Reducing delays in radiotherapy initiation post CT simulation: a quality improvement study from a rural cancer center in India
Pragyat Thakur1, Nagarjun Ballari1, Anureet Kaur2a, Tapas Kumar Dora1, I Vedamanasa1, Arshdeep Kaur1 and Ashish Gulia3
1Department of Radiation Oncology, Homi Bhabha Cancer Hospital and Research Centre (HBCHRC), Tata Memorial Centre, Affiliated to Homi Bhabha National Institute (HBNI), Mullanpur, Punjab, India
2Department of Radiation Oncology, Homi Bhabha Cancer Hospital (HBCH), Tata Memorial Centre, Affiliated to Baba Farid University of Health Sciences (BFUHS), Sangrur, Punjab, India
3Department of Surgical Oncology, Homi Bhabha Cancer Hospital and Research Centre (HBCHRC), Tata Memorial Center, Affiliated to Homi Bhabha National Institute (HBNI), Mullanpur, Punjab, India
a https://orcid.org/0009-0008-6823-4395
Abstract
Radiation therapy (RT) is a cornerstone in the treatment of solid tumours, with more than half of all cancer patients requiring it for curative or palliative intent. However, delays in initiating RT after CT simulation (CT sim) can significantly impact clinical outcomes by increasing recurrence risk, triggering re-simulation due to anatomical shifts and causing psychological and logistical distress for patients and caregivers. This prospective quality improvement (QI) study was conducted at a rural cancer center in India from April to December 2022, aiming to reduce the time from CT sim to RT initiation. Using the A3 methodology in collaboration with Enable Quality, Improve Patient Care India, Stanford Medicine and the National Cancer Grid, a root cause analysis was conducted, followed by key driver identification via Pareto analysis. A series of Plan-Do-Study-Act (PDSA) cycles led to targeted interventions, including written standard operating procedures (SOPs) for scheduling, standardised patient instructions, clearly defined staff roles and stakeholder education. Data from 200 patients planned for radical treatment were analysed weekly, with treatment timelines plotted on a run chart. At baseline, the median delay from simulation to treatment initiation was 18 days. After implementation of interventions in July 2022, this was reduced to 12 days by September and further to 10 days by November, a 44.4% reduction. Additionally, the re-simulation rate dropped from 10% to less than 1%. No patient experienced a delay beyond the prescribed date, and importantly, staff feedback confirmed that the revised workflow did not increase perceived workload. The interventions were institutionalised through SOPs and monitored via real time dashboards, ensuring sustainability into 2024. This study demonstrates that targeted, systemlevel interventions developed using accessible QI methodologies can lead to meaningful reductions in RT delays and operational inefficiencies in low-resource environments. The model is feasible, cost-effective and adaptable to other cancer centers aiming to optimise timely RT delivery and improve patient outcomes.
Keywords: radiotherapy, quality improvement, CT simulation, time to treatment, workflow redesign, root cause analysis, treatment delay
Correspondence to: Anureet Kaur
Email: parmaranureet3@gmail.com
Published: 01/05/2026
Received: 24/06/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Radiation therapy (RT) is a cornerstone in the management of solid tumours, with over half of all cancer patients receiving it at some point during their treatment course, either alone or in combination with other modalities [1–3]. The timely initiation of RT is critical for optimising clinical outcomes, especially in settings where treatment delays are common.
Time to treatment initiation (TTI), the interval between diagnosis and the start of therapy, is a well-established determinant of cancer-related outcomes [4]. In head and neck squamous cell carcinoma (HNSCC), timely postoperative RT is essential for achieving locoregional control and improving survival, particularly in patients with advanced stage or high-risk disease [5, 6]. Delays in initiating RT have been consistently linked to higher recurrence rates [7, 8] and increased cancer specific and overall mortality [9–11]. Beyond oncologic outcomes, delays may also lead to increased rates of resimulation due to changes in patient anatomy, increased psychological distress and place a greater burden on healthcare systems [12].
Despite significant advancements in RT technology, delays between CT simulation (CT Sim) and treatment initiation remain a persistent challenge, particularly in low-resource or rural healthcare settings. While existing literature has largely focused on delays from diagnosis to treatment [13] or postoperative RT timing, the specific interval between CT sim and the delivery of the first RT fraction remains understudied.
This quality improvement (QI) study was conducted at a rural cancer center in India with the objective of identifying key factors contributing to post simulation delays in RT initiation. Using structured QI tools, we aimed to implement targeted interventions to reduce the delay between CT sim and initiation of RT for cancer patients across various tumour sites .
