A hospital-based study of survival in colon cancer patients of Tata Memorial Hospital (Mumbai)
Tushar Ambekar1,2,3, Amey Oak1,2,3, Sivaranjini Kannusamy1,2,3, Sandhya Cheulkar1,2,3, Riya R John1,2,3, Shalmali Chavan1,2,3, Saket Mendhe1,2,3, Supriya Kadam1,2,3, Mamta Lade1,2,3, Ganesh Balasubramanium2,3, Ashwin Desouza1,2,3, Avanish Saklani1,2,3, Rajesh Dikshit1,2,3, Pankaj Chaturvedi1,2,3 and Sudeep Gupta1,2,3,4
1Division of Cancer Care, HBCR and Survival Studies, Centre for Cancer Epidemiology (CCE)
2Centre for Cancer Epidemiology (CCE)
3ACTREC, Tata Memorial Centre, Homi Bhabha National Institute (HBNI), Navi Mumbai 400094, India
4Tata Memorial Centre, Homi Bhabha National Institute (HBNI), Mumbai 400094, India
Abstract
Introduction: Colon cancer, a type of cancer affecting the large intestine, is the fourth most common cancer worldwide, with 1,142,286 new cases and the fifth leading cause of cancer death, with 538,167 deaths reported in 2022 (GLOBOCAN). This retrospective study examines overall survival (OS) and the impact of sociodemographic and clinical factors on colon cancer patients.
Methodology: The study included colon cancer patients diagnosed and treated at Tata Memorial Hospital, Mumbai, between January and December 2017, with a 5-year follow-up until 2022. Of the 761 cases registered, 434 received treatment and were eligible for analysis. OS and its variation by socio-demographics were assessed using the Kaplan-Meier method and log-rank test, while the Cox proportional hazard model variables indicate the impact of multiple factors on survival.
Results: Among 434 patients (mean age 50 ± 13.6 years, 64% male), adenocarcinoma was the predominant histology (81.7%), with 98% microscopically confirmed diagnoses. Regional and distant disease were present in 32.9% and 23.5%, respectively. The 1-, 3- and 5-year OS rates were 88%, 77% and 71%. Survival was significantly higher in early-stage disease than advanced disease (5-year OS: 89% versus 43%). On multivariable analysis, advanced clinical extent (aHR 7.42; 95% CI: 3.67–14.9) and signet ring cell histology (aHR 1.95; 95% CI: 1.06–3.59) were independently associated with increased mortality, while combined-modality treatment was independently protective (aHR 0.48; 95% CI: 0.25–0.91).
Conclusion: The study suggests that improving socioeconomic conditions and encouraging proactive treatment-seeking behaviour are key to increasing colon cancer survival. Cancer hospitals, together with India’s public healthcare system, including clinicians and policymakers, should implement these strategies to strengthen cancer care in low- and middle-income countries.
Keywords: colon cancer, sociodemographic factors, clinical factors, registry, survival, India
Correspondence to: Amey Oak
Email: ameyoak55@gmail.com
Published: 13/05/2026
Received: 17/09/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
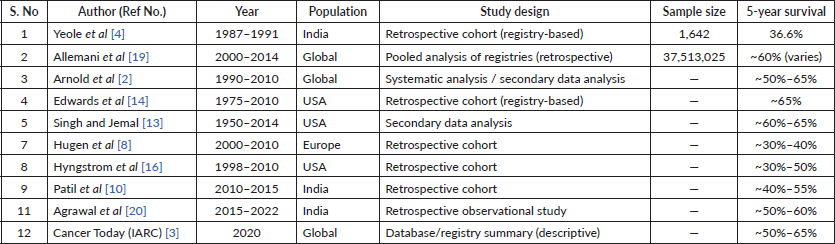
Colon cancer is an important contributor to global burden and remains a significant cause of cancer-related morbidity and mortality. According to global cancer estimates, colon cancer ranks among the leading malignancies worldwide and continues to demonstrate rising incidence in several developing regions. The increasing burden of colon cancer has been linked to urbanisation, dietary transitions characterised by high consumption of processed food and red meat, sedentary lifestyle, obesity and increasing life expectancy [1, 2]. According to GLOBOCAN 2022 estimates, colon cancer incidence ranks fourth with 1,142,286 cases and 10.7 age standardised rate (ASR), while in terms of mortality, it ranks fifth with 538,167 deaths and 4.7 ASR globally, whereas it ranks ninth with 34,046 cases in terms of incidence with 2.4 ASR, 20,629 cases while in terms of mortality, with 1.5 ASR in India [3].
