tolfvenstam T, et al (2016) Frequent respiratory viral infections in children with febrile neutropenia—a prospective follow-up study PLoS One 11(6) e0157398 https://doi.org/10.1371/journal.pone.0157398
7. Pillaiyar T, Manickam M, and Namasivayam V, et al (2016) An overview of severe acute respiratory syndrome-coronavirus (SARS-CoV) 3CL protease inhibitors: peptidomimetics and small molecule chemotherapy J Med Chem 59(14) 6595–6628 https://doi.org/10.1021/acs.jmedchem.5b01461 PMID: 26878082 PMCID: 7075650
8. Hijano DR, Maron G, and Hayden RT (2018) Respiratory viral infections in patients with cancer or undergoing hematopoietic cell transplant Front Microbiol 9 3097 https://doi.org/10.3389/fmicb.2018.03097
9. Milano F, Campbell AP, and Guthrie KA, et al (2010) Human rhinovirus and coronavirus detection among allogeneic hematopoietic stem cell transplantation recipients Blood 115(10) 2088–2094 https://doi.org/10.1182/blood-2009-09-244152 PMID: 20042728 PMCID: 2837322
10. Ogimi C, Waghmare AA, and Kuypers JM, et al (2017) Clinical significance of human coronavirus in bronchoalveolar lavage samples from hematopoietic cell transplant recipients and patients with hematologic malignancies Clin Infect Dis 64(11) 1532–1539 https://doi.org/10.1093/cid/cix160 PMID: 28329354 PMCID: 5434339
11. Ghosh AK, Xi K, and Johnson ME, et al (2007) Progress in anti-SARS coronavirus chemistry, biology and chemotherapy Annu Rep Med Chem 41 183–196 PMID: 19649165 PMCID: 2718771
12. LaFemina RL (2014) Alternative screening approaches for discovery of middle east respiratory syndrome coronavirus inhibitors Antimicrob Agents Chemother 58(8) 4251 https://doi.org/10.1128/AAC.03406-14 PMID: 24867994 PMCID: 4135998
13. Benites EC, Cabrini DP, and Silva AC, et al (2014) Acute respiratory viral infections in pediatric cancer patients undergoing chemotherapy J Pediatr (Rio J) 90(4) 370–376 https://doi.org/10.1016/j.jped.2014.01.006
14. Balzarini J (2007) Carbohydrate-binding agents: a potential future cornerstone for the chemotherapy of enveloped viruses? Antivir Chem Chemother 18(1) 1–11 https://doi.org/10.1177/095632020701800101 PMID: 17354647
15. Chen X, Chou CY, and Chang GG (2009) Thiopurine analogue inhibitors of severe acute respiratory syndrome-coronavirus papain-like protease, a deubiquitinating and deISGylating enzyme Antivir Chem Chemother 19(4) 151–156 https://doi.org/10.1177/095632020901900402 PMID: 19374142
16. Chebotkevich VN, Bessmel’tsev SS, and Volkov AV, et al (2006) [Molecular biology investigation of respiratory viruses as a factor of infectious complications in hemoblastosis and myelodepression] Vopr Onkol 52(4) 421–426 PMID: 17024815
17. Kesel AJ (2005) Synthesis of novel test compounds for antiviral chemotherapy of severe acute respiratory syndrome (SARS) Curr Med Chem 12(18) 2095–2162 https://doi.org/10.2174/0929867054637644 PMID: 16101496
18. Wang C, Pang B-S, and National Research Project for Sars BG (2003) [Dynamic changes and the meanings of blood cytokines in severe acute respiratory syndrome] Zhonghua jie he he hu xi za zhi = Zhonghua jiehe he huxi zazhi = Chin J Tuberc Respir Dis 26(10) 586–589
19. Jin H, Xiao C, and Chen Z, et al (2005) Induction of Th1 type response by DNA vaccinations with N, M, and E genes against SARS-CoV in mice Biochem Biophys Res Commun 328(4) 979–986 https://doi.org/10.1016/j.bbrc.2005.01.048 PMID: 15707974
20. Kesel AJ (2003) A system of protein target sequences for anti-RNA-viral chemotherapy by a vitamin B6-derived zinc-chelating trioxa-adamantane-triol Bioorg Med Chem 11(21) 4599–4613 https://doi.org/10.1016/S0968-0896(03)00500-5 PMID: 14527557