Materials and methods
The study spanned a 9 month period, from April to December 2022, and enrolled a total of 200 patients who underwent CT sim for planned external beam RT.
The QI initiative followed the A3 methodology, a structured problem-solving approach widely used in healthcare process optimisation. It was implemented in collaboration with the Enable Quality, Improve Patient Care (EQUIP India) program of Stanford Medicine and the National Cancer Grid, India. The A3 process involved defining the problem, analysing the root causes, implementing targeted interventions and assessing outcomes.
A Gemba Walk, an observational process in which team members visit the clinical workspace to directly observe workflows (Figure 1), was conducted during regular departmental hours. This helped identify systemic and process-level inefficiencies contributing to delays in treatment initiation.
A root cause analysis (RCA) was subsequently performed, involving multidisciplinary stakeholders including radiation oncologists, medical physicists, radiotherapy technologists (RTTs), nursing and administrative staff. An EQUIP questionnaire (Table 1) was specifically developed to assess various contributing factors to RT initiation delays using a standardised five point Likert scale (Table 2), allowing respondents to rate their level of agreement with each identified cause. The questionnaire was developed through an iterative, consensus-driven process involving frontline stakeholders, including radiation oncologists, medical physicists and RTTs with direct experience in workflow bottlenecks. Initial item generation was informed by insights from Gemba walks, fishbone analysis and literature on radiation workflow delays. To ensure content validity, the draft questionnaire was reviewed in two structured feedback sessions by a panel of five senior faculty members with expertise in radiation oncology operations and QI. Items were revised based on clarity, relevance and redundancy. A pilot administration was then conducted among a representative sample of ten staff members across different roles (RTTs, residents and nursing staff) to assess response variability and comprehension. Internal consistency was evaluated using Cronbach’s alpha, which demonstrated acceptable reliability (α = 0.81). The final version focused on identifying specific factors delaying RT initiation post CT sim.
A fishbone diagram (Ishikawa analysis) was constructed to categorise contributing factors into major domains. Pareto analysis, based on the 80/20 principle, which posits that approximately 80% of delays can be attributed to 20% of the underlying causes, was employed to prioritise high-impact drivers of delay according to their frequency and relative contribution. Informed by the RCA and Pareto analysis, a multidisciplinary team developed Targeted interventions using the Plan-Do-Study-Act (PDSA) framework. Interventions were tested iteratively over several cycles to evaluate feasibility, scalability and effectiveness.
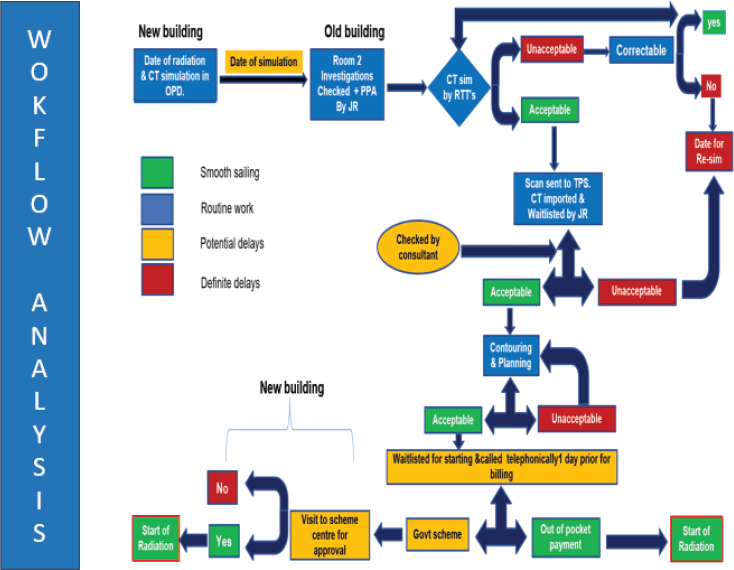
Figure 1. CT sim to RT workflow derived from Gemba Walk analysis. This figure illustrates the end-to-end workflow from CT sim to the initiation of RT at our center, as mapped during a Gemba Walk exercise. Key steps, such as simulation, planning and scheduling, are highlighted along with critical transition points and bottlenecks contributing to delays. Abbreviations: CT, Computed Tomography; JR, Junior Resident; OPD, Outpatient Department; PPA, Pre-Planning Audit; Re-sim, Resimulation; RTT, Radiation Therapy Technologist; Sim, Simulation; TPS, Treatment Planning System.
The primary outcome defined for this QI initiative was the proportion of patients scheduled to initiate RT within 10 days of CT sim, based on the institutional benchmark for timely treatment initiation.