In India, colon cancer incidence has shown a gradual increase in over recent decades, particularly in Urban populations. Although the incidence remains lower than that reported in Western countries, Survival outcomes remain suboptimal. Population-based cancer registry data from India have reported 5-year survival rates of colon cancer ranging between approximately 30%–40%, which is considerably lower than survival rates reported in high-income countries [4, 5]. The disparity in survival outcomes is attributable to multiple factors, including delayed diagnosis, limited awareness of early symptoms, lack of organised screening programs and unequal access to specialised oncology services [6].
Stage at diagnosis is the most important determinant of colon cancer prognosis. Early-stage colon is associated with favourable outcomes and high curative potential, primarily through surgical resection. In contrast, advanced-stage disease is associated with increased recurrence and mortality [7]. Apart from stage, several pathological characteristics influence survival outcomes. Tumour differentiation and histological subtype are recognised prognostic indicators, with aggressive variants such as signet ring cell carcinoma demonstrating poorer survival due to advanced stage at presentation and aggressive biological behaviour [8].
Treatment plays a critical role in determining survival outcomes in colon cancer. Surgical resection remains the cornerstone of curative management, while adjuvant chemotherapy has been shown to significantly improve survival in patients with high-risk disease and advanced stages. Multimodality treatment approaches have been associated with improved disease control and overall survival (OS) [9].
Despite the increasing burden of colon cancers in India, there remains limited evidence describing survival outcomes in prognostic factors specifically among colon cancer patients treated in tertiary care settings. Evaluating survival patterns and associated determinants is essential for identifying gaps in cancer care and improving treatment strategies. Therefore, the present study was undertaken to assess survival outcomes among colon cancer patients and to identify demographic, clinical, pathological and treatment related predictors of OS in a tertiary care centre, Tata Memorial Hospital (TMH), Mumbai, one of India’s cancer institutes which offers cancer diagnosis, treatment, education and research with access to a variety of cancer-related studies, treatments and patient follow-ups. TMH has a system of digital medical records that provides numerous research opportunities. This study will utilise data available in the hospital medical records on colon cancer from the year 2017 to 2022, with a follow-up period of 5 years, for the patients registered in the year 2017 at TMH, Mumbai.
Materials and methods
This study was conducted at TMH, Mumbai. It included all colon cancer patients who were registered and received treatment at TMH between 1 January 2017, and 31 December 2017. The study was based on a review of hospital records from the TMH Cancer Registry. Data were collected using a detailed questionnaire with three sections: sociodemographic details, clinical information and follow-up. The following information was recorded: age, gender, region, occupation, education (illiterate = unable to read or write), marital status, income, tumour site, tumour type, grade, stage, type of case (new patients treated at TMH or old patients treated elsewhere before coming to TMH) and type of treatment. Colon cancer is categorised into Caecum, Appendix, Ascending colon (Right colon), Hepatic flexure, Transverse colon, Splenic flexure, Descending colon (Left colon) Sigmoid colon (Sigmoid, NOS; Sigmoid flexure; Pelvic colon), Overlapping lesions, Colon, NOS; Large intestine; Large bowel, NOS, Rectosigmoid junction, Rectosigmoid, NOS Rectosigmoid, Colon and rectum Pelvic-rectal junction. Surgery, radiation, chemotherapy or combined therapy, which is a combination of both therapies, have all been considered in the treatment analysis. Patients were followed up every 6 months. The follow-up period lasted from January 2017 to December 2022. Follow-up data were collected through the electronic medical record system and phone calls. OS was defined as the time from diagnosis to death or last follow-up. Five-year OS was calculated for patients diagnosed between January 2017 and December 2022. The study examined how different sociodemographic and clinical factors affected survival. All data were kept confidential. Data entry was done using in-house software, and analysis was performed using STATA version 15.0. Results were presented using means, standard deviations, medians, ranges, proportions and 95% confidence intervals. Survival was analysed using Kaplan–Meier and log-rank tests. The Cox proportional hazards model was used to assess the effect of multiple factors. A p-value of less than 0.05 was considered statistically significant. R software version 4.52 was used to generate Kaplan–Meier graphs.