21. Folz RJ and Elkordy MA (1999) Coronavirus pneumonia following autologous bone marrow transplantation for breast cancer Chest 115(3) 901–905 https://doi.org/10.1378/chest.115.3.901 PMID: 10084516
22. Fu Y, Cheng Y, and Wu Y (2020) Understanding SARS-CoV-2-mediated inflammatory responses: from mechanisms to potential therapeutic tools Virol Sin https://doi.org/10.1007/s12250-020-00207-4 PMID: 32125642 PMCID: 7035235
23. Acland HM, Gunson DE, and Gillette DM (1983) Ulcerative duodenitis in foals Vet Pathol 20(6) 653–661 https://doi.org/10.1177/030098588302000601 PMID: 6649336
24. Fang L, Karakiulakis G, and Roth M (2020) Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med https://doi.org/10.1016/S2213-2600(20)30116-8 PMID: 32171062
25. Lu H (2020) Drug treatment options for the 2019-new coronavirus (2019-nCoV) Biosci Trends https://doi.org/10.5582/bst.2020.01020
26. Lam CWK, Chan MHM, and Wong CK (2004) Severe acute respiratory syndrome: clinical and laboratory manifestations Clin Biochem Rev 25(2) 121–132
27. Mahmud-Al-Rafat A, Majumder A, and Taufiqur Rahman KM, et al (2019) Decoding the enigma of antiviral crisis: does one target molecule regulate all? Cytokine Cytokine 115 13–23 https://doi.org/10.1016/j.cyto.2018.12.008 PMID: 30616034
28. Wong SSY and Yuen K-Y (2008) The management of coronavirus infections with particular reference to SARS J Antimicrob Chemother 62(3) 437–441 https://doi.org/10.1093/jac/dkn243 PMID: 18565970
29. Zhang X, Alekseev K, and Jung K, et al (2008) Cytokine responses in porcine respiratory coronavirus-infected pigs treated with corticosteroids as a model for severe acute respiratory syndrome J Virol82(9) 4420–4428 https://doi.org/10.1128/JVI.02190-07 PMID: 18287230 PMCID: 2293053
30. Amici C, Di Caro A, and Ciucci A, et al (2006) Indomethacin has a potent antiviral activity against SARS coronavirus Antivir Ther 11(8) 1021–1030
31. Hao D, He L-X, and Qu J-M, et al (2005) [A study of pulmonary inflammatory reaction induced by N-protein of SARS-CoV in rat models and effects of glucocorticoids on it] Zhonghua nei ke za zhi 44(12) 890–893
32. Chihrin S and Loutfy MR (2005) Overview of antiviral and anti-inflammatory treatment for severe acute respiratory syndrome Expert Rev Anti-infect Ther 3(2) 251–262 https://doi.org/10.1586/14787210.3.2.251 PMID: 15918782
33. Cinatl J, Jr., Michaelis M, and Morgenstern B, et al (2005) High-dose hydrocortisone reduces expression of the pro-inflammatory chemokines CXCL8 and CXCL10 in SARS coronavirus-infected intestinal cells Int J Mol Med 15(2) 323–327 PMID: 15647850
34. Wong CK, Lam CWK, and Wu AKL, et al (2004) Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome Clin Exp Immunol 136(1) 95–103 https://doi.org/10.1111/j.1365-2249.2004.02415.x PMID: 15030519 PMCID: 1808997
35. Zhao Z, Zhang F, and Xu M, et al (2003) Description and clinical treatment of an early outbreak of severe acute respiratory syndrome (SARS) in Guangzhou, PR China J Med Microbiol 52(Pt 8) 715–720 https://doi.org/10.1099/jmm.0.05320-0 PMID: 12867568
36. Tobinick E (2004) TNF-alpha inhibition for potential therapeutic modulation of SARS coronavirus infection Curr Med Res Opin 20(1) 39–40 https://doi.org/10.1185/030079903125002757 PMID: 14741070
37. Atanasova K, Van Gucht S, and Van Reeth K (2010) Anti-TNF-alpha therapy does not ameliorate disease in a model of acute virus-endotoxin mediated respiratory disease in pigs Vet Immunol Immunopathol 137(1–2) 12–9 https://doi.org/10.1016/j.vetimm.2010.04.003 PMID: 20466438 PMCID: 2922464