The secondary outcomes included predefined process measures:
the rate of re-simulations,
patient compliance with scheduled appointments,
patient satisfaction with the workflow and
the degree of workflow redundancy and administrative load on staff.
Table 1. EqUIP questionnaire to identify factors contributing to delays in RT initiation post-CT sim.

Table 2. Likert scale to score the EqUIP questionnaire.

To evaluate these outcomes, weekly CT Sim to RT initiation intervals were prospectively recorded throughout the study period. These data were plotted on run charts as part of routine QI monitoring. Interventions implemented during the PDSA cycles were annotated directly onto the charts to allow temporal association between process changes and subsequent variations in outcome measures.
Following the intervention phase, a structured postimplementation monitoring plan was introduced in January 2023. This included weekly audit rounds, integration of key performance indicators (e.g., CT Simtotreatment interval, re-simulation rate) into departmental dashboards and structured orientation of incoming staff to the revised Standard Operating Procedures (SOPs). These activities were integrated into routine departmental operations and continued through 2024 to promote sustainability and institutionalisation of the intervention bundle.
Data collection and analysis were performed using Microsoft Excel (Microsoft Corporation, Redmond, WA). Median waiting time from CT sim to RT initiation before and after intervention were compared descriptively to assess the effectiveness of the QI initiative. Feedback from departmental staff and patients was also documented to assess perceived acceptability and operational impact. This study was conducted as part of an institutional QI program and adhered to ethical standards for QI research.
Results
At baseline, the median interval from CT sim to the initiation of RT at our institution was 18 days.
As part of the RCA, a Fishbone (Ishikawa) diagram (Figure 2) was constructed to visually organise the contributing factors underlying delays in RT initiation postCT sim. These factors were systematically grouped into five primary domains: personnel (e.g., unclear staff responsibilities), methods (e.g., lack of standardised SOPs), machines (e.g., scan import delays), logistics (e.g., government documentation bottlenecks) and patient-related issues (e.g., inadequate instruction comprehension).
RCA was further supported by Pareto analysis (Figure 3), which identified four major causes of delay:
Inefficiencies in RT appointment scheduling
Inadequate patient comprehension of preparatory instructions
Ambiguity in staff roles and responsibilities
Gaps in coordinated care delivery across services, including delays in processing government documentation, which were addressed through the KEVAT desk, a dedicated patient navigation system designed to streamline access to health schemes.
To address these, a series of targeted, stepwise interventions (Figure 4) were implemented through the PDSA framework. These included:
Creation of a centralised scheduling system with daily caps on simulation numbers
Disease-specific SOPs with clearly printed simulation instructions
Mandatory weekly simulation list updates by RTTs
Pre-planning audits (PPAs) by junior residents (JRs)
Daily scan import and verification protocols
Advance communication with patients regarding logistics and preparation
On-site completion of financial documentation on the day of simulation
Consultant-assigned site responsibilities with regular review audits
A run chart (Figure 5) was used to plot the weekly RT initiation times across the 9-month period.
A second run chart (Figure 6) tracked weekly median delays from CT sim to RT initiation following the implementation of targeted interventions. Progressive reductions were observed with each cycle. The baseline average delay of 18 days (red line) showed a consistent decline across October and November 2022, corresponding with key interventions such as centralised scheduling (items 1, 3, 4, 6, 7, 8), consultant-led review processes (item 10) and integration of KEVAT documentation on the day of simulation (item 11). These targeted interventions aligned with key driver categories, scheduling, instruction clarity, role definition and coordinated planning, and contributed to achieving the predefined target of 10 days (green line) by mid-November. Weekly metrics fell below the target line in the latter part of the study, indicating durable improvement.
By September 2022, the median time from simulation to RT initiation dropped from 18 to 12 days. Following further process refinements, including consultant performance review meetings and streamlined KEVAT services for the timely completion of documents for government schemes, the median duration was reduced to the target of 10 days by November 2022, representing a 44.4% reduction in waiting time.
In addition to the marked reduction in median simulation to treatment interval, several secondary outcome measures demonstrated significant improvement, underscoring the effectiveness and sustainability of the implemented interventions. Notably, the proportion of patients initiating RT within 10 days of CT sim increased from 30% at baseline to 78% by the end of the study period. This shift reflects not only an improvement in central tendency but also a favourable change in the overall distribution of waiting times, indicating broader systemic impact.
Re-simulation rates, a critical quality indicator reflecting planning inefficiencies and treatment delays, declined from 10% to less than 1%. This reduction was primarily attributable to improved standardisation of simulation protocols, pre-planning audits and timely identification of imaging or positioning issues, which minimised the need for duplicate scans and redundant planning efforts.