Results
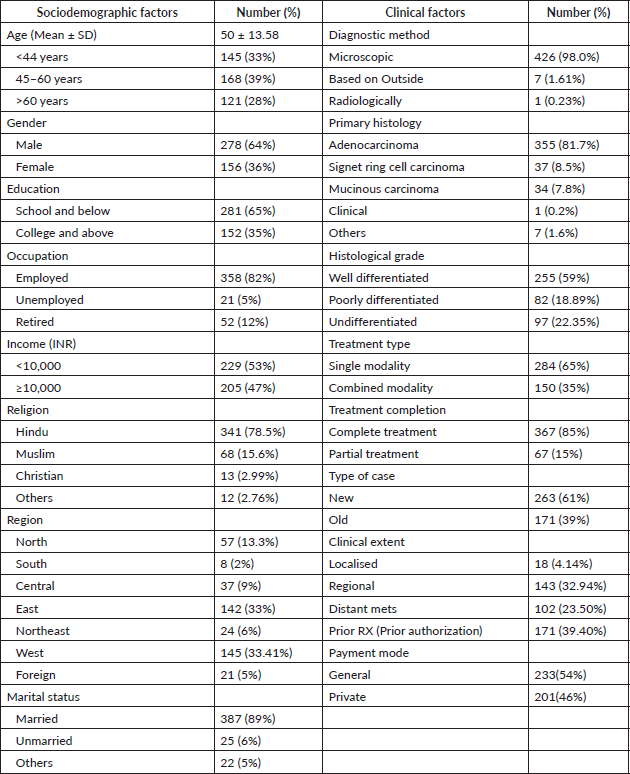
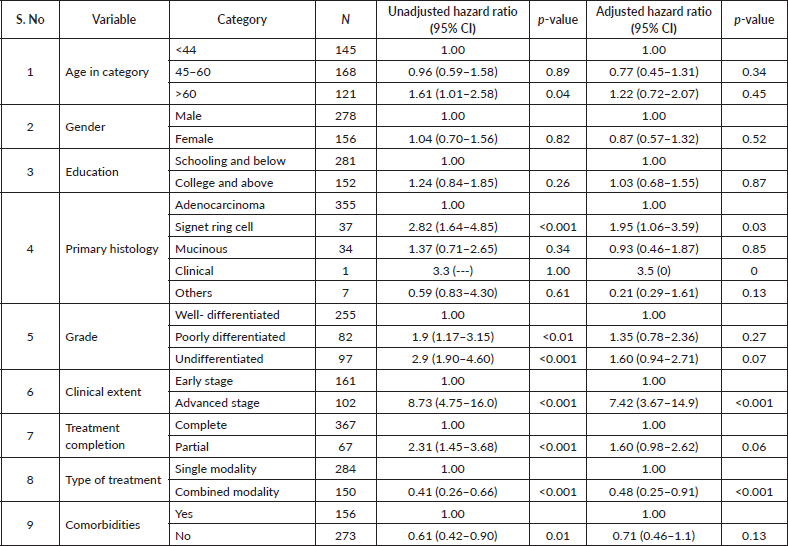
The mean age was 50 ± 13.58 years; 39% (168) were aged 45–60 years and 64% (278) were male. Most patients had education up to school level (65%, 281), were employed (82%, 358), had a monthly income <INR 10,000 (53%, 229) and were married (89%, 387). Patients predominantly originated from the West (33.4%, 145) and East (33%, 142) regions. Diagnosis was confirmed microscopically in 98% (426) of cases. Adenocarcinoma was the most common histology (81.7%, 355). Tumours were well differentiated in 59% (255), while 22.4% (97) were undifferentiated. Single-modality treatment was administered to 65% (284), with 85% (367) completing treatment. New cases comprised 61% (263). At presentation, 32.9% (143) had regional disease and 23.5% (102) had distant metastases (Table 1).