38. Gao Y, Li T, and Han M, et al (2020) Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19 J Med Virol https://doi.org/10.1002/jmv.25770 PMID: 32181911
39. Conti P, Ronconi G, and Caraffa A, et al (2020) Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by COVID-19: anti-inflammatory strategies J Biol Regul Homeost Agents 34(2) pii:1
40. Yamaya M, Nishimura H, and Deng X, et al (2020) Inhibitory effects of glycopyrronium, formoterol, and budesonide on coronavirus HCoV-229E replication and cytokine production by primary cultures of human nasal and tracheal epithelial cells Respir Investig https://doi.org/10.1016/j.resinv.2019.12.005 PMID: 32094077
41. Chen L, Liu HG, and Liu W, et al (2020) [Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia] Zhonghua jie he he hu xi za zhi = Zhonghua jiehe he huxi zazhi = Chin J Tuberc Respir Dis 43(0) E005
42. Kim ES, Choe PG, and Park WB, et al (2016) Clinical progression and cytokine profiles of middle east respiratory syndrome coronavirus infection J Korean Medi Sci 31(11) 1717–1725 https://doi.org/10.3346/jkms.2016.31.11.1717
43. Li Y, Chen M, and Cao H, et al (2013) Extraordinary GU-rich single-strand RNA identified from SARS coronavirus contributes an excessive innate immune response Microb Infect 15(2) 88–95 https://doi.org/10.1016/j.micinf.2012.10.008
44. Liao Y, Wang X, and Huang M, et al (2011) Regulation of the p38 mitogen-activated protein kinase and dual-specificity phosphatase 1 feedback loop modulates the induction of interleukin 6 and 8 in cells infected with coronavirus infectious bronchitis virus Virology 420(2) 106–116 https://doi.org/10.1016/j.virol.2011.09.003 PMID: 21959016
45. Day CW, Baric R, and Cai SX, et al (2009) A new mouse-adapted strain of SARS-CoV as a lethal model for evaluating antiviral agents in vitro and in vivo Virology 395(2) 210–222 https://doi.org/10.1016/j.virol.2009.09.023 PMID: 19853271 PMCID: 2787736
46. Ishii K, Hasegawa H, and Nagata N, et al Neutralizing antibody against severe acute respiratory syndrome (SARS)-coronavirus spike is highly effective for the protection of mice in the murine SARS model Microbiol Immunol 53(2) 75–82 PMID: 19291090
47. Yoshikawa T, Hill T, and Li K, et al (2009) Severe acute respiratory syndrome (SARS) coronavirus-induced lung epithelial cytokines exacerbate SARS pathogenesis by modulating intrinsic functions of monocyte-derived macrophages and dendritic cells J Virol 83(7) 3039–3048 https://doi.org/10.1128/JVI.01792-08 PMCID: 2655569
48. Zhang X, Wu K, and Wang D, et al (2007) Nucleocapsid protein of SARS-CoV activates interleukin-6 expression through cellular transcription factor NF-kappaB Virology 365(2) 324–335 https://doi.org/10.1016/j.virol.2007.04.009 PMID: 17490702
49. Okabayashi T, Kariwa H, and Yokota S-I, et al (2006) Cytokine regulation in SARS coronavirus infection compared to other respiratory virus infections J Med Virol 78(4) 417–424 https://doi.org/10.1002/jmv.20556 PMID: 16482545
50. Sheng W-H, Chiang B-L, and Chang S-C, et al (2005) Clinical manifestations and inflammatory cytokine responses in patients with severe acute respiratory syndrome J Formosan Med Assoc = Taiwan yi zhi 104(10) 715–723 PMID: 16385373
51. Tseng C-TK, Perrone LA, and Zhu H, et al (2005) Severe acute respiratory syndrome and the innate immune responses: modulation of effector cell function without productive infection J Immunol 174(12) 7977–7985 https://doi.org/10.4049/jimmunol.174.12.7977 PMID: 15944304
52. Huang K-J, Su I-J, and Theron M, et al (2005) An interferon-gamma-related cytokine storm in SARS patients J Med Virol 75(2) 185–194 https://doi.org/10.1002/jmv.20255