Importantly, from October 2022 onward, no patient experienced a delay in RT initiation beyond their clinically indicated start date, reflecting adherence to planned schedules and enhanced operational control.
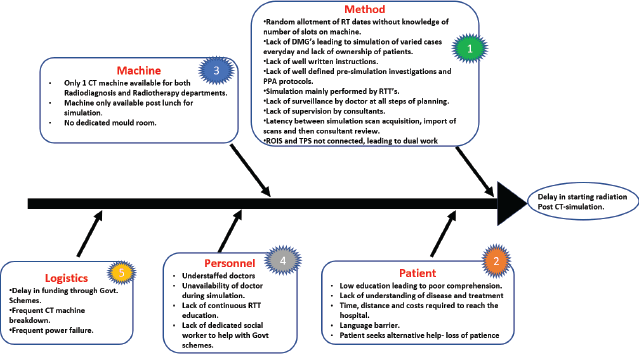
Figure 2. Fishbone analysis categorising systemic causes of delay in RT initiation into five domains. Abbreviations: CT, computed tomography; DMG, Disease Management Group; PPA, Pre Planning Audit; ROIS, radiation oncology information system; RT, radiation therapy; RTT, radiation therapy technologist; TPS, treatment planning system.
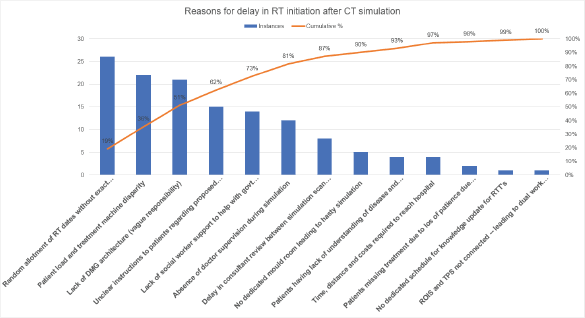
Figure 3. Pareto chart summarising the cumulative percentage of major causes contributing to delay in RT initiation post-simulation. Abbreviations: DMG, Disease Management Group; ROIS, radiation oncology information system; RT, radiation therapy; RTT, radiation therapy technologist; TPS, treatment planning system.
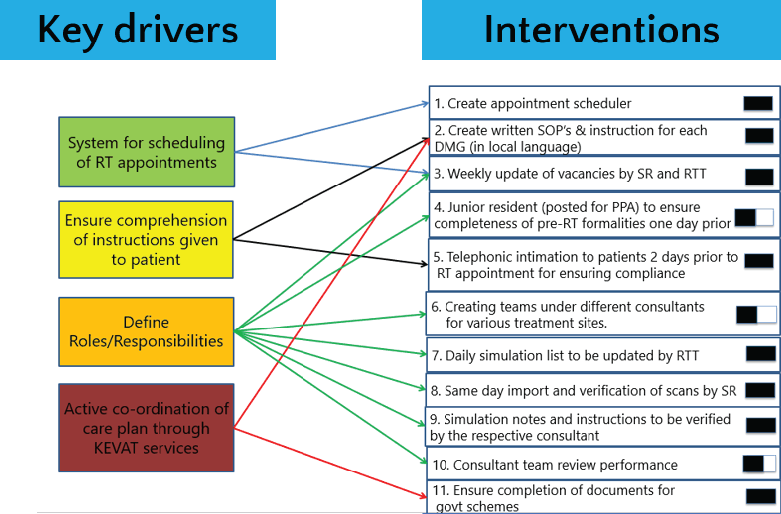
Figure 4. Root causes and interventions for delays in RT initiation. This figure outlines root causes of delay, categorised into four primary drivers and the corresponding corrective interventions implemented. Each color-coded driver (e.g., scheduling, communication, role clarity, KEVAT coordination) is linked to specific actionable steps designed to address the identified bottlenecks. Half and full bars denote the degree of implementation of each intervention (partial versus complete). Abbreviations: DMG, Disease Management Group; PPA, Pre-Planning Audit; RTT, Radiation Therapy Technologist; SOPs, Standard Operating Procedures; SR, Senior Resident.
The revised SOPs were institutionalised in January 2023 and consistently followed over the subsequent 2 years. Departmental audits during this period confirmed that the median CT Sim to treatment interval remained stable within the 10–12 day range, and re-simulation rates were sustained below 2%, compared to 10% at baseline. Additionally, structured quarterly feedback was obtained from clinical staff, including nursing personnel, RTTs, medical physicists and radiation oncology residents. Among 36 anonymised responses collected between March 2023 and December 2024, 89% (n = 32) reported no increase in perceived workload, 81% (n = 29) noted improved task clarity and 83% (n = 30) reported smoother interdepartmental coordination. These findings underscore the long-term operational feasibility and cross disciplinary acceptance of the intervention bundle.