Table 2 and Figure 1 show the survival results. The overall 1-, 3- and 5-year OS rates were 88%, 77% and 71%, respectively (n = 434). Five-year OS by age was 75% (<44 years), 77% (45–60 years) and 58% (>60 years) (p = 0.04), with no significant gender difference (71% in both males and females; p = 0.82). Patients with early-stage disease had markedly superior survival (1-, 3-, 5-year OS: 97%, 93%, 89%) compared with those with advanced disease (73%, 50%, 43%; p < 0.001). By histology, adenocarcinoma showed 1-, 3- and 5-year OS of 89%, 80% and 74%, signet ring cell carcinoma 68%, 42% and 42%, and mucinous carcinoma 87%, 64% and 64% (p = 0.002). Survival declined with poorer differentiation (well-differentiated: 90%, 85%, 79%; poorly differentiated: 86%, 68%, 66%; undifferentiated: 81%, 53%, 51%; p < 0.001). Patients receiving combined-modality treatment had higher survival (93%, 85%, 81%) than those receiving single-modality treatment (84%, 71%, 64%; p = 0.001). Treatment completion was associated with improved outcomes (89%, 80%, 74% versus 77%, 50%, 50%; p < 0.001). Patients without comorbidities had better survival (89%, 79%, 75%) compared with those with comorbidities (83%, 71%, 63%; p = 0.02). The Kaplan–Meier graphs in Figure 1 show the graphical representation of significant variables.
On unadjusted analysis, age >60 years was associated with higher mortality (HR 1.61; p = 0.04), but this was not significant after adjustment. Gender and education showed no association with survival. Signet ring cell carcinoma remained an independent predictor of mortality (aHR 1.95; p = 0.03), while mucinous histology was not significant. Higher tumour grade was associated with poorer survival on unadjusted analysis only. Advanced clinical extent was the strongest independent predictor (aHR 7.42; p < 0.001). Combined-modality treatment was independently protective (aHR 0.48; p < 0.001). Treatment completion and comorbidities were not independently associated with survival after adjustment (Table 3).
Discussion
The present study evaluated the survival outcomes among the patients diagnosed with colon cancer and identified key demographic, clinical and treatment-related predictors influencing prognosis. Our findings highlight that survival in colon cancer is strongly influenced by stage at diagnosis, treatment modalities and patient-related sociodemographic and lifestyle factor which is consistent with global and Indian literature. In our study, of the 434 patients, 24% died, while 31% were categorised as censored. The overall 1-, 3- and 5- year survival rates in our study were 88%, 77% and 71%, respectively, which appear higher than the previously reported population-based survival estimates from India. Earlier registry-based analysis have reported 5-year survival estimates for colorectal cancer ranging from 34% to 42%, reflecting poor outcomes in the general population compared to specialised tertiary centres [4 ,5]. The relatively favourable survival observed in our cohort may be attributed to improved diagnostic facilities, availability of multimodality treatment and treatment adherence in a tertiary cancer care setting.
Table 1. Sociodemographic and clinical characteristics of study participants (N = 434).
In our study, the mean age at diagnosis was 50 years, which is lower than that reported ion western populations where colorectal cancer typically occurs after 60 years of age. Similar trends of younger age at presentation have been documented in Indian studies, which may reflect demographic differences, genetic susceptibility, lifestyle transitions and changing epidemiological patterns [10, 11]. Although older age (>60 years) was associated with increased mortality in the unadjusted hazard ratio, the association was not significant after adjustment, suggesting that treatment and stage at diagnosis may play a more critical role in determining survival than age alone. Gender did not demonstrate a significant association with survival in our study. While some international studies have reported slightly better survival among females, Indian studies have shown inconsistent findings likely due to sociocultural differences, variations in healthcare utilisation and biological heterogeneity [10, 12].