53. Wang W-K, Chen S-Y, and Liu IJ, et al (2004) Temporal relationship of viral load, ribavirin, interleukin (IL)-6, IL-8, and clinical progression in patients with severe acute respiratory syndrome Clin Infect Dis 39(7) 1071–1075 https://doi.org/10.1086/423808 PMID: 15472864
54. Zhang Y, Li J, and Zhan Y, et al (2004) Analysis of serum cytokines in patients with severe acute respiratory syndrome Infect Immun 72(8) 4410–4415 https://doi.org/10.1128/IAI.72.8.4410-4415.2004 PMID: 15271897 PMCID: 470699
55. Wang C and Pang B (2003) Dynamic changes and the meanings of blood cytokines in severe acute respiratory syndrome Chin J Tuberc Respir Dis 26(10) 586–589
56. Ng PC, Lam CWK, and Li AM, et al (2004) Inflammatory cytokine profile in children with severe acute respiratory syndrome Pediatrics 113 e7–e14 https://doi.org/10.1542/peds.113.1.e7 PMID: 14702488
57. Richardson P, Griffin I, and Tucker C, et al (2020) Baricitinib as potential treatment for 2019-nCoV acute respiratory disease Lancet 395(10223) e30–e1 https://doi.org/10.1016/S0140-6736(20)30304-4 PMID: 32032529
58. Chen N, Zhou M, and Dong X, et al (2020) Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study Lancet 395(10223) 507–513 https://doi.org/10.1016/S0140-6736(20)30211-7 PMID: 32007143
59. Ma Y, Wang C, and Xue M, et al (2018) The coronavirus transmissible gastroenteritis virus evades the type I interferon response through IRE1alpha-mediated manipulation of the microRNA miR-30a-5p/SOCS1/3 axis J Virol 92(22) https://doi.org/10.1128/JVI.00728-18
60. Yang C-W, Lee Y-Z, and Hsu H-Y, et al (2017) Targeting coronaviral replication and cellular JAK2 mediated dominant NF-κB activation for comprehensive and ultimate inhibition of coronaviral activity Sci Rep 7(1) 4105 https://doi.org/10.1038/s41598-017-04203-9 PMID: 28642467 PMCID: 5481340
61. Wathelet MG, Orr M, and Frieman MB, et al (2007) Severe acute respiratory syndrome coronavirus evades antiviral signaling: role of nsp1 and rational design of an attenuated strain J Virol 81(21) 11620–11633 https://doi.org/10.1128/JVI.00702-07 PMID: 17715225 PMCID: 2168762
62. Chen L, Liu HG, and Liu W, et al (2020) [Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia] Zhonghua jie he he hu xi za zhi = Zhonghua jiehe he huxi zazhi = Chin J Tuberc Respir Dis 43(0) E005
63. Miura TA, Wang J, and Holmes KV, et al (2007) Rat coronaviruses infect rat alveolar type I epithelial cells and induce expression of CXC chemokines Virology 369(2) 288–298 https://doi.org/10.1016/j.virol.2007.07.030 PMID: 17804032 PMCID: 2170429
64. Zalinger ZB, Elliott R, and Weiss SR (2017) Role of the inflammasome-related cytokines Il-1 and Il-18 during infection with murine coronavirus J Neurovirol 23(6) 845–854 https://doi.org/10.1007/s13365-017-0574-4 PMID: 28895072 PMCID: 5726909
65. Chen IY, Moriyama M, and Chang M-F, et al (2019) Severe acute respiratory syndrome coronavirus viroporin 3a activates the nlrp3 inflammasome Front Microbiol 10 50 https://doi.org/10.3389/fmicb.2019.00050 PMID: 30761102 PMCID: 6361828
66. Kritas SK, Ronconi G, and Caraffa A, et al (2020) Mast cells contribute to coronavirus-induced inflammation: new anti-inflammatory strategy J Biol Regul Homeostat Agents 34(1)
67. Zhu M (2004) SARS immunity and vaccination Cell Mol Immunol 1(3) 193–198
68. Hart BJ, Dyall J, and Postnikova E, et al (2014) Interferon-beta and mycophenolic acid are potent inhibitors of Middle East respiratory syndrome coronavirus in cell-based assays J Gen Virol 95(Pt 3) 571–577 https://doi.org/10.1099/vir.0.061911-0 PMCID: 3929173
