Melding the genetic and cellular analysis of tumours with how they appear in medical images could give physicians and other cancer therapy specialists new insights into how to best treat patients, especially those with brain cancer, according to a new study led by the Translational Genomics Research Institute (TGen), an affiliate of City of Hope.
Published in the scientific journal PLOS ONE, this study suggests that the tumour microenvironment -- essentially all the cells both in and surrounding a tumour -- play a vastly under-studied role in the development and growth of cancer.
"This study is a bridge between genetic sequencing, single-cell analysis and high-resolution medical imaging," said Dr. Michael Berens, Professor and Director of TGen's Cancer and Cell Biology Division, head of the institute's Glioma Research Lab, and one of the study's lead authors.
"By literally focusing on how tumours look on the outside, as well as spelling out their DNA cell characteristics on the inside, we believe we can provide physicians, oncologists, radiologists, surgeons and others with timely information about how to best attack each patient's cancer."
This type of analysis is especially needed for brain cancer patients, whose fast-growing tumours are among the most difficult to diagnose, treat, remove and monitor.
Because of the way brain tumours infiltrate surrounding healthy brain tissue, it is difficult for surgeons to remove all of the cancer without causing potentially catastrophic damage to a patient's memory and ability to function.
And because of a matrix of tiny blood vessels that surround and protect the brain, only the smallest molecules can enter, limiting the types of drugs that could help shrink brain tumours.
Adding to the difficulty is the fact that each patient's brain tumour cells are different. Even the individual cancer cells within the tumour can vary.
"(The) diverse cellular composition and cellular interactions have not been well characterized," according to the study.
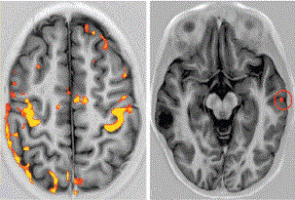
To better understand these cancers, Dr. Berens and others correlated the genetic and protein fingerprints of brain cancer cells with how those cells, and surrounding cells looked, using MRI (magnetic resonance imaging), a test that is routinely performed as soon as brain cancer is suspected.
Drawback of DNA analysis without imaging
Currently, to characterise tumours at the molecular level, scientists commonly grind up many cells from a biopsy and extract DNA, RNA and other genomic materials so that the tumour can be sequenced and researchers can tell what genes, or set of genes, might be misbehaving to cause the cancer.
But they can't tell how those cancer cells may have interacted with other nearby cells.
The spatial and cellular context is lost.
Even the new revolution of single-cell sequencing leaves researchers with no context as to what cells were adjacent to the individual diseased cells they analyse.
"The resolution of MRI can't 'see' individual cell differences. But we were able to find evidence for correlations between genetic and cellular changes. We can see the consequences of specific genetic changes in brain cancer tumours that show up on a medical image," Dr. Berens said.
This type of information could potentially help surgeons decide how much tissue must be removed to extract the cancer, the dosage and frequency a radiologist might use to treat the cancer, and what specific drugs might be best suited for each patient at differing points in time.
It could help answer other questions.
For example: How is the tumour infiltrating adjacent tissue?
How is it interfering with the body's immune system?
How is it generating new adjacent blood vessels to obtain a surge of nutrients to keep it growing?
Using fluorescent tags to identify biomarkers
To help answer these questions, TGen's partnering investigators at General Electric Global Research Center deployed a novel imaging tool, which they developed over the past decade together with GE Healthcare, called "Cell DIVE™," or Multiplexed immunofluorescence imaging (MxIF), which is used to repeatedly stain tumour samples with antibodies attached to fluorescent dyes.
In an iterative process of staining and imaging, the method allows cell level quantification of over 60 cell biomarkers in a single sample.
In this study, researchers analysed more than 100,000 cells in brain tumour cases, using MxIF to uncover differences between two types of brain tumours based on mutations in the gene IDH1.
"Using this platform, we can visualise and analyse various cell types and cell states present in the tumour tissue as well as how they interact with each other and their microenvironment," said Dr. Anup Sood, a senior scientist at GE Global Research, and also a lead author of the study.
"Visualising the microenvironment, especially, is key to understanding tumour behaviour and the response to therapy, which has been difficult to analyse with conventional methods," Dr. Sood said.
"The platform's unique capabilities, which allows deeper insights into cancer, were the result of a more than decade's long effort by a multidisciplinary team of more than 50 chemists, biologists, software and hardware engineers, computer scientists, statisticians with key industrial and academic partners."
Dr. Fiona Ginty, another GE senior scientist and the study's senior author, added: "The more cell-level data we analyse, the more we learn about tumour biology, cell-to-cell interactions, immune response and how tumours progress. Further, with the integration of cellular, medical imaging and genomic data, we gain a more holistic understanding of why certain tumour types progress more rapidly, and others are more slow-growing, and ultimately which drugs a patient may respond to."
Researchers' next step will be to use this new technology on a large cohort of patients to prove that it works.
We are an independent charity and are not backed by a large company or society. We raise every penny ourselves to improve the standards of cancer care through education. You can help us continue our work to address inequalities in cancer care by making a donation.
Any donation, however small, contributes directly towards the costs of creating and sharing free oncology education.
Together we can get better outcomes for patients by tackling global inequalities in access to the results of cancer research.
Thank you for your support.