The demographic profile of our patients showed that the majority belonged to lower socioeconomic strata, with low income and educational levels. Socioeconomic status has been recognised as an important determinant of cancer outcomes, influencing health-seeking behaviour, access to diagnostic services and treatment adherence [13]. In India, disparities in healthcare infrastructure and financial constraints often contribute to delays in diagnosis and treatment, thereby adversely affecting survival outcomes [6]. Presence of comorbidities was associated with poor survival initially but was not independently predictive after adjustment. Previous studies have reported that comorbid conditions may influence treatment decisions, therapy tolerance and overall prognosis, particularly among elderly patients [14, 15].
Histological subtype significantly influenced survival outcomes in our cohort. Signet ring cell carcinoma was independently associated with increased mortality risk, whereas mucinous carcinoma was not significant after adjustment. Previous studies have demonstrated that signet ring cell carcinoma is associated with aggressive biological behaviour, advanced stage at presentation and poor response to conventional therapies, leading to inferior survival outcomes [16, 17]. Additionally, we observed declining survival with poor tumour differentiation, which is consistent with established evidence linking high-grade tumours with aggressive disease and unfavourable prognosis [18]. Stage at diagnosis emerged as the strongest independent predictor of survival in our study, with advanced-stage disease showing significantly higher mortality risk. Patients with early-stage disease demonstrated markedly superior survival compared to those with global and Indian evidence, highlighting that late-stage presentation is a major contributor to poor colorectal cancer outcomes in low- and middle-income countries [5, 19]. In India, the absence of organised population-based screening programs and limited awareness regarding early symptoms often result in diagnosis at advanced stages [20].
Table 2. OS based on sociodemographic and clinical characteristics.
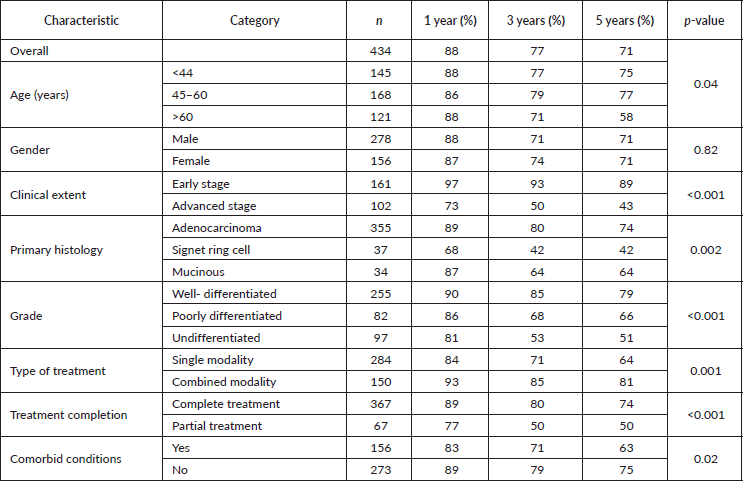
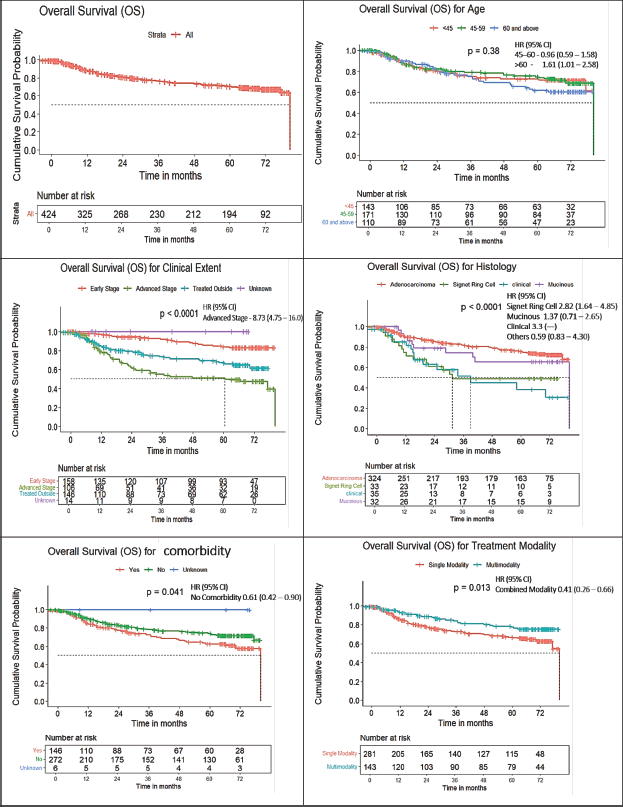
Figure 1. Five-year OS, survival based on the age, clinical extent, histology, comorbidity and type of treatment of colon cancer patients at TMH Mumbai, in 2017 (N = 434).