Discussion
Delays in initiating RT are a well-recognised challenge in oncology; they are frequent, multifactorial and associated with worse clinical outcomes [14]. Even modest postponements may allow for tumour proliferation and disease upstaging, compromising local control and survival, particularly in aggressive malignancies such as head and neck and cervical cancers [15, 16].
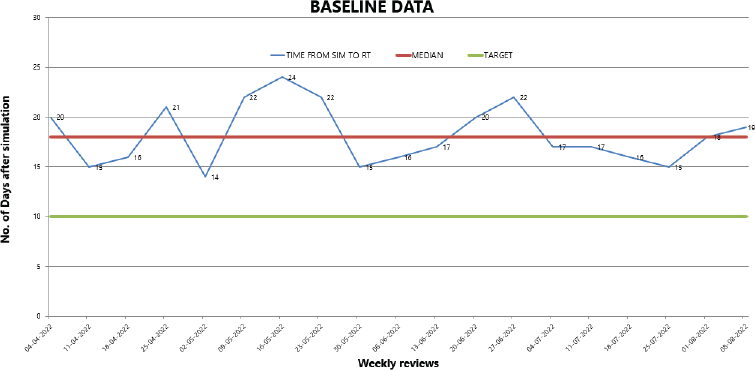
Figure 5. Run Chart displaying the weekly median number of days between CT sim and RT initiation prior to intervention implementation. The blue line represents weekly values, the red line indicates the pre-intervention median (18 days) and the green line marks the target goal of 10 days.
In prior work, Xu et al [13] employed Failure Mode and Effects Analysis to evaluate vulnerabilities in the RT workflow, particularly in the interval between CT sim and treatment delivery. However, their work remained largely diagnostic. In contrast, our study went beyond risk identification to implement proactive, data-driven interventions aimed at reducing delays. The resulting median CT Sim to treatment interval of 10 days postintervention not only represents an operational success but also carries potential clinical implications in minimising tumour progression during waiting periods.
Delays in treatment initiation are a particularly significant concern in India, where systemic and infrastructural limitations prolong TTI. A recent multicenter study reported a median TTI of 20 days (interquartile range (IQR) 7–39) from diagnosis to therapy, with a median of 27.5 days (IQR 10–49) for RT-specific starts [17]. Against this national benchmark, our reduction of the simulation to treatment gap to 10 days is both substantial and encouraging. It illustrates how targeted workflow improvements can mitigate larger systemic delays. Internationally, high-income countries often enforce stricter timeliness standards.
Rural cancer centers in low and middle-income countries (LMICs) face unique challenges. RT infrastructure in India is often concentrated in urban areas, leading to travel burdens and significant financial and logistical strain for rural patients. These barriers can result in treatment abandonment, missed appointments and worse outcomes. Prior to our intervention, patients frequently waited over 2 weeks after simulation, requiring multiple hospital visits or extended stays, which imposed considerable personal and economic hardship. These challenges align with evidence linking travel burden to poor treatment adherence and survival outcomes among rural cancer patients [18]. Our intervention directly addressed these disparities. A shorter median wait time of 10 days improves the likelihood of patients completing curative RT timely manner, thereby reducing dropout risk and the window for disease progression.
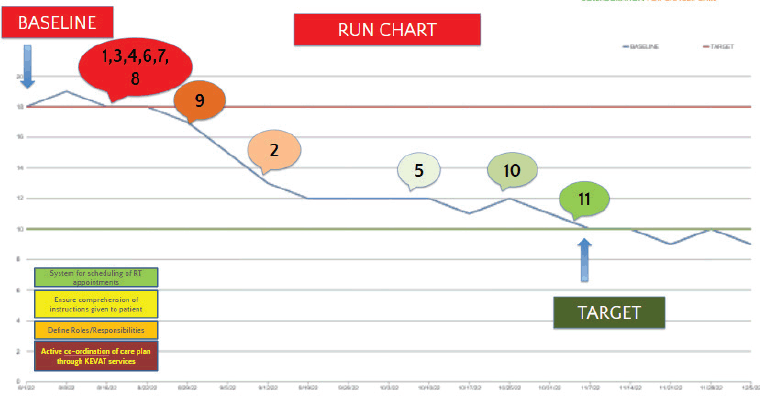
Figure 6. Run chart demonstrating weekly reduction in RT initiation delay with Annotated interventions. The red line marked the baseline average delay of 18 days, while the green line indicated the target of 10 days. Numeric annotations (e.g., 1, 3, 4, 6, 7 and 8) correspond to specific interventions introduced at various times. Larger circles like 9, 2, 5, 10 and 11 highlight critical intervention milestones. The blue trend line shows the progression of delay reduction over time.
