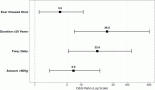
This study aims to determine the current state of clinical trials regarding HPV-positive head and neck squamous cell carcinoma (HNSCC). Clinical trials were filtered to fit the study’s aim using Clinicaltrials.gov: trials concerning HNSCC specifically those related to HPV done between January 2005 and December 2020 were extracted and information regarding location, duration, phases, patient recruitment, trial status, results, primary outcome, type of intervention and publication status were collected and analysed. As a result, 123 trials were included. North American countries (USA and Canada) conducted more than two-thirds of the trials (72.4%) compared to European countries and the rest of the world. Trials in phase II constituted more than half of those included in this study (53.7%). From the 123 trials included in this study, only 30 had their NCT identification number linked to publications, but less than half (46.7%) of the publications stemmed from trials with results. Drug combination was the most widely studied treatment modality. Despite falling in the middle of the spectrum with respect to the number of trials when compared to other diseases, our research highlights the need for even more trials tackling multiple aspects of HPV-positive HNSCC.