Table 3. Unadjusted and adjusted hazard ratios for OS.
Treatment-related factors played a crucial role in survival outcomes. Patients receiving combined modality treatment demonstrated significantly improved survival and remained independently protective after adjustment. Multimodal therapy, including surgery combined with chemotherapy and/or radiotherapy, has been shown to improve survival by reducing recurrence and improving disease control, particularly in advanced-stage disease [7, 9]. These findings underscore the importance of multidisciplinary cancer management and timely initiation of appropriate therapy. Although treatment completion was associated with improved survival on unadjusted analysis, it did not remain independently significant after adjustment, suggesting that treatment modality and stage at diagnosis may have a stronger influence on outcomes.
The strengths of our study include a relatively large cohort, comprehensive survival analysis and evaluation of multiple demographic, clinical and treatment-related predictors. However, certain limitations should be acknowledged. Being a retrospective hospital-based study, the findings may not be fully generalisable to the population level. Additionally, data on molecular markers, lifestyle factors and quality of life outcomes were limited. Future multicentric prospective studies incorporating molecular profiling and survivorship outcomes are required for a better understanding of the disease biology and to optimise treatment strategies in the Indian population. Several studies that were referenced are mentioned in Supplementary Table 1.
Overall, our findings emphasise the critical importance of early detection, timely initiation of multimodality treatment and strengthening cancer care infrastructure to improve colon cancer survival outcomes in India.
Conclusion and recommendations
Colon cancer survival in this tertiary care cohort was relatively high, with a 5-year OS of 71%. Advanced stage at diagnosis and signet ring cell histology were significant predictors of poor survival, while multimodality treatment independently improved outcomes.
The study highlights the critical influence of stage at diagnosis and tumour biology on survival outcomes. It provides evidence that survival can be substantially improved in resource-limited settings when patients receive comprehensive, multidisciplinary treatment. The findings also suggest that the demographic and socioeconomic factors may indirectly influence outcomes through their impact on stage at presentation and treatment.
Strengthening early detection strategies, promoting awareness of symptoms and ensuring timely access to multidisciplinary treatment are essential to improve survival outcomes. Expanding screening initiatives and improving treatment accessibility, particularly for advanced disease, should be in the Indian healthcare setting.
Conflicts of interest
The authors have no relevant financial or non-financial interests to disclose.
Funding
The authors declare that no funds, grants or other support were received during the preparation of this manuscript.