Moreover, shorter waiting periods lower the need for repeat simulations, commonly necessitated by anatomical or tumour changes during prolonged delays. Re-simulations are not only resource intensive but also contribute to additional radiation exposure. One multi clinic study reported a median treatment delay of seven days due to re-simulation and may exacerbate patient anxiety during treatment planning [12]. In our cohort, reduced wait times coincided with lower re-simulation rates, allowing more patients to proceed with the original treatment plan. This is especially critical in resource-limited environments, where scanner availability and staff time are constrained. Avoiding unnecessary re-planning enhances clinical throughput and reduces indirect costs for both institutions and patients.
In addition to improved median wait times, we observed a shift in the entire distribution of waiting periods, with a larger proportion of patients initiating treatment within the desired 10-day timeframe. These improvements mirror results from similar QI initiatives. For example, Simons et al [19] achieved a 4-day reduction in median RT wait times (from 20.2 to 16.3 days) via Lean transformation in a Dutch center, with a notable drop in treatment breaches. In their setting, breaches for palliative RT beyond 10 days decreased from 35% to 16%, while breaches of the 28-day limit for curative RT dropped from 17% to 8% [19]. Likewise, Divi et al [20] reported success with a structured QI program to expedite adjuvant RT in oral cancer, increasing the proportion of patients initiating radiation within 6 weeks from 62% to 73% and significantly reducing avoidable delays [20].
Despite differing clinical and geographic settings, a common thread across these QI projects is the effectiveness of structured, methodical interventions in improving oncology care timelines. Our postintervention performance demonstrates that RT wait times are not immutable; they can be optimised through deliberate system redesign. The delay drivers may vary, from scheduling inefficiencies to communication lapses, but the diagnostic and improvement methodology remains universally applicable.
Our study leveraged the A3 Lean framework in conjunction with Fishbone analysis, Pareto prioritisation and iterative PDSA cycles. These tools provided a structured pathway for identifying root causes, testing change ideas and monitoring outcomes. The Fishbone diagram helped categorise delay factors across domains like human resources, equipment availability, procedural inefficiencies and patient-related issues. PDSA cycles allowed incremental, evidence-based adaptation of interventions. Staff feedback during PDSA cycles indicated improved workflow clarity and job satisfaction, particularly due to the resolution of previously stress-inducing bottlenecks.
Such agile cycles are widely recommended in healthcare QI efforts for their ability to accommodate local constraints and evolve with feedback. In line with previous work, including that of Divi et al [20], who explicitly employed the A3 Lean framework to mitigate delays in surgical to radiation transitions, our methodology proved adaptable and scalable in a rural LMIC setting.
Crucially, the interventions required minimal financial investment and no additional infrastructure. Many LMIC settings suffer not just from a lack of resources but from inefficient utilisation of existing capacity. Our process-oriented approach demonstrates that targeted workflow improvements can bridge some of these gaps. Moreover, this model can be extended across departments, for example, to improve time from biopsy to chemotherapy initiation, or to higher volume institutions with appropriate contextual adaptations.
Finally, reducing CT Sim to treatment intervals is not an isolated operational goal but part of a broader strategy to improve equity in cancer care. Timely initiation of therapy is a recognised quality metric that directly affects patient outcomes. In rural and underserved populations, reducing delays helps mitigate geographic and economic barriers, contributing to improved compliance, better experiences and potentially enhanced clinical outcomes. Our findings show that even in under-resourced contexts, QI methodologies can bring RT performance in line with international standards. This not only supports the mission of equitable cancer care but provides a replicable framework for other LMIC centers striving to optimise oncology workflows.
The sustained implementation of these interventions through 2024 demonstrates strong institutional buy-in and operational resilience. Continued improvements in key metrics, coupled with positive staff feedback across roles, reinforce the feasibility of long-term adoption and replication of this model in other high-volume, resource-constrained oncology setting.
Strengths
This study is the first to integrate the A3 methodology with complementary QI tools such as Gemba walks, Fishbone analysis, Pareto charts and PDSA cycles to systematically address simulation-to-treatment delays within a rural Indian cancer center. The interventions implemented were practical, resource-sensitive and easily reproducible in similar low-resource settings. Notably, the initiative led to measurable and sustainable improvements in both clinical outcomes, such as reduced waiting times and lower re-simulation rates, and operational performance, including enhanced workflow efficiency and positive staff feedback, underscoring its effectiveness and scalability.