69. Lin MH, Moses DC, and Hsieh CH, et al (2018) Disulfiram can inhibit MERS and SARS coronavirus papain-like proteases via different modes Antiviral Res 150 155–163 https://doi.org/10.1016/j.antiviral.2017.12.015 PMID: 29289665
70. Cheng KW, Cheng SC, and Chen WY, et al (2015) Thiopurine analogs and mycophenolic acid synergistically inhibit the papain-like protease of Middle East respiratory syndrome coronavirus Antiviral Res 115 9–16 https://doi.org/10.1016/j.antiviral.2014.12.011
71. Chan JF, Chan KH, and Kao RY, et al (2013) Broad-spectrum antivirals for the emerging Middle East respiratory syndrome coronavirus J Infect 67(6) 606–616 https://doi.org/10.1016/j.jinf.2013.09.029 PMID: 24096239
72. Shen L, Niu J, and Wang C, et al (2019) High-throughput screening and identification of potent broad-spectrum inhibitors of coronaviruses J Virol 93(12) https://doi.org/10.1128/JVI.00023-19
73. Cinatl J, Morgenstern B, and Bauer G, et al (2003) Glycyrrhizin, an active component of liquorice roots, and replication of SARS-associated coronavirus Lancet 361(9374) 2045–2046 https://doi.org/10.1016/S0140-6736(03)13615-X PMID: 12814717
74. Al Ghamdi M, Alghamdi KM, and Ghandoora Y, et al (2016) Treatment outcomes for patients with Middle Eastern Respiratory Syndrome Coronavirus (MERS CoV) infection at a coronavirus referral center in the Kingdom of Saudi Arabia BMC Infect Dis 16(1) 174 https://doi.org/10.1186/s12879-016-1492-4 PMID: 27097824 PMCID: 4839124
75. Chan JF, Yao Y, and Yeung ML, et al (2015) Treatment with lopinavir/ritonavir or interferon-beta1b improves outcome of MERS-CoV infection in a nonhuman primate model of common marmoset J Infect Dis 212(12) 1904–1913 https://doi.org/10.1093/infdis/jiv392 PMID: 26198719
76. Barnard DL, Day CW, and Bailey K, et al (2006) Enhancement of the infectivity of SARS-CoV in BALB/c mice by IMP dehydrogenase inhibitors, including ribavirin Antiviral Res 71(1) 53–63 https://doi.org/10.1016/j.antiviral.2006.03.001 PMID: 16621037
77. Mo Y and Fisher D (2016) A review of treatment modalities for Middle East Respiratory Syndrome J Antimicrob Chemother 71(12) 3340–3350 https://doi.org/10.1093/jac/dkw338 PMID: 27585965
78. Al-Tawfiq JA and Memish ZA (2017) Update on therapeutic options for Middle East Respiratory Syndrome Coronavirus (MERS-CoV) Expert Rev Anti Infect Ther 15(3) 269–275 https://doi.org/10.1080/14787210.2017.1271712
79. Chan JF, Lau SK, and To KK, et al (2015) Middle East respiratory syndrome coronavirus: another zoonotic betacoronavirus causing SARS-like disease Clin Microbiol Rev 28(2) 465–522 https://doi.org/10.1128/CMR.00102-14 PMID: 25810418 PMCID: 4402954
80. Rabaan AA, Alahmed SH, and Bazzi AM, et al (2017) A review of candidate therapies for Middle East respiratory syndrome from a molecular perspective J Med Microbiol 66(9) 1261–1274 https://doi.org/10.1099/jmm.0.000565 PMID: 28855003 PMCID: 7079582
81. Carbajo-Lozoya J, Müller MA, and Kallies S, et al (2012) Replication of human coronaviruses SARS-CoV, HCoV-NL63 and HCoV-229E is inhibited by the drug FK506 Virus Res 165(1) 112–117 https://doi.org/10.1016/j.virusres.2012.02.002 PMID: 22349148
82. AlGhamdi M, Mushtaq F, and Awn N, et al (2015) MERS CoV infection in two renal transplant recipients: case report Am J Transplant 15(4) 1101–1104 https://doi.org/10.1111/ajt.13085 PMID: 25716741
83. Carbajo-Lozoya J, Ma-Lauer Y, and Malesevic M, et al Human coronavirus NL63 replication is cyclophilin A-dependent and inhibited by non-immunosuppressive cyclosporine A-derivatives including Alisporivir Virus Res 184 44–53 PMID: 24566223