Reference
1. Sung H, Ferlay J, and Siegel RL, et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [Internet] CA Cancer J For Clin 2026 [https://acsjournals.onlinelibrary.wiley.com/doi/10.3322/caac.21660]
2. Arnold M, Sierra MS, and Laversanne M, et al (2017) Global patterns and trends in colorectal cancer incidence and mortality Gut 66(4) 683–691 https://doi.org/10.1136/gutjnl-2015-310912
3. Cancer Today [Internet] [https://gco.iarc.who.int/today/] Date accessed: 16/04/26
4. Yeole BB, Sunny L, and Swaminathan R, et al (2001) Population-based survival from colorectal cancer in Mumbai, (Bombay) India Eur J Cancer 37(11) 1402–1408 https://doi.org/10.1016/S0959-8049(01)00108-3 PMID: 11435072
5. Shivshankar S, Patil PS, and Deodhar K, et al (2025) Epidemiology of colorectal cancer: a review with special emphasis on India Indian J Gastroenterol 44(2) 142–153 PMID: 39928255 PMCID: 11953156
6. Mallath MK, Taylor DG, and Badwe RA, et al (2014) The growing burden of cancer in India: epidemiology and social context Lancet Oncol 15(6) e205–e212 https://doi.org/10.1016/S1470-2045(14)70115-9 PMID: 24731885
7. Benson AB, Venook AP, and Al-Hawary MM, et al (2021) Colon cancer, version 2.2021, NCCN clinical practice guidelines in oncology J Natl Compr Canc Netw 19(3) 329–359 [https://doi.org/10.6004/jnccn.2021.0012]
8. Hugen N, Verhoeven RH, and Lemmens VE, et al (2015) Colorectal signet-ring cell carcinoma: benefit from adjuvant chemotherapy but a poor prognostic factor Int J Cancer 136(2) 333–339 https://doi.org/10.1002/ijc.28981
9. Dekker E, Tanis PJ, and Vleugels JLA, et al (2019) Colorectal cancer Lancet 394(10207) 1467–1480 https://doi.org/10.1016/S0140-6736(19)32319-0 PMID: 31631858
10. Patil PS, Saklani A, and Gambhire P, et al (2017) Colorectal cancer in India: an audit from a Tertiary Center in a Low Prevalence Area Indian J Surg Oncol 8(4) 484–490 https://doi.org/10.1007/s13193-017-0655-0 PMID: 29203978 PMCID: 5705504
11. Lavingia V and Gore AA (2021) Time for colorectal cancer screening in India! Indian J Cancer 58(3) 315–316 [https://doi.org/10.4103/ijc.ijc_918_21]
12. Brenner H, Kloor M, and Pox CP (2014) Colorectal cancer Lancet 383(9927) 1490–1502 https://doi.org/10.1016/S0140-6736(13)61649-9
13. Singh GK and Jemal A (2017) Socioeconomic and racial/ethnic disparities in cancer mortality, incidence, and survival in the United States, 1950–2014: over six decades of changing patterns and widening inequalities J Environ Public Health 2017 2819372 https://doi.org/10.1155/2017/2819372
14. Edwards BK, Noone AM, and Mariotto AB, et al (2014) Annual report to the nation on the status of cancer, 1975-2010, featuring prevalence of comorbidity and impact on survival among persons with lung, colorectal, breast or prostate cancer Cancer 120(9) 1290–1314 https://doi.org/10.1002/cncr.28509
15. Sarfati D, Koczwara B, and Jackson C The impact of comorbidity on cancer and its treatment [Internet] [https://doi.org/10.3322/caac.21342]
16. Hyngstrom JR, Hu CY, and Xing Y, et al (2012) Clinicopathology and outcomes for mucinous and signet ring colorectal adenocarcinoma: analysis from the national cancer data base Ann Surg Oncol 19(9) 2814–2821 https://doi.org/10.1245/s10434-012-2321-7 PMID: 22476818 PMCID: 3688065
17. Hugen N, Verhoeven RH, and Lemmens VE, et al (2015) Colorectal signet‐ring cell carcinoma: benefit from adjuvant chemotherapy but a poor prognostic factor [Internet] Int J Cancer 2026 https://doi.org/10.1002/ijc.28981
18. Compton CC (2003) Colorectal carcinoma: diagnostic, prognostic, and molecular features Mod Pathol 16(4) 376–388 https://doi.org/10.1097/01.MP.0000062859.46942.93 PMID: 12692203
19. Allemani C, Matsuda T, and Di Carlo V, et al (2018) Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries Lancet 391(10125) 1023–1075 https://doi.org/10.1016/S0140-6736(17)33326-3 PMID: 29395269 PMCID: 5879496
20. Agrawal M, Surendran AK, and Venkatesh KK, et al (2025) Screening for colorectal carcinoma in india: real-world scenario, pitfalls, and solutions South Asian J Cancer 13(4) 229–235 PMID: 40060340 PMCID: 11888814
Supplementary information
Supplementary Table 1. Colon cancer survival rates from published studies.