Limitations
While the interventions yielded measurable improvements in workflow and simulation-to-treatment timelines, long-term patient outcomes such as local control and survival were not assessed and warrant future investigation. Formal statistical testing to compare pre and postintervention data was not performed, which limits causal inference. Also, the introduction of a centralised simulation scheduling system with daily caps may have modestly increased the interval between oncologist recommendation and simulation; however, this parameter was not formally evaluated in the present study. Additionally, patient satisfaction and psychological outcomes such as anxiety were not evaluated using validated assessment tools, representing another area for future QI.
Future directives
Future efforts should aim to validate the effectiveness and adaptability of QI interventions across a broader range of oncology centers, including high-volume urban institutions, to assess their scalability and generalisability. Also, it should prospectively assess how daily simulation caps influence both recommendation-to-simulation intervals and overall treatment initiation timelines, ensuring continued workflow optimisation. Incorporating formal statistical analyses and longitudinal monitoring of key clinical outcomes, such as survival, local control will enhance causal inference and provide stronger evidence for impact.
Additionally, evaluating patient-centered outcomes is crucial. A structured patient satisfaction survey, particularly focusing on components such as the KEVAT patient navigation services, should be conducted to capture feedback on workflow improvements, patient communication and perceived quality of care. Such feedback will offer valuable insights for iterative refinements and ensure that future interventions remain aligned with patient needs.
By coupling robust evaluation with system-level integration, these steps will help ensure the durability and continued evolution of our QI framework.
Conclusion
This study demonstrates that delays in initiating RT following CT sim, an often under-recognised barrier to timely cancer care, can be effectively mitigated through a structured, data-driven QI approach, even within the constraints of a rural healthcare setting. By integrating A3 problem-solving methodology with RCA tools (Fishbone and Pareto) and iterative PDSA cycles, we identified critical workflow inefficiencies and implemented resource-appropriate, scalable interventions. Targeted modifications in scheduling practices, role clarity, patient counselling and process checkpoints led to a 44.4% reduction in the median simulation-to-treatment interval (from 18 to 10 days), without adding burden to existing clinical staff. These improvements were accompanied by enhanced workflow efficiency, fewer re-simulations and improved patient adherence.
Importantly, this QI model offers a reproducible and adaptable framework for RT departments in low-resource settings, aiming to optimise treatment timeliness. Long-term sustainability will require continued process monitoring, integration into institutional protocols and periodic re-evaluation to align with evolving clinical demands. Ultimately, timely RT initiation should be viewed not only as an operational goal but as a key determinant of equitable, patient-centered oncology care. Our findings affirm that systemic delays are modifiable and that structured QI initiatives can yield clinically meaningful gains even in resource-limited environments.
List of abbreviations
A3, A3 Problem Solving Framework (Lean methodology); CTSIM, Computed Tomography Simulation; DMG, Disease Management Group; EQUIP, Enable Quality Improve Patient Care; HNSCC, Head and Neck Squamous Cell Carcinoma; IQR, Interquartile Range; JR, Junior Resident; KEVAT, Patient-navigation and support system of Tata Memorial Centre that assists patients with government schemes, financial documentation and logistical coordination; LMIC, Low and Middle Income Countries; PDSA, Plan-Do-Study-Act; PPA, Pre Planning Audit; QI, Quality Improvement; RCA, Root Cause Analysis; ROIS, Radiation Oncology Information System; RT, radiation therapy; RTT, Radiation Therapy Technologist; SOPs, Standard Operating Procedures; SR, Senior Resident; TPS, Treatment Planning System; TTI, Time to Treatment Initiation.
Acknowledgments
The authors gratefully acknowledge the support of the EQUIP India team, Stanford Medicine and the National Cancer Grid (NCG), India, for their technical guidance and mentorship throughout the quality improvement process. The authors also thank all participating clinical and administrative staff for their active involvement, constructive feedback and continued support in implementing and sustaining the interventions described in this study.
Conflicts of interest
The authors declare that they have no conflicts of interest relevant to the content of this article. No financial relationships, commercial affiliations or personal interests influenced the design, execution or reporting of this study.
Funding
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
1. Bernier J, Hall EJ, and Giaccia A (2004) Radiation oncology: a century of achievements Nat Rev Cancer 4(9) 737–747 https://doi.org/10.1038/nrc1451 PMID: 15343280
2. Bentzen SM (2006) Preventing or reducing late side effects of radiation therapy: radiobiology meets molecular pathology Nat Rev Cancer 6 702–713 https://doi.org/10.1038/nrc1950 PMID: 16929324
3. Das P, Cantor SB, and Parker CL, et al (2010) Long-term quality of life after radiotherapy for the treatment of anal cancer Cancer 116(4) 822–829 https://doi.org/10.1002/cncr.24906
4. Walter F, Webster A, and Scott S, et al (2012) The Andersen model of total patient delay: a systematic review of its application in cancer diagnosis J Health Serv Res Policy 17(2) 110–118 https://doi.org/10.1258/jhsrp.2011.010113 PMCID: 3336942
5. Bernier J, Domenge C, and Ozsahin M, et al (2004) Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer N Engl J Med 350(19) 1945–1952 https://doi.org/10.1056/NEJMoa032641 PMID: 15128894
6. Cooper JS, Pajak TF, and Forastiere AA, et al (2004) Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck N Engl J Med 350(19) 1937–1944 https://doi.org/10.1056/NEJMoa032646 PMID: 15128893
7. Žumer B, Pohar Perme M, and Jereb S, et al (2020) Impact of delays in radiotherapy of head and neck cancer on outcome Radiat Oncol 15(1) 202 https://doi.org/10.1186/s13014-020-01645-w PMID: 32819389 PMCID: 7441656
8. Huang J, Barbera L, and Brouwers M, et al (2003) Does delay in starting treatment affect the outcomes of radiotherapy? A systematic review J Clin Oncol 21(3) 555–563 https://doi.org/10.1200/JCO.2003.04.171 PMID: 12560449
9. Morrill KE, Robles-Morales R, and Lopez-Pentecost M, et al (2022) Factors associated with cancer treatment delay: a protocol for a systematic review and meta-analysis BMJ Open 12(6) 61121 https://doi.org/10.1136/bmjopen-2022-061121
10. Hanna TP, King WD, and Thibodeau S, et al (2020) Mortality due to cancer treatment delay: systematic review and meta-analysis BMJ 371 4087 https://doi.org/10.1136/bmj.m4087
11. Bleicher RJ, Ruth K, and Sigurdson ER, et al (2016) Time to surgery and breast cancer survival in the United States JAMA Oncol 2(3) 330–339 https://doi.org/10.1001/jamaoncol.2015.4508
12. Metzger A, Renz P, and Hasan S, et al (2019) Unforeseen computed tomography resimulation for initial radiation planning: associated factors and clinical impact Adv Radiat Oncol 4(4) 716–721 https://doi.org/10.1016/j.adro.2019.06.002 PMID: 31673665 PMCID: 6817516
13. Xu Z, Lee S, and Albani D, et al (2019) Evaluating radiotherapy treatment delay using failure mode and effects analysis (FMEA) Radiother Oncol 137 102–109 https://doi.org/10.1016/j.radonc.2019.04.016 PMID: 31082631
14. Graboyes EM, Kompelli AR, and Neskey DM, et al (2019) Association between treatment delays and oncologic outcomes in head and neck cancer: a systematic review Head Neck 41(2) 572–584 https://doi.org/10.1002/hed.25409
15. Murphy CT, Galloway TJ, and Handorf EA, et al (2017) Adherence to NCCN guidelines for the time interval between surgery and postoperative radiation therapy in head and neck cancer Cancer 123(14) 2651–2660 [10.1002/cncr.30672] https://doi.org/10.1002/cncr.30651
16. Vitzthum L, Yuan J, and Jones D, et al (2019) Reducing prolonged chemoradiation treatment times for cervical cancer BMJ Open Qual 8(3) 516 https://doi.org/10.1136/bmjoq-2018-000516
17. Halder P, Dixit J, and Gupta N, et al (2024) Access to timely cancer treatment initiation in India: extent, determinants and trends Lancet Regional Health Southeast Asia 32 100514 https://doi.org/10.1016/j.lansea.2024.100514
18. Gangopadhyay A (2022) Radiotherapy travel times – is time running out in India? Ecancermedicalscience 16 ed122 https://doi.org/10.3332/ecancer.2022.ed122
19. Simons P, Backes H, and Bergs J, et al (2017) The effects of a lean transition on process times, patients and employees Int J Health Care Qual Assur 30(2) 103–118 https://doi.org/10.1108/IJHCQA-08-2015-0106 PMID: 28256930
20. Divi V, Chen MM, and Hara W, et al (2018) Implementing a standardized care pathway for head and neck cancer patients: improving timeliness of care Otolaryngol Head Neck Surg 158(6) 1026–1032 https://doi.org/10.1177/0194599818